Abstract
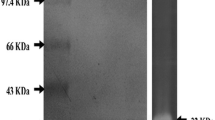
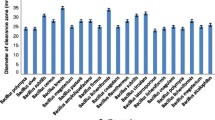
The study was carried out to purify protease produced by Bacillus subtilis ATCC 6633 strain and to examine the effect of the crude saponin extract (CSE) obtained from Gypsophila pilulifera Boiss. & Heldr. on the purified enzyme. In addition, the present study dealt with the evaluation of antioxidant and DNA cleavage potentials of the extract. Protease from B. subtilis ATCC 6633 was produced and purified using ammonium sulfate precipitation, gel filtration chromatography with 27.2-fold. The purified enzyme showed a single band on SDS-PAGE, and it was determined that its molecular weight was 30.1 kDa. In addition, to examine the effect of saponin as natural surfactant on the protease activity, crude saponin from G. pilulifera was extracted in methanol and the purified protease was incubated with the extract. It was observed that the activity of enzyme increased in the presence of extract. In the in vitro assays, while total phenolic content of the extract was determined by Folin–Ciocalteau method, antioxidant properties were evaluated by DPPH (1,1-diphenyl-2-picryl-hydrazyl) free radical scavenging activity, FRAP (ferric-ion reducing antioxidant parameter), CUPRAC (cupric ion reducing antioxidant capacity) and β-caroten-linoleic acid method. The results obtained in this study showed that the extract possesses low antioxidant activity for studied all in vitro models. Also, effect of the extract on plasmid DNA was determined with agarose gel electrophoresis. The extract caused conversion of supercoiled DNA structure to other forms. As a result, the extract exhibited DNA cleavage activity as dose-dependent.


Similar content being viewed by others
References
Adinarayana K, Ellaiah P, Siva Prasad D (2003) Purification and partial characterization of thermostable serine alkaline protease from a newly isolated Bacillus subtilis PE-11. AAPS Pharmscitech 4(4):1–9
Apak R, Güçlü K, Özyürek M, Karademir SE (2004) Novel total antioxidant index for dietary polyphenols and vitamins C and E, using their cupric ion reducing capability in the presence of neocuproine CUPRAC. Method J Agric Food Chem 52:7970–7981
Arslan I, Celik A, Chol JH (2012) A cytotoxic triterpenoid saponin from under-ground parts of Gypsophila pilulifera Boiss. & Heldr. Fitoterapia 83(4):699–703
Beg KQ, Gupta R (2003) Purification and characterization of an oxidation-stable, thiol-dependent serine alkaline protease from Bacillus mojavensi. Enzym Microb Technol 32:294–304
Castanon M, Wilke CR (1981) Effects of the surfactant tween 80 on enzymatic hydrolysis of newspaper. Biotechnol Bioeng 23:1365–1372
Cheeke PR (2000) Actual and potential applications of Yucca schidigera and Quillaja saponaria saponins in human and animal nutrition. J Anim Sci 77:1–10
Chen YF, Roan HY, Lii CK, Huang YC, Wang TS (2011) Relationship between antioxidant and antiglycation ability of saponins, polyphenols, and polysaccharides in Chinese herbal medicines used to treat diabetes. J Med Plants Res 5(11):2322–2331
Cupp-Enyard C (2008) Sigma’s non-specific protease activity assay-casein as a substrate. J Vis Exp 19:e899. https://doi.org/10.3791/899
Dias DR, Vilela DM, Silvestre MPC, Schwan RF (2008) Alkaline protease from Bacillus sp. isolated from coffee bean grown on cheese whey. World J Microbiol Biotechnol 24:2027–2034
Espósito TS, Amaral IPG, Marcuschi M, Carvalho JR, Bezerra RS (2009) Surfactants and oxidants-resistant-alkaline proteases from common carp (Cyprınus carpio L.) Processing. Waste J Food Biochem 33:821–834
Fidan AF, Dundar Y (2008) The effects of Yucca schidigera and Quillaja saponaria on DNA damage, protein oxidation, lipid peroxidation, and some biochemical parameters in streptozotocin-induced diabetic rats. J Diabetes Complicat 22(5):348–356
Fujiwara N, Masui A, Imanaka T (1993) Purification and properties of the highly thermostable alkaline protease from an alkaliphilic and thermophilic Bacillus sp. J Biotechnol 30(2):245–256
Gevrenova R, Stancheva T, Voynikov Y, Laurain-Mattar D, Henry M (2010) Root in vitro cultures of six Gypsophila species and their saponin contents. Enzym Microb Technol 47(3):97–104
Gupta R, Gupta K, Saxena RK, Khan S (1999) Bleach-stable, alkaline protease from Bacillus sp. Biotechnol Lett 21:135–138
Helle SS, Duff SJB, Cooper DG (1993) Effect of surfactants on cellulose hydrolysis. Biotechnol Bioeng 42:611–617
Huong NTT, Matsumoto K, Kasai R, Yamasaki K, Watanabe H (1998) In vitro antioxidant activity of vietnamese ginseng saponin and its components. Biol Pharm Bull 21:978–981
Johnvesly B, Naik GR (2001) Studies on production of thermostable alkaline protease from thermophilic and alkaliphilic Bacillus sp. JB-99 in a chemically defined medium. Process Biochem 37:139–144
Kamande GM, Baah J, Cheng KJ, McAllister TA, Shelford JA (2000) Effects of tween 60 and tween 80 on protease activity, thiol group reactivity, protein adsorption, and cellulose degradation by rumen microbial enzymes. J Dairy Sci 83:536–542
Khanam Z, Sam KH, Zakaria NHBM, Ching CH, Bhat IUH (2015) Determination of polyphenolic content, HPLC analyses and DNA cleavage activity of Malaysian Averrhoa carambola L. fruit extracts. J King Saud Univ Sci 27(4):331–337
Laemmli DK (1970) Cleavage of structural proteins during in assembly of the head of bacteriophage T4. Nature 227:680–683
Lee JH, Jeon JK, Kim SG, Kim SH, Chun T, Imm JY (2011) Comparative analyses of total phenols, flavonoids, saponins and antioxidant activity in yellow soy beans and mung beans. Int J Food Sci Technol 46:2513–2519
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Moreira KA, Albuqerque BF, Teixeira MFS, Porto ALF, Lima FJL (2002) Application of protease from Nocardiopsis sp. as a laundry detergent additive. World J Microbiol Biotechnol 18:309–315
Mujeeb F, Bajpai P, Pathak N (2014) Phytochemical evaluation, antimicrobial activity, and determination of bioactive components from leaves of Aegle marmelos. Biomed Res Int. https://doi.org/10.1155/2014/497606
Nadeem M, Qazi JI, Syed Q, Gulsher M (2013) Purification and characterization of an alkaline protease from Bacillus licheniformis UV-9 for detergent formulations. Songklanakarin J Sci Technol 35(2):187–195
Oberoi R, Beg QK, Puri S, Saxena RK, Gupta R (2001) Characterization and wash performance analysis of an SDS-stable alkaline protease from a Bacillus sp. World J Microbiol Biotechnol 17:493–497
Otang WM, Grierson DS, Ndip RN (2012) Phytochemical studies and antioxidant activity of two South African medicinal plants traditionally used for the management of opportunistic fungal infections in HIV/AIDS patients. BMC Complement Altern Med 12:43–50
Oyaizu M (1986) Studies on products of browning reaction antioxidative activity of products of browning reaction. Jpn J Nutr 40:307–315
Rajput R, Sharma R, Gupta R (2010) Biochemical characterization of a thiol activated, oxidation stable keratinase from Bacillus pumilus KS12. Enzym Res. https://doi.org/10.4061/2010/132148
Sahin S, Ozmen I, Kir E (2015) Purification, immobilization, and characterization of protease from local Bacillus subtilis M-11. Asia Pac J Chem Eng 10(2):241–247
Sanchez-Moreno C, Larrauri JA, Saura-Calixto FA (1998) Procedure to measure the antiradical efficiency of polyphenols. J Sci Food Agric 76:270–276
Serteser A, Kargıoğlu M, Gök V, Bağcı Y, Özcan M, Arslan D (2009) Antioxidant properties of some plants growing wild in Turkey. Grasas Aceites 60(2):147–154
Singh R, Kumar M, Mittal A, Mehta PK (2016) Microbial enzymes: industrial progress in 21st century. 3 Biotech 6(2):174. https://doi.org/10.1007/s13205-016-0485-8
Singleton V, Orthofer R, Lamuela-Raventos R (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin ciocalteu reagent. Method Enzymol 299:152–178
Sinha R, Khare SK (2013) Thermostable proteases. In: Satyanarayana T, Littlechild J, Kawarabayasi Y (eds) Thermophilic microbes in environmental and industrial biotechnology, 2nd edn. Springer, Dordrecht, pp 859–880
Subramanian M, Chander R, Chattopadhyay S (2006) A novel naturally occurring tripyrrole with potential nuclease and anti-tumour properties. Bioorg Med Chem 14(8):2480–2486
Tepe B, Daferera B, Tepe A, Polissiou M, Sokmen A (2007) Antioxidant activity of the essential oil and various extracts of Nepeta Flavida HubMor from Turkey. Food Chem 103:1358–1364
Xu Q, Nakajima M, Liu Z, Shiina T (2011) Biosurfactants for microbubble preparation and application. Int J Mol Sci 12(1):462–475
Yan W, Ohtani K, Kasai R, Yamasaki K (1996) Steroidal saponins from fruits of Tribulus terrestris. Phytochemistry 42:1417–1422
Yernale NG, Bennikallu Hire Mathada M (2014) Synthesis, characterization, antimicrobial, dna cleavage, and in vitro cytotoxic studies of some metal complexes of schiff base ligand derived from thiazole and quinoline moiety. Bioinorg Chem Appl. https://doi.org/10.1155/2014/314963
Zheng W, Wang SY (2001) Antioxidant activity and phenolic compounds in selected herbs. J Agric Food Chem 49(11):5165–5170
Acknowledgements
Funding was provided by Suleyman Demirel University Research Funds.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yazici, S.Ö., Özmen, İ. Effect of the Crude Saponin Extract from Gypsophila pilulifera Boiss. & Heldr. on Protease from Bacillus subtilis ATCC 6633 and Antioxidant Properties of the Extract. Iran J Sci Technol Trans Sci 42, 1707–1713 (2018). https://doi.org/10.1007/s40995-017-0366-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-017-0366-y