Abstract
Rural populations often depend on small reservoirs for their water supply. These reservoirs are engineered systems designed to serve specific purposes. Water from these sources is not only utilized for drinking purposes, but also for commercial and industrial use. Thus, a thorough limnological study is essential. The present study is carried out at Bakreswar reservoir in Birbhum district, which was created by the dam, built on Bakreswar River. The major purpose of the reservoir is to supply drinking water to the surrounding villages and to use Bakreswar Thermal Power Station for cooling their power plant. A previous study on this reservoir has shown temporal variation of environmental factors as well as good assemblage of zooplankton. In order to get a more detailed preview on zooplankton groups, seasonal diversity is calculated for four major groups of zooplanktons (Cladocera, Ostracoda, Copepoda and Rotifera) with the software PAST. Spatial pattern analysis of zooplankton community is also performed to propose possible hypotheses explaining reasons underlying the causal factors for any such variation. Moreover, community ordination is done using SPSS 20 to understand the overall ecosystem functioning of the reservoir. The results show that Station I and Station III are more diversified in all the seasons compared to Station II. The mode of distribution is clumped, and the degree of clumping varied with different functional groups of studied zooplankton as seen in the two methods used. So it can be concluded that the zooplankton community of this reservoir is diverse and shows horizontal clumped distribution pattern and niche separation among the groups to avoid competition. The results help in formulating proper strategies for advanced water quality management and conservation of reservoir ecosystem.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Reservoirs are impoundments of flowing waters. Unlike natural lakes, reservoirs and dams are engineered systems designed to serve specific purpose (Kennedy 1999). Dams create new ecosystems that have hybrid characteristics of lakes and rivers (Soballe et al. 1992), and they appear as longitudinal zones in reservoirs.
Dams create new ecosystems that historically reservoirs were built to serve a single purpose, i.e., irrigation. Nowadays with demanding need for water in different aspects of life, reservoirs are constructed to serve multiple purposes (Straškraba and Tundisi 1999). India is a developing country of Asia and is facing an outburst of population growth. As a result, industrial and agricultural development is at full pace. Social demands for different resources are increasing fast. Thus, to fulfill the necessity of water supply, reservoirs and dams are constructed throughout the country as it harbors some of world’s longest rivers. Today India has 4710 completed large dams and reservoirs and 341 are under construction (NRLD, 2014); South India has the maximum number of reservoirs, especially Tamil Nadu and Karnataka.
The food web of a reservoir consists mainly of phytoplankton, zooplankton, bacterioplankton, other invertebrates and fish in the open water zone. The littoral zone consists of the macrophytes. Usually, algae and cyanobacteria are the common phytoplankton in a reservoir (Geraghty et al. 1973). Prior studies were carried out in the selected reservoir that dealt with temporal variation of environmental factors and zooplankton which focused on its water quality, along with a study on the trophic relationship and ecosystem functioning of Bakreswar reservoir (Banerjee et al. 2015, 2016). Zooplankton is an important aspect of freshwater ecosystems as it serves as a consumer of phytoplankton and is a main link in the food web. Zooplankton forms a major link in the energy transfer at secondary level in aquatic food webs between autotrophs and heterotrophs (Deivanai et al. 2004). Studies of zooplankton diversity in any water body also indicate the water quality. Zooplankton shows a vertical as well as horizontal distribution in aquatic environments. Reservoirs are no exception to that. Diel vertical migration driven mainly by predation and damaging ultraviolet radiation is a common occurrence (Dini and Carpenter 1992; Lienesch and Matthews 2000; Lampert et al. 2003). Variability in species assemblage and density of zooplankton on a horizontal scale has also been reported in multiple systems (Wurtsbaugh and Li 1985; Patalas and Salki 1993; Geraldes and Boavida 2004; Vijanen et al. 2009). Zooplankton comprising of rotifers, cladocerans, copepods and ostracods are considered to be most important in terms of population density, biomass production, grazing and nutrient regeneration in any aquatic ecosystem (Sehgal et al. 2013).
The spatial pattern of plants and animals is an important characteristic of ecological communities (Connell 1963). There are three distinct patterns: random, clumped and uniform. Once a pattern has been identified, it is easy to generate a hypothesis concerning the structure of ecological communities (Williams 1976). Another important aspect of ecological community is variation in species abundance of species distribution often modeled with environmental effects (Sarkar et al. 2008; Venturino et al. 2016). An easy way of describing species abundance relation is through study of diversity indices. Based on these studies, community organization can be done, where communities can be grouped based on their similarities through principle component analysis (Ludwig and Reynolds 1988). Community structure changes abruptly during construction of a dam.
In the present study, Bakreswar reservoir of Birbhum district has been selected as study site. The dam is constructed over Bakreswar River. The major purpose of the reservoir is to supply drinking water to the surrounding villages and for thermal power generation at the Bakreswar Thermal Power Station, under West Bengal Power Development Corporation. In a previous study, a thorough analysis of the environmental variables is done to get an overview of the water quality of the reservoir, which shows temporal variation of environmental factors and zooplankton (Banerjee et al. 2015). This study is an extension of the previous work which focuses on the diversity and distribution pattern of zooplankton to get an overview of the community organization. This will be useful for formulation of long-term management strategies ensuring the ecosystem health for not only the aforesaid reservoir but other such ecosystems as well.
2 Material and methods
2.1 Study site
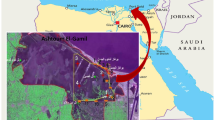
Bakreswar reservoir of Birbhum district, West Bengal (lat. 23°50.519′N: long. 87°24.612′E), was selected as the study site. Constructed on river Bakreswar, the dam functions as a backup water supply for the Bakreswar Thermal Power Station under the supervision of West Bengal Power Development Corporation in 1999 (Fig. 1) with the main purpose being the supply of drinking water to the neighboring villages of Chinpai and Bhurkuna Gram Panchayat. It is located 3 km northwest of the power station and has a storage volume of 2.29 million m3, retention time of 90 days and surface area of 6.38 km2 (BKTPS 2012). The reservoir has a rich diversity of macrophytes (aquatic plant growing near water body is either submergent, emergent or floating) like sedge (Scirpus spp.), reed (Fragmites spp.), pond weeds (Potamogeton spp.), hornworts (Ceratophyllum spp.), water lettuce (Enteromorpha sp., Pistia sp.), Kans grass (Saccharumspontaneum) (Sinha et al. 2012). It also supports rich diversity of arthropods, mollusks and fish (Sinha et al. 2011). A detailed study on the bird species revealed that it harbors breeding grounds for Great Cormorant (Phalacrocoraxcarbo), Great Crested Grebe (Podicepscristatus), Ruddy Shellduck (Tadornaferruginea), Gadwall (Anasstrepera) and Common Coot (Fulicaatra) which are considered to be indicators of Bakreswar as a wetland (Sinha et al. 2011), some of them being common winter migrants which visit Bakreswar every winter. This site is a second abode for migratory birds after the deterioration of Tilpara Barrage located 17.8 km from Bakreswar which was also considered to be the prime source of water supply to the thermal power station.
2.2 Sampling
In order to study whether the zooplankton community of the selected reservoir shows random, uniform or clumped distribution, at first the reservoir is divided into three sampling stations, Station I, Station II and Station III. Then, for a better understanding of the distribution pattern the total embankment area of the reservoir is divided into nine stations keeping a gap of about 500 m. Sampling was done from March 2012 to February 2014. The whole study period was divided into three seasons: pre-monsoon (PM; March–June), monsoon (M, July–October) and post-monsoon (PTM, November–February). Samples were collected fortnightly from the above-mentioned stations (three samples/station) during each visit. Plankton samples were collected using a zooplankton net of mesh size 200 µm. They were stored in small 30-ml vials and preserved in 4% formalin. Four major zooplankton groups (Cladocera, Copepoda, Ostracoda and Rotifera) were counted in a Sedgwick Rafter counter and expressed in numbers/liter (No/L).
2.3 Statistical analysis
Species abundance relation was calculated in terms of diversity index. The common indices calculated were Shannon–Weiner diversity index (H), Simpson index of dominance (D), Evenness index (e) and Margalef’s richness (R) index using the software PAST (Palentological Statistics 2.02). The values of D and e always range between 0 and 1. More the value tends toward 1, more the community is said to have high species richness. H value more than 1 indicates a diversified community. The formulae are presented below (Ludwig and Reynolds 1988):
where p i are the proportional abundance of the ith species
where \( S = \sum\nolimits_{i = 1}^{S} {p_{i}^{2} } \)
where S is the number of species
where S is the number of species and n is the number of individuals in each species
For many years, Fischer (1960) coefficient of dispersion [the ratio of sample variance (s 2) to sample mean (\( \overline{X} \))] was used by plankton ecologists, both as a means of detecting patchiness and as a measure of the degree of patchiness. When it becomes necessary to quantify different patterns of dispersion in populations, indices based on the variance/mean ratio are usually inappropriate since they are nearly all influenced by the number of individuals in the sample. A more generally acceptable measure of patchiness is that based on the index of ‘mean crowding’ (Lloyd 1967).
The formulae are presented below:
In the above-mentioned expressions: \( S_{2} \left( {\text{variance}} \right) = \frac{{\sum Xi^{2} {-}\left( {\sum Xi} \right)^{2} /N}}{N - 1} \), where X = number of individuals in a sample, \( \overline{X} = \frac{\sum Xi}{N} \) and N = number of samples. Species of a particular site was considered to be clumped, aggregated or contagious in a distribution if value of \( \dot{X} \) exceeds the value of \( \overline{X} \). When \( \dot{X} \) was equal to \( \overline{X} \), distribution pattern was considered to be random, and when \( \dot{X} \) was less than \( \overline{X} \), it was considered to be tending toward regularity.
The first-quadrat variance model called block quadrat variance method (BQV) was proposed by (Greig-Smith 1952) and later modified by (Kershaw 1977). Here, strips of contiguous quadrats were blocked in successive power of two and variances of non-overlapping blocks of different sizes were calculated. But as this method has a limitation of block size based on power of two, a more relevant and advanced method was later introduced by (Goodall 1974) and Ludwig and (Goodall 1978). The paired-quadrat variance method (PQV) is based on selected specified spacing or distance and does not depend on the block size. The present study involved nine stations creating block spacing each of approximately 500 m. Here, pairs of quadrats are selected at a specific interval (500 m) to estimate variances. Variance for each of the spacing is calculated as
where VAR (x) = variance of x, N = number of quadrats, X = number of individuals in each quadrat and x = number of spacing. As there are nine quadrats of size approximately 500 m, four block spacings are selected as given in Table 1.
In order to understand the ordination of the studied stations based on zooplankton abundance, principal component analysis and hierarchical cluster are applied. PCA is the most appropriate method for dimension reduction in space and indirect gradient analysis for improved data interpretation. PCA relies upon an eigenvector decomposition of the covariance or correlation matrix, and correlation matrix is usually preferred since the units or variables are greatly different (Bengraïne and Marhaba 2003). In this study, PCA was run on correlation matrix of centered and standardized data using SPSS 20, IBM Corp, 2011, in order to identify the important components that explain most of the variance in reservoir ecosystem. To analyze the similarity in seasonal samples, agglomerative hierarchical cluster analysis (nearest neighbor method) was done.
3 Results
Analysis of diversity indices of first study period (2012–2013) showed that Shannon–Weiner diversity index is high (1.36) in Station I during the pre-monsoon period, whereas Station III recorded higher value (1.32) in the monsoon period. Dominance indices showed similar range (0.34–0.36) in all three seasons in Station II, but high value was recorded in Station I in post-monsoon and similar values (0.34) in Station III for both pre-monsoon and post-monsoon. Evenness index also showed similar range (0.81–0.82) in Station II in all three seasons. High values of evenness index were obtained in pre-monsoon from Station I (0.97) and in monsoon (0.94) from Station III. Richness values are more or less similar (0.51–0.57) in all three stations for all seasons (Table 2).
The results of diversity indices of the second study period (2013–2014) showed that Shannon–Weiner index has high value (>1.00) in pre-monsoon for all three stations, and also the evenness index (>0.80). Dominance values are high (>0.40) in post-monsoon for all three seasons. Richness is more or less familiar (0.52–0.58) in all the three stations and does not vary through seasons (Table 2).
The analysis of Fischer’s coefficient of dispersion and mean crowding was made to assess the pattern of dispersion of zooplankton. Spatial pattern analysis showed a clumped mode of dispersion in all three stations (Table 3).
Paired-quadrat variance method also revealed clumped mode of distribution for the study period for each group, and the spacing of clumping varied between the four zooplankton groups. Cladocerans show high degree of clumping at block size 3 along with copepods, but higher degree of clumping is found in block size 2 and 4 for Rotifera and Ostracoda, respectively (Fig. 2a–d). There is no distinct change in the clumping pattern of all these four groups for the studied two years.
In order to understand the ordination of the three studied stations in space, PCA is applied. Results show that two principal components are extracted and have defined the stations based on the abundance pattern of four zooplankton groups. The eigenvalues shown after varimax rotation in Table 3 indicate that Stations I and III are positively correlated with component 1 and Station II has similar positive correlation with component 2. Total variance explained by PCA ordination is 99.93%. The rotated component matrix based on varimax method has clearly separated Station II from the other two stations.
A clear spatial ordination of the three studied stations is well documented in the PCA bi-plot. It is clear that Station I and Station III are spatially correlated with high abundance of copepods and cladocerans. Station II is mostly represented by Ostracoda species. Rotifers do not play a crucial role in spatial separation of the three stations and indicate their uniform abundance pattern in all three stations (Fig. 3). A similar result is also provided by hierarchical cluster where Stations I and III form an in-group and Station II is the out-group (Fig. 4). Thus, from the dendogram it is observed that Stations I and III are very closely associated as the calculated distance between them is very small compared to Station II. Thus, the results reveal that Stations I and III are spatially correlated and have similar diversity profile and distribution pattern (Table 4).
4 Discussion
Zooplankton community is considered to be the strategic component of the aquatic ecosystem, and it maintains and orients the aquatic food webs. Thus, its positioning in the food chain with its high degree of connection to the primary producers makes it highly susceptible to different structural heterogeneity in the system (Eskinazi-SantAnna et al. 2013). It is essential to characterize the community structure of the zooplankton. The easiest and convenient way of analyzing community characteristics is to use diversity indices. The results mentioned in Table 2 have shown a high diversity profile of zooplankton in Station I and Station III as compared to Station II. This can be due to the fact that this station is situated just near the dam and log gate where a disturbance in the water current is caused due to regular discharge of water. Water is pumped out at regular interval to the thermal power cooling plant. As a result, the depth of the reservoir near Station II fluctuates between 72 and 75 m in pre- and post-monsoon period. Due to excessive rainfall during the second study period, flushing of water was more frequent; as a result, diversity is low during monsoon. Lowering of plankton diversity with heavy precipitation was reported from the works of Rajagopal et al. (2010). A similar work from Dekhu reservoir Aurangabad (Sontakke and Mokashe 2014) also supports the observations seen in Bakreswar reservoir. Lowering of diversity can be related to the fact that rotifer density usually decreases with heavy rainfall which allows the growth of cladocerans and copepod population increasing the dominance index (Shivashankar and Venkataramana 2013).
Spatial pattern analysis is done in order to know whether a system shows homogeneous or heterogeneous distribution pattern. Major studies were made on the temporal dynamics of a reservoir, but little is made on the spatial dynamics of reservoir ecosystem in India. The distribution pattern of zooplankton groups can be considered as an essential tool for such studies as established by the works of (Bini et al. 1997) at Broa reservoir, Sao Paulo, Brazil. Similar studies in different eutrophic reservoirs also suggest that zooplankton show a heterogeneous, i.e., clumped dispersion pattern (Eskinazi-Sant’Anna et al. 2013). Fischer’s coefficient of dispersion used in the present study has also showed a clumped pattern of distribution of zooplankton. Whenever environment is uniform, a random pattern of distribution is exhibited, but Odum and Barrett (1971) have quoted ‘Clumping of varying degree represents by far the commonest pattern, almost the rule when individuals are considered.’ Accordingly, highest clumping is seen in Station III by cladocerans and copepods followed by Station I. Rotifers and ostracods show high degree of clumping in Station II. The possible reason is that Stations I and III harbor high zooplankton assemblage as indicated by the calculated diversity indices. These stations also have a rich nutrient base for the growth of phytoplankton that in turn supports the growth of zooplankton. It is already been established that resource availability is the primary cause of clumping in a particular area, the other reasons being predation (Townsend et al. 2003). Field survey has also revealed that above-mentioned stations give maximum success in fishing, which also indicates the availability of resource in this area that supports the cause of clumping of zooplanktons. It has been already established that fine-scale horizontal variability in zooplankton distribution is related to the presence and feeding of planktivorous fish (Pinel-Alloul 1995; Jeppesen et al. 1997; Romero et al. 2004). In Bakreswar reservoir, clumping of cladocerans and copepods is high in Stations I and III as already discussed. Station II is a log gate area of the dam and has a depth between 71 and 75 m. Visman et al. (1994) have already pointed out that cladocerans, especially daphnids (which is mostly found in Bakreswar reservoir as well) and cyclopoid copepods, prefer shallow stations. The results of Bakreswar reservoir also support this fact as both Stations I and III are shallow stations with less water depth.
Index of dispersion is based on sampling units and its size and thus has some limitations in predicting the exact distribution pattern for organisms with continuous distribution. It provides a preliminary idea about the degree of patchiness. Thus, a more appropriate method based on arbitrary sampling units in terms of blocks or quadrats is now common. Paired-quadrat method is used in this present study in order to identify the horizontal spacing or distance of clumping for the four zooplankton groups. The results have reflected a strong clumping pattern (Fig. 2). The peak of the variance calculated for each block size is equivalent to the radius of the clump mean area, and the average distance from center of clumps will be twice the block size spacing (Ludwig and Reynolds 1988). In our present study, it was observed that cladocerans and copepods PQV graph has a variance peak at block size 3, whereas rotifer has a peak at block size 2 and Ostracoda at block size 4. It can be concluded that cladoceran and copepod clumps are spaced at spacing of 3000 m (block spacing = twice the block spacing 3, i.e., 500 × 3 = 3000) from the point of origin of the first quadrate. Rotifer clumps are spaced at intervals of 2000 m (block spacing = twice the block spacing 2, i.e., 500 × 2 = 1000) and Ostracoda clumps spaced at 4000 m (block spacing = twice the block spacing 4, i.e., 500 × 4 = 2000). Thus, a well-documented variability in horizontal dispersion pattern is present in Bakreswar reservoir. The zooplankton groups have clumps of distribution that are non-overlapping which suggests that they avoid the chance of competition and utilize resource to its fullest. Vijanen et al. (2009) have already suggested a coexistence of Cladocera and Copepoda in similar environmental conditions. Thus, occurrence of their clumps at similar intervals can be easily explained. Rajashekhar et al. (2010) have already reported a negative relation in pattern of rotifer and cladocera from the study of a freshwater reservoir in Karnataka. As rotifers can explore, several environmental habitats clump at small spacing is expected. On the other hand, ostracods being the least abundant group among zooplankton and more keen to slight changes in environmental conditions show clumps spaced at large distances (Rajashekhar et al. 2010; Jadhav et al. 2012; Ahmed et al. 2013).
To sum up, it can be concluded from the study that Bakreswar reservoir is a healthy ecosystem. It can be safely used as a source of drinking water for the surrounding village. It also has a good zooplankton diversity, which indicates the presence of both phytoplankton and fish in the system. Thus, the reservoir is sustainable, and with this knowledge of reservoir functioning, proper management strategies can be taken up for future concern. As Stations I and III have a higher clumping pattern, this area can further be exploited through subsampling study in order to get a clear picture of the possible cause of such distribution pattern.
References
Ahmed Y, Hussain A, Hussain G et al (2013) Diversity and seasonal variations of zooplanktons in Pahuj reservoir at Jhansi (U.P) India. Int J Pharm Biol Arch 4:100–104
Banerjee M, Mukherjee J, Banerjee A et al (2015) Impact of environmental factors on maintaining water quality of Bakreswar reservoir, India. Comput Ecol Softw 5:239–253
Banerjee A, Banerjee M, Mukherjee J et al (2016) Trophic relationships and ecosystem functioning of Bakreswar reservoir, India. Ecol Inform 36:50–60. doi:10.1016/j.ecoinf.2016.09.006
Bengraïne K, Marhaba TF (2003) Using principal component analysis to monitor spatial and temporal changes in water quality. J Hazard Mater 100:179–195
Bini LM, Tundisi JG, Matsumura-Tundisi T, Matheus CE (1997) Spatial variation of zooplankton groups in a tropical reservoir (Broa reservoir, São Paulo State-Brazil). Hydrobiologia 357:89–98
Bktps PD (2012) Bakreswar thermal power project (1) and (2). Bakerswar, India
Connell JH (1963) Territorial behavior and dispersion in some marine invertebrates. Res Popul Ecol (Kyoto) 5:87–101
Deivanai K, Arunprasath S, Rajan MK, Baskaran S (2004) Biodiversity of phyto and zooplankton in relation to water quality parameters in a sewage polluted pond at Ellayirampannai, Virudhunagar district. In: The proceedings of national symposium on biodiversity resources management and sustainable use, organized by the center for biodiversity and Forest studies. Madurai Kamaraj University, Madurai
Dini ML, Carpenter SR (1992) Fish predators, food availability and diel vertical migration in Daphnia. J Plankton Res 14:359–377
Eskinazi-SantAnna EM, Menezes R, Costa IS et al (2013) Zooplankton assemblages in eutrophic reservoirs of the Brazilian semi-arid. Braz J Biol 73:37–52
Fischer AG (1960) Latitudinal variations in organic diversity. Evolution (N Y) 14:64–81
Geraghty JJ, Miller DW, Van Der Leenden F, Troise FL (1973) Water atlas of the United States. Water Information Center, Port Washington, New York
Geraldes AM, Boavida MJ (2004) Do littoral macrophytes influence crustacean zooplankton distribution? Limnetica 23:57–63
Goodall DW (1974) A new method for the analysis of spatial pattern by random pairing of quadrats. Vegetatio 29:135–146
Goodall DW (1978) Sample similarity and species correlation. In: Whittaker RH (ed) Ordination of plant communities. Springer, Netherlands, pp 99–149
Greig-Smith P (1952) The use of random and contiguous quadrats in the study of the structure of plant communities. Ann Bot 16(62):293–316
Jadhav S, Borde S, Jadhav D, Humbe A (2012) Seasonal variations of zooplankton community in Sina Kolegoan Dam Osmanabad district, Maharashtra, India. J Exp Sci 3:19–22
Jeppesen E, Jensen JP, Søndergaard M et al (1997) Top-down control in freshwater lakes: the role of nutrient state, submerged macrophytes and water depth. In: Kufel L, Prejs A, Rybak JI (eds) Shallow Lakes 95. Springer, Netherlands, pp 151–164
Kennedy RH (1999) Reservoir design and operation: limnological implications and management opportunities. Theor Reserv Ecol Appl 9:1–28
Kershaw KA (1977) Physiological-environmental interactions in lichens. New Phytol 79:377–390
Lampert W, McCauley E, Manly BFJ (2003) Trade-offs in the vertical distribution of zooplankton: ideal free distribution with costs? Proc R Soc Londn B Biol Sci 270:765–773
Lienesch PW, Matthews WJ (2000) Daily fish and zooplankton abundances in the littoral zone of Lake Texoma, Oklahoma-Texas, in relation to abiotic variables. Environ Biol Fish 59:271–283
Lloyd M (1967) Mean crowding. J Anim Ecol 36(1):1–30
Ludwig JA, Reynolds JF (1988) Statistical ecology: a primer in methods and computing. Wiley, New York
Odum EP, Barrett GW (1971) Fundamentals of ecology. Saunders, Philadelphia
Patalas K, Salki A (1993) Spatial variation of crustacean plankton in lakes of different size. Can J Fish Aquat Sci 50:2626–2640
Pinel-Alloul B (1995) Spatial heterogeneity as a multiscale characteristic of zooplankton community. Hydrobiologia 300(1):17–42
Rajagopal T, Thangamani A, Sevarkodiyone SP et al (2010) Zooplankton diversity and physico-chemical conditions in three perennial ponds of Virudhunagar district, Tamilnadu. J Environ Biol 31:265–272
Rajashekhar M, Vijaykumar K, Paerveen Z (2010) Seasonal variations of zooplankton community in freshwater reservoir Gulbarga district, Karnataka South India. Int J Syst Biol 2:6
Romero JR, Hipsey MR, Antenucci JP, Hamilton D (2004) Computational aquatic ecosystem dynamics model: CAEDYM v2, science manual. Centre for Water Research, University of Western Australia, Nedlands, WA, Australia
Sarkar RR, Pal J, Das KP, Chattopadhyay J (2008) Control of harmful algal blooms through input of competitive phytoplankton and the effect of environmental variability. J Calcutta Math Soc 4:1–8
Sehgal K, Phadke GG, Chakraborty SK, Reddy SVK (2013) Studies on zooplakton diversity of Dimbhe reservoir, Maharashtra, India. Adv Appl Sci Res 4:417–420
Shivashankar P, Venkataramana GV (2013) Zooplankton diversity and their seasonal variations of Bhadra reservoir, Karnataka, India. Int Res J Environ Sci 2:87–91
Sinha A, Hazra P, Khan TN (2011) Population trends and spatiotemporal changes to the community structure of waterbirds in Birbhum district, West Bengal, India. Proc Zool Soc 64:96–108. doi:10.1007/s12595-011-0018-8
Sinha A, Hazra P, Khan TN (2012) Emergence of a wetland with the potential for an avian abode of global significance in South Bengal, India. Curr Sci 102:613–615
Soballe DM, Kimmel BL, Kennedy RH, Gaugush RF (1992) Reservoirs. Biodivers Southeast United States Aquat communities Wiley 421–476
Sontakke G, Mokashe S (2014) Diversity of zooplankton in Dekhu reservoir from Aurangabad, Maharashtra. J Appl Nat Sci 6:131–133
Straškraba M, Tundisi JG (1999) Guidelines of lake management: Reservoir water quality management, vol 9. Int Lake Environ Comm 1–60
Townsend CR, Begon M, Harper JL (2003) Essentials of ecology. Blackwell Science, Oxford, UK
Venturino E, Roy PK, Al Basir F, Datta A (2016) A model for the control of the mosaic virus disease in Jatropha curcas plantations. Energy Ecol Environ 1:360–369
Vijanen M, Holopainen A-L, Rahkola-Sorsa M, Avinsky V, Ruuska M, Leppänen S, Rasmus K, Voutilainen A (2009) Temporal and spatial heterogeneity of pelagic plankton in Lake Pyhäselkä, Finland. Boreal Environ Res 14:903–913
Visman V, McQueen DJ, Demers E (1994) Zooplankton spatial patterns in two lakes with contrasting fish community structure. Hydrobiologia 284:177–191
Williams WT (1976) Pattern analysis in agricultural science
Wurtsbaugh W, Li H (1985) Diel migrations of (Menidia beryllina) in relation to the distribution of its prey in. Limnol Ocean 30:565476
Acknowledgements
The author Moitreyee Chakrabarty would like to thank Dr. Goutam Bandyopadhyay, Department of Management Studies, NIT, Durgapur, for his contribution in the statistical analysis. She would also like to thank Head, Department of Chemistry, NIT, Durgapur, for providing the necessary laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors hereby declare that there are no conflicts of interest whatsoever.
Rights and permissions
About this article
Cite this article
Chakrabarty, M., Banerjee, A., Mukherjee, J. et al. Spatial pattern analysis of zooplankton community of Bakreswar reservoir, India. Energ. Ecol. Environ. 2, 198–206 (2017). https://doi.org/10.1007/s40974-017-0057-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40974-017-0057-8