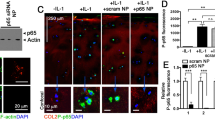
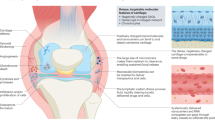
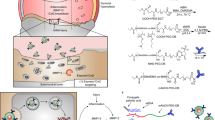
Abstract
Articular cartilage injury, as a hallmark of arthritic diseases, is difficult to repair and causes joint pain, stiffness, and loss of mobility. Over the years, the most significant problems for the drug-based treatment of arthritis have been related to drug administration and delivery. In recent years, much research has been devoted to developing new strategies for repairing or regenerating the damaged osteoarticular tissue. The RNA interference (RNAi) has been suggested to have the potential for implementation in targeted therapy in which the faulty gene can be edited by delivering its complementary short interfering RNA (siRNA) at the posttranscriptional stage. The successful editing of a specific gene by the delivered siRNA might slow or halt osteoarthritic diseases without side effects caused by chemical inhibitors. However, cartilage siRNA delivery remains a challenging objective because cartilage is an avascular and very dense tissue with very low permeability. Furthermore, RNA is prone to degradation by serum nucleases (such as RNase H and RNase A) due to an extra hydroxyl group in its phosphodiester backbone. Therefore, successful delivery is the first and most crucial requirement for efficient RNAi therapy. Nanomaterials have emerged as highly advantage tools for these studies, as they can be engineered to protect siRNA from degrading, address barriers in siRNA delivery to joints, and target specific cells. This review will discuss recent breakthroughs of different siRNA delivery technologies for cartilage diseases.
Lay Summary
Articular cartilage breakdown is a hallmark of osteoarthritis, which is difficult to repair and treat. Currently, there is no disease-modifying therapeutic approved by FDA to treat osteoarthritis. In recent years, RNAi drugs have been suggested to have the potential for repairing or regenerating the damaged articular cartilage. However, the effective delivery of small RNAs remains a significant challenge. To overcome these obstacles, nanomaterial delivery systems including polymers, lipids, peptides, and oligonucleotide nanoparticles have been developed to enhance the effectiveness of RNAi drugs. Here, we review recent progress in using nanomaterials to deliver small RNAs for cartilage disease therapeutics. In the future, nanomaterials can be specifically designed to form small-sized delivery vehicles with excellent penetration properties. RNAi therapeutics based on these nanomaterials will have great promise to treat cartilage diseases.


Similar content being viewed by others
References
Felson DT, Zhang YQ. An update on the epidemiology of knee and hip osteoarthritis with a view to prevention. Arthritis Rheum. 1998;41(8):1343–55.
Strombeck B, Petersson IF, Vlieland TPMV, Grp ENW. Health care quality indicators on the management of rheumatoid arthritis and osteoarthritis: a literature review. Rheumatology. 2013;52(2):382–90.
Bajpayee AG, Grodzinsky AJ. Cartilage-targeting drug delivery: can electrostatic interactions help? Nat Rev Rheumatol. 2017;13(3):183–93.
Rothenfluh DA, Bermudez H, O'Neil CP, Hubbell JA. Biofunctional polymer nanoparticles for intra-articular targeting and retention in cartilage. Nat Mater. 2008;7(3):248–54.
Gary DJ, Puri N, Won YY. Polymer-based siRNA delivery: perspectives on the fundamental and phenomenological distinctions from polymer-based DNA delivery. J Control Release. 2007;121(1–2):64–73.
Fire A, Xu SQ, Montgomery MK, Kostas SA, Driver SE, Mello CC. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature. 1998;391(6669):806–11.
Wang SW, Wei XC, Sun XJ, Chen CW, Zhou JM, Zhang G, et al. A novel therapeutic strategy for cartilage diseases based on lipid nanoparticle-RNAi delivery system. Int J Nanomedicine. 2018;13:617–31.
Zhang G, Guo BS, Wu H, Tang T, Zhang BT, Zheng LZ, et al. A delivery system targeting bone formation surfaces to facilitate RNAi-based anabolic therapy. Nat Med. 2012;18(2):307–14.
Abdallah B, Hassan A, Benoist C, Goula D, Behr JP, Demeneix BA. A powerful nonviral vector for in vivo gene transfer into the adult mammalian brain: Polyethylenimine. Hum Gene Ther. 1996;7(16):1947–54.
Trubetskoy VS, Torchilin VP, Kennel SJ, Huang L. Use of N-terminal modified poly(L-lysine) antibody conjugate as a carrier for targeted gene delivery in mouse lung endothelial-cells. Bioconjug Chem. 1992;3(4):323–7.
Cho C-S. Design and development of degradable polyethylenimines for delivery of DNA and small interfering RNA: an updated review. ISRN Mater Sci. 2012;2012:1–24.
Cao HQ, Jiang X, Chai C, Chew SY. RNA interference by nanofiber-based siRNA delivery system. J Control Release. 2010;144(2):203–12.
Liu L, Zheng M, Librizzi D, Renette T, Merkel OM, Kissel T. Efficient and tumor targeted siRNA delivery by polyethylenimine-graft-polycaprolactone-block-poly(ethylene glycol)-folate (PEI-PCL-PEG-Fol). Mol Pharm. 2016;13(1):134–43.
Feng CL, Han YX, Guo HH, Ma XL, Wang ZQ, Wang LL, et al. Self-assembling HA/PEI/dsRNA-p21 ternary complexes for CD44 mediated small active RNA delivery to colorectal cancer. Drug Deliv. 2017;24(1):1537–48.
Sezlev Bilecen D, Uludag H, Hasirci V. Development of PEI-RANK siRNA complex loaded PLGA nanocapsules for the treatment of osteoporosis. Tissue Eng Part A. 2019;25(1–2):34–43.
Zhang SB, Xu YM, Wang B, Qiao WH, Liu DL, Li ZS. Cationic compounds used in lipoplexes and polyplexes for gene delivery. J Control Release. 2004;100(2):165–80.
Guo JF, Cheng WP, Gu JX, Ding CX, Qu XZ, Yang ZZ, et al. Systemic delivery of therapeutic small interfering RNA using a pH-triggered amphiphilic poly-L-lysine nanocarrier to suppress prostate cancer growth in mice. Eur J Pharm Sci. 2012;45(5):521–32.
Hartono SB, Gu WY, Kleitz F, Liu J, He LZ, Middelberg APJ, et al. Poly-L-lysine functionalized large pore cubic mesostructured silica nanoparticles as biocompatible carriers for gene delivery. ACS Nano. 2012;6(3):2104–17.
Inoue Y, Kurihara R, Tsuchida A, Hasegawa M, Nagashima T, Mori T, et al. Efficient delivery of siRNA using dendritic poly(L-lysine) for loss-of-function analysis. J Control Release. 2008;126(1):59–66.
Boussif O, Lezoualch F, Zanta MA, Mergny MD, Scherman D, Demeneix B, et al. A versatile vector for gene and oligonucleotide transfer into cells in culture and in-vivo - polyethylenimine. P Natl Acad Sci USA. 1995;92(16):7297–301.
Rodriguez M, Lapierre J, Ojha CR, Kaushik A, Batrakova E, Kashanchi F, et al. Intranasal drug delivery of small interfering RNA targeting Beclin1 encapsulated with polyethylenimine (PEI) in mouse brain to achieve HIV attenuation. Sci Rep. 2017;7(1):1862.
Urban-Klein B, Werth S, Abuharbeid S, Czubayko F, Aigner A. RNAi-mediated gene-targeting through systemic application of polyethylenimine (PEI)-complexed siRNA in vivo. Gene Ther. 2005;12(5):461–6.
Lacave JM, Fanjul A, Bilbao E, Gutierrez N, Barrio I, Arostegui I, et al. Acute toxicity, bioaccumulation and effects of dietary transfer of silver from brine shrimp exposed to PVP/PEI-coated silver nanoparticles to zebrafish. Comp Biochem Physiol C Toxicol Pharmacol. 2017;199:69–80.
Israel LL, Lellouche E, Ostrovsky S, Yarmiayev V, Bechor M, Michaeli S, et al. Acute in vivo toxicity mitigation of PEI-coated maghemite nanoparticles using controlled oxidation and surface modifications toward siRNA delivery. ACS Appl Mater Interfaces. 2015;7(28):15240–55.
Breunig M, Hozsa C, Lungwitz U, Watanabe K, Umeda I, Kato H, et al. Mechanistic investigation of poly(ethylene imine)-based siRNA delivery: disulfide bonds boost intracellular release of the cargo. J Control Release. 2008;130(1):57–63.
Wen YT, Pan SR, Luo X, Zhang X, Zhang W, Feng M. A biodegradable low molecular weight polyethylenimine derivative as low toxicity and efficient gene vector. Bioconjug Chem. 2009;20(2):322–32.
Zhang TT, Bai XH, Mao XH. Systemic delivery of small interfering RNA targeting the interleukin-2/15 receptor beta chain prevents disease progression in experimental arthritis. PLoS One. 2013;8(11):e78619.
Lv H, Zhang S, Wang B, Cui S, Yan J. Toxicity of cationic lipids and cationic polymers in gene delivery. J Control Release. 2006;114(1):100–9.
Shi Q, Rondon-Cavanzo EP, Dalla Picola IP, Tiera MJ, Zhang XL, Dai KR, et al. In vivo therapeutic efficacy of TNF alpha silencing by folate-PEG-chitosan-DEAE/siRNA nanoparticles in arthritic mice. Int J Nanomedicine. 2018;13:387–402.
Howard KA, Rahbek UL, Liu X, Damgaard CK, Glud SZ, Andersen MO, et al. RNA interference in vitro and in vivo using a novel chitosan/siRNA nanoparticle system. Mol Ther. 2006;14(4):476–84.
Raftery R, O'Brien FJ, Cryan SA. Chitosan for gene delivery and orthopedic tissue engineering applications. Molecules. 2013;18(5):5611–47.
Felgner PL, Gadek TR, Holm M, Roman R, Chan HW, Wenz M, et al. Lipofection - a highly efficient, lipid-mediated DNA-transfection procedure. Proc Natl Acad Sci U S A. 1987;84(21):7413–7.
Kesharwani P, Gajbhiye V, Jain NK. A review of nanocarriers for the delivery of small interfering RNA. Biomaterials. 2012;33(29):7138–50.
Xue HY, Guo P, Wen WC, Wong HL. Lipid-based nnocarriers for RNA delivery. Curr Pharm Des. 2015;21(22):3140–7.
Parashar D, Rajendran V, Shukla R, Sistla R. Lipid-based nanocarriers for delivery of small interfering RNA for therapeutic use. Eur J Pharm Sci. 2020;142:105159.
Xu YH, Szoka FC. Mechanism of DNA release from cationic liposome/DNA complexes used in cell transfection. Biochemistry-Us. 1996;35(18):5616–23.
Wittrup A, Ai A, Liu X, Hamar P, Trifonova R, Charisse K, et al. Visualizing lipid-formulated siRNA release from endosomes and target gene knockdown. Nat Biotechnol. 2015;33(8):870–6.
Burnett JC, Rossi JJ. RNA-based therapeutics: current progress and future prospects. Chem Biol. 2012;19(1):60–71.
Semple SC, Akinc A, Chen JX, Sandhu AP, Mui BL, Cho CK, et al. Rational design of cationic lipids for siRNA delivery. Nat Biotechnol. 2010;28(2):172-U18.
Schroeder A, Levins CG, Cortez C, Langer R, Anderson DG. Lipid-based nanotherapeutics for siRNA delivery. J Intern Med. 2010;267(1):9–21.
Huang L, Liu Y. In vivo delivery of RNAi with lipid-based nanoparticles. Annu Rev Biomed Eng. 2011;13:507–30.
Tseng YC, Mozumdar S, Huang L. Lipid-based systemic delivery of siRNA. Adv Drug Deliv Rev. 2009;61(9):721–31.
Kanasty R, Dorkin JR, Vegas A, Anderson D. Delivery materials for siRNA therapeutics. Nat Mater. 2013;12(11):967–77.
Kolli S, Wong SP, Harbottle R, Johnston B, Thanou M, Miller AD. pH-triggered nanoparticle mediated delivery of siRNA to liver cells in vitro and in vivo. Bioconjug Chem. 2013;24(3):314–32.
Lin SY, Zhao WY, Tsai HC, Hsu WH, Lo CL, Hsiue GH. Sterically polymer-based liposomal complexes with dual-Shell structure for enhancing the siRNA delivery. Biomacromolecules. 2012;13(3):664–75.
Frankel AD, Pabo CO. Cellular uptake of the tat protein from human immunodeficiency virus. Cell. 1988;55(6):1189–93.
Endoh T, Ohtsuki T. Cellular siRNA delivery using cell-penetrating peptides modified for endosomal escape. Adv Drug Deliv Rev. 2009;61(9):704–9.
Meade BR, Dowdy SF. Exogenous siRNA delivery using peptide transduction domains/cell penetrating peptides. Adv Drug Deliv Rev. 2007;59(2–3):134–40.
Ullah I, Chung K, Beloor J, Kim J, Cho M, Kim N, et al. Trileucine residues in a ligand-CPP-based siRNA delivery platform improve endosomal escape of siRNA. J Drug Target. 2017;25(4):320–9.
Simeoni F, Morris MC, Heitz F, Divita G. Insight into the mechanism of the peptide-based gene delivery system MPG: implications for delivery of siRNA into mammalian cells. Nucleic Acids Res. 2003;31(11):2717–24.
Oehlke J, Scheller A, Wiesner B, Krause E, Beyermann M, Klauschenz E, et al. Cellular uptake of an alpha-helical amphipathic model peptide with the potential to deliver polar compounds into the cell interior non-endocytically. Bba-Biomembranes. 1998;1414(1–2):127–39.
Ye J, Liu E, Gong J, Wang J, Huang Y, He H, et al. High-yield synthesis of monomeric LMWP(CPP)-siRNA covalent conjugate for effective cytosolic delivery of siRNA. Theranostics. 2017;7(9):2495–508.
Xie X, Yang Y, Lin W, Liu H, Liu H, Yang Y, et al. Cell-penetrating peptide-siRNA conjugate loaded YSA-modified nanobubbles for ultrasound triggered siRNA delivery. Colloids Surf B: Biointerfaces. 2015;136:641–50.
Lee SH, Kang YY, Jang HE, Mok H. Current preclinical small interfering RNA (siRNA)-based conjugate systems for RNA therapeutics. Adv Drug Deliv Rev. 2016;104:78–92.
Bolhassani A. Potential efficacy of cell-penetrating peptides for nucleic acid and drug delivery in cancer. Biochim Biophys Acta Rev Cancer. 2011;1816(2):232–46.
Bhatia D, Surana S, Chakraborty S, Koushika SP, Krishnan Y. A synthetic icosahedral DNA-based host-cargo complex for functional in vivo imaging. Nat Commun. 2011;2.
Keum JW, Ahn JH, Bermudez H. Design, assembly, and activity of antisense DNA nanostructures. Small. 2011;7(24):3529–35.
Chen YP, Song S, Yan ZM, Fenniri H, Webster TJ. Self-assembled rosette nanotubes encapsulate and slowly release dexamethasone. Int J Nanomedicine. 2011;6:1035–44.
Song S, Chen YP, Yan ZM, Fenniri H, Webster TJ. Self-assembled rosette nanotubes for incorporating hydrophobic drugs in physiological environments. Int J Nanomedicine. 2011;6:101–7.
Lee H, Lytton-Jean AK, Chen Y, Love KT, Park AI, Karagiannis ED, et al. Molecularly self-assembled nucleic acid nanoparticles for targeted in vivo siRNA delivery. Nat Nanotechnol. 2012;7(6):389–93.
Shu Y, Shu D, Haque F, Guo PX. Fabrication of pRNA nanoparticles to deliver therapeutic RNAs and bioactive compounds into tumor cells. Nat Protoc. 2013;8(9):1635–59.
Sun X, Chen Y, Yu H, Machan JT, Alladin A, Ramirez J, et al. Anti-miRNA oligonucleotide therapy for chondrosarcoma. Mol Cancer Ther. 2019;18(11):2021–9.
Webster TJ, Chen Q, Chen Y, Fenniri H, Hemraz UD. Nanotubes as carriers of nucleic acids into cells. US10364440B2, 2014.
Chen Q, Chen Y, Yu H, Ehrlich MG. Nanocarriers And Their Processing For Diagnostics And Therapeutics. US9775842B2, 2015.
Chen Q, Yu H, Chen Y. Nanomaterials Compositions, Synthesis, and Assembly. US20170362238A1, 2017.
Dobrovolskaia MA. Self-assembled DNA/RNA nanoparticles as a new generation of therapeutic nucleic acids: immunological compatibility and other translational considerations. DNA RNA Nanotechnol. 2016;3(1):1–10.
Nixon AJ, Goodrich LR, Scimeca MS, Witte TH, Schnabel LV, Watts AE, et al. Gene therapy in musculoskeletal repair. Ann N Y Acad Sci. 2007;1117:310–27.
Andersson G. The burden of musculoskeletal diseases in the United States: prevalence. In: Societal and Economic Cost. Rosemont: American Academy of Orthopaedic Surgeons; 2008.
Evans CH, Huard J. Gene therapy approaches to regenerating the musculoskeletal system. Nat Rev Rheumatol. 2015;11(4):234–42.
Funding
This work is funded by 7R01AR072024 and 7R03AR069383 from DHHS/NIH/NIAMS and the CAREER award (1905785) from NSF.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Yupeng Chen is co-founder of NanoDe Therapeutics.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhou, L., Rubin, L.E., Liu, C. et al. Short Interfering RNA (siRNA)-Based Therapeutics for Cartilage Diseases. Regen. Eng. Transl. Med. 7, 283–290 (2021). https://doi.org/10.1007/s40883-020-00149-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40883-020-00149-z