Abstract
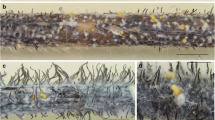
Southern Brazil has witnessed significant outbreaks of leaf blotch disease in barley over the last decade, leading to substantial losses in grain yield and quality. The increasing importance of this disease is mainly attributed to the shift towards cultivars with higher grain yields and superior malting quality, but they are more susceptible to leaf blotch. A field survey was conducted in the major barley-producing areas of Paraná state using DNA barcoding techniques to identify Bipolaris sorokiniana isolates, the causative agent of spot blotch, aiming to determine the extent of pathogenic variability among the isolates. DNA barcoding and phylogenetic analyses were based on internal transcribed spacers 1 and 2 with the 5.8S region (ITS) of rDNA, glyceraldehyde-3-phosphate dehydrogenase (gapdh), and translation elongation factor 1-α (tef1) genes. Out of the 124 isolates examined, which were collected from 20 commercial barley fields each year in 2020 and 2021, 116 isolates were identified as B. sorokiniana and eight isolates as B. gossypina. Koch's postulates confirmed the pathogenicity of B. gossypina in barley, representing a novel occurrence worldwide. The fungus causes the development of elongated brown lesions surrounded by irregular yellow halos, starting at minute points. Subtle differences between these symptoms and spot blotch caused by B. sorokiniana are discussed. This study also assessed the aggressiveness of 16 B. sorokiniana isolates on potted barley grown under controlled conditions, using a visual infection rate (IR) scale ranging from 1 to 9. Significant differences in aggressiveness were recorded among the isolates, with the IR ranging from 5.1 to 7.4 in the cultivar ANA03 and 5.7 to 8.1 in the cultivar Imperatriz. The interaction between the isolates and cultivars was not significant. These findings could support breeding programs aiming to develop cultivars with genetic resistance to spot blotch disease in Brazil.





Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author upon reasonable request.
References
Agostinetto L, Casa RT, Bogo A, Sachs C, Souza CA, Reis EM, da Cunha IC (2015) Barley spot blotch intensity, damage, and control response to foliar fungicide application in southern Brazil. Crop Protection 67:7–12
Ahmadpour A, Castell-Miller C, Javan-Nikkhah M, Naghavi MR, Dehkaei FP, Leng Y, Puri KD, Zhong S (2018) Population structure, genetic diversity, and sexual state of the rice brown spot pathogen Bipolaris oryzae from three Asian countries. Plant Pathology 67:181–192
Alkan M, Bayraktar H, İmren M, Özdemir F, Lahlali R, Mokrini F, Paulitz T, Dababat AA, Özer G (2022) Monitoring of host suitability and defense-related genes in wheat to Bipolaris sorokiniana. Journal of Fungi 8:149
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. Journal of Molecular Biology 215:403–410
Arnhold E (2013) Package in the R environment for analysis of variance and complementary analyses. Brazilian Journal of Veterinary Research and Animal Science 50:488–492
Berbee ML, Pirseyedi M, Hubbard S (1999) Cochliobolus phylogenetics and the origin of known, highly virulent pathogens, inferred from ITS and glyceraldehyde-3-phosphate dehydrogenase gene sequences. Mycologia 91:964–977
Bhunjun CS, Dong Y, Jayawardena RS, Jeewon R, Phukhamsakda C, Bundhun D, Hyde KD, Sheng J (2020) A polyphasic approach to delineate species in Bipolaris. Fungal Diversity 102:225–256
Cipollone J, Mourelos C, Sisterna M (2020) First report of Bipolaris zeicola on barley worldwide. Crop Protection 135:105188
CONAB (2023) Séries históricas, cevada. Available at: https://www.conab.gov.br/info-agro/safras/serie-historica-das-safras/itemlist/category/904-cevada. Accessed on June 07, 2023
Crous PW, Hawksworth DL, Wingfield MJ (2015) Identifying and naming plant-pathogenic fungi: Past, present, and future. Annual Review of Phytopathology 53:247–267
Crous PW, Shivas RG, Wingfield MJ, Summerell BA, Rossman AY, Alves JL, Adams GC, Barreto RW, Bell A, Coutinho ML, Flory SL, Gates G, Grice KR, Hardy GE, Kleczewski NM, Lombard L, Longa CM, Louis-Seize G, Macedo F, Mahoney DP, Maresi G, Martin-Sanchez PM, Marvanová L, Minnis AM, Morgado LN, Noordeloos ME, Phillips AJ, Quaedvlieg W, Ryan PG, Saiz-Jimenez C, Seifert KA, Swart WJ, Tan YP, Tanney JB, Thu PQ, Videira SI, Walker DM, Groenewald JZ (2012) Fungal planet description sheets: 128–153. Persoonia 29:146–201
FAOSTAT (2022) Crops. Available at: https://www.fao.org/faostat/en/#data/QCL/visualize. Accessed on December 15, 2022
Farr DF, Rossman AY (2023) Fungal Databases, U.S. National Fungus Collections, ARS, USDA. Available at: https://nt.ars-grin.gov/fungaldatabases/. Accessed on January 16, 2023
Ferdinandez HS, Manamgoda DS, Udayanga D, Deshappriya N, Munasinghe MS, Castlebury LA (2022) Molecular phylogeny and morphology reveal two new graminicolous species, Bipolaris adikaramae sp. nov and B. petchii sp. nov., with new records of fungi from cultivated rice and weedy grass hosts. Mycological Progress 21:59
Fetch TG, Steffenson BJ (1994) Identification of Cochliobolus sativus isolates expressing differential virulence on two-row barley genotypes from North Dakota. Canadian Journal Plant Pathology 16:202–206
Fetch TG, Steffenson BJ (1999) Rating scales for assessing infection responses of barley infected with Cochliobolus sativus. Plant Disease 83(3):213–217
Gamba FM, Finckh MR, Backes G (2020) Pathogenic variability of a Uruguayan population of Bipolaris sorokiniana in barley suggests a mix of quantitative and qualitative interactions. Journal of Plant Diseases and Protection 127:25–33
Ghazvini H (2018) The host-pathogen interaction between barley and casual agent of spot blotch (Bipolaris sorokiniana) disease: a review. Crop Breeding Journal 8:1–15
Ghazvini H, Tekauz A (2007) Virulence diversity in the population of Bipolaris sorokiniana. Plant Disease 91:814–821
Ghazvini H, Tekauz A (2008) Host–pathogen interactions among barley genotypes and Bipolaris sorokiniana isolates. Plant Disease 92:225–233
Hall T (2011) BioEdit: an important software for molecular biology. GERF Bulletin Biosciences 2:60–61
Kumar J, Schäfer P, Hückelhoven R, Langen G, Baltruschat H, Stein E, Nagarajan S, Kogel K (2002) Bipolaris sorokiniana, a cereal pathogen of global concern: cytological and molecular approaches towards better control. Molecular Plant Pathology 3:185–195
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: Molecular evolutionary genetis analysis across computing platforms. Molecular Biology and Evolution 35:1547–1949
Lourenço CCG, Alves JL, Guatimosim E, Colman A, Barreto RW (2017) Bipolaris marantae sp. nov., a novel Helminthosporoid species causing foliage blight of the garden plant Maranta leuconeura in Brazil. Mycobiology 45:123–128
Manamgoda DS, Rossman AY, Castlebury LA, Crous PW, Madrid H, Chukeatirote E, Hyde KD (2014) The genus Bipolaris. Studies in Mycology 79:221–288
Marin-Felix Y, Groenewald JZ, Cai L, Chen Q, Marincowitz S, Barnes I, Bensch K, Braun U, Camporesi E, Damm U, De Beer ZW, Dissanayake A, Edwards J, Giraldo A, Hernández-Restrepo M, Hyde KD, Jayawardena RS, Lombard L, Luangsa-Ard J, Mctaggart AR, Rossman AY, Sandoval-Denis M, Shen M, Shivas RG, Tan YP, van der Linde EJ, Wingfield MJ, Wood AR, Zhang JQ, Zhang Y, Crous PW (2017) Genera of phytopathogenic fungi: GOPHY 1. Studies in Mycology 86:99–216
Mathre DE (1997) Compendium of barley diseases. The American Phytopathological Society, Saint Paul
Miller MA, Pfeiffer W, Schwartz T (2010) Creating the CIPRES science gateway for inference of large phylogenetic trees. In: Proceedings of the gateway computing environments workshop (GCE), 14 Nov 2010. Gateway Computing Environments Workshop, New Orleans, pp 1–8. https://doi.org/10.1109/GCE.2010.5676129
Neupane A, Tamang P, Brueggeman RS, Friesen TL (2015) Evaluation of a barley core collection for spot form net blotch reaction reveals distinct genotype-specific pathogen virulence and host susceptibility. Phytopathology 105:509–517
R Core Team (2022) R: A language and environment for statistical computing, Vienna. Available at: https://www.R-project.org/. Accessed December 10, 2022
Rambaut A (2009) FigTree v1.4.0: Tree figure drawing tool, molecular evolution phylogenetics and epidemiology. Available at: http://tree.bio.ed.ac.uk/software/figtree/. Accessed December 15, 2022
Rehner SA, Buckley EA (2005) Beauveria phylogeny inferred from nuclear ITS and EF1-α sequences: Evidence for cryptic diversification and links to Cordyceps teleomorphs. Mycologia 97:84–98
Ronquist F, Huelsenbeck JP, Teslenko M (2012) MrBayes version 3.2 manual, tutorials and model sumaries, MrBayes on XSEDE. Available at: https://www.phylo.org/index.php/. CIPRESS Science Gateway. Accessed on December 19, 2022
Sharma P, Mishra S, Singroha G, Kumar RS, Singh SK, Singh GP (2022) Phylogeographic diversity analysis of Bipolaris sorokiniana (Sacc.) Shoemaker causing spot blotch disease in wheat and barley. Genes 13:2206
Sivanesan A (1985) New species of Bipolaris. Transactions of the British Mycological Society 84:403–421
Sivanesan A (1987) Graminicolus species of Bipolaris, Curvularia, Drechslera, Exerohilum and their teleomorphs. CAB International, Wallingford
Tan YP, Crous PW, Shivas RG (2016) Eight novel Bipolaris species identified from John L. Alcorn’s collections at the Queensland plant pathology herbarium (BRIP). Mycological Progress 15:1203–1214
Tan YP, Madrid H, Crous PW, Shivas RG (2014) Johnalcornia gen. et. comb. nov., and nine new combinations in Curvularia based on molecular phylogenetic analysis. Australasian Plant Pathology 43:589–603
White DG (ed) (1999) Compendium of corn diseases, vol 78. American Phytopathological Society Press, Saint Paul
White TJ, Bruns T, Lees TJ (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols, a guide to methods and applications. Academic Press, San Diego, California, pp 315–322
Zadoks JC, Chang TT, Konzak CF (1974) A decimal code for the growth stages of cereals. Weed Research 14:415–421
Acknowledgements
We thank Agrária Industrial Cooperativa of Guarapuava, PR, Brazil, for their help with sample collection.
Funding
This work was financially supported by the National Council for Scientific and Technological Development (CNPq) of Brazil (grant no. 308948/2020–3), the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior of Brazil (CAPES; finance code 001), the Fundação Agrária de Pesquisa Agropecuária (FAPA), and the Cooperativa Agrária Agroindustrial.
Author information
Authors and Affiliations
Contributions
Conceptualization, Rodrigues PCS and Tessmann DJ; investigation, Rodrigues PCS, Angelotti-Zampar P, and Seron G; writing and review, Feksa HR; supervision and project administration, Tessmann, DJ. All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
dos Santos Rodrigues, P.C., Angelotti-Zampar, P., Feksa, H.R. et al. DNA barcoding, aggressiveness of Bipolaris sorokiniana isolates, and pathogenicity of emerging B. gossypina in barley in subtropical southern Brazil. Trop. plant pathol. 49, 268–278 (2024). https://doi.org/10.1007/s40858-023-00628-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40858-023-00628-y