Abstract
Purpose
Diagnosis and treatment of Parkinson’s Disease (PD) are typically supported by a kinematic gait analysis. Nonetheless, the main drawbacks of the classical analysis, based on a reduced set of markers, are the loss of small dynamical changes, the invasive methodology, and the sparse representation from few points, restricting the disease analysis. This work aims to perform a robust regional kinematic characterization, which may result in a potential digital biomarker of the disease to complement personalized analysis, treatment and monitoring of PD.
Methods
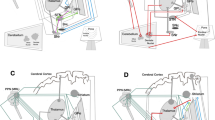
This work introduces a markerless computational framework based on a full body-segment kinematic characterization related with PD motor alterations. Firstly, a set of dense motion trajectories are computed to represent locomotion. Such trajectories are grouped using a deep learning based body segmentation, that partitions the human silhouette into regions corresponding to the head, trunk and limbs. Each resultant region is described using dartboard-like kinematic histograms computed along the trajectories.
Results
The proposed approach was validated using different pretrained classification models. The proposed method was evaluated on a set of 11 control subjects and 11 PD patients, achieving an average accuracy of \(99.62\%\) for lower-limbs and head regions.
Conclusion
This work proved to be effective to classify Parkinsonian patterns w.r.t control gaits. A major contribution of the proposed strategy is the capability to recover kinematic patterns in different body segments, particularly, for head and trunk regions, which turned out to be a decisive PD biomarker.






Similar content being viewed by others
References
Collaborators of, Global, G. B. D. (2016) Regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the global burden of disease study 2016. The Lancet 390:1211–1259.
Dorsey, E. R., & Bloem, B. R. (2018). The Parkinson pandemic-a call to action. JAMA Neurology, 75(1), 9–10.
Halliday, S. E., Winter, D. A., Frank, J. S., Patla, A. E., & Prince, F. (1998). The initiation of gait in young, elderly, and Parkinson’s disease subjects. Gait & Posture, 8(1), 8–14.
Duncan, R. P., et al. (2015). Balance differences in people with Parkinson disease with and without freezing of gait. Gait & Posture, 42(3), 306–309.
Cole, M. H., Naughton, G. A., & Silburn, P. A. (2017). Neuromuscular impairments are associated with impaired head and trunk stability during gait in Parkinson fallers. Neurorehabilitation and Neural Repair, 31(1), 34–47.
Winter, D. A. (1995). Human balance and posture control during standing and walking. Gait & Posture, 3(4), 193–214.
Sutherland, D. H. (2002). The evolution of clinical gait analysis: Part ii kinematics. Gait & Posture, 16(2), 159–179.
Ceseracciu, E., Sawacha, Z., & Cobelli, C. (2014). Comparison of markerless and marker-based motion capture technologies through simultaneous data collection during gait: proof of concept. PloS ONE, 9(3), e87640.
Pistacchi, M., et al. (2017). Gait analysis and clinical correlations in early Parkinson’s disease. Functional Neurology, 32(1), 28.
Naghavi, N., & Wade, E. (2019). Prediction of freezing of gait in Parkinson’s disease using statistical inference and lower-limb acceleration data. IEEE Transactions on Neural Systems and Rehabilitation Engineering, 27(5), 947–955.
Buckley, C., Galna, B., Rochester, L., & Mazzà, C. (2017). Quantification of upper body movements during gait in older adults and in those with Parkinson’s disease: Impact of acceleration realignment methodologies. Gait & Posture, 52, 265–271.
Barbe, M. T., et al. (2014). Gait and upper limb variability in Parkinson’s disease patients with and without freezing of gait. Journal of Neurology, 261(2), 330–342.
Dillmann, U., et al. (2014). Principal component analysis of gait in Parkinson’s disease: Relevance of gait velocity. Gait & Posture, 39(3), 882–887.
Vallabhajosula, S., Buckley, T. A., Tillman, M. D., & Hass, C. J. (2013). Age and Parkinson’s disease related kinematic alterations during multi-directional gait initiation. Gait & Posture, 37(2), 280–286.
Parisi, F., et al. (2015). Body-sensor-network-based kinematic characterization and comparative outlook of updrs scoring in leg agility, sit-to-stand, and gait tasks in Parkinson’s disease. IEEE Journal of Biomedical and Health Informatics, 19(6), 1777–1793.
Bovonsunthonchai, S., Vachalathiti, R., Pisarnpong, A., Khobhun, F., & Hiengkaew, V. (2014). Spatiotemporal gait parameters for patients with Parkinson’s disease compared with normal individuals. Physiotherapy Research International, 19(3), 158–165.
Verlekar, T. T., Soares, L. D., & Correia, P. L. (2018). Automatic classification of gait impairments using a markerless 2d video-based system. Sensors, 18(9), 2743.
Buckley, C., Galna, B., Rochester, L., & Mazzà, C. (2019). Upper body accelerations as a biomarker of gait impairment in the early stages of Parkinson’s disease. Gait & Posture, 71, 289–295.
Buckley, C., Galna, B., Rochester, L., & Mazzà, C. (2015). Attenuation of upper body accelerations during gait: piloting an innovative assessment tool for Parkinson’s disease. BioMed Research International, 2015, 865873.
Baby, M. S., Saji, A. & Kumar, C. S. (2017). Parkinsons disease classification using wavelet transform based feature extraction of gait data. In International conference on circuit, power and computing technologies (pp. 1–6). IEEE.
Lee, S.-H., & Lim, J. S. (2012). Parkinson’s disease classification using gait characteristics and wavelet-based feature extraction. Expert Systems with Applications, 39(8), 7338–7344.
Sarbaz, Y., Towhidkhah, F., Gharibzadeh, S., & Jafari, A. (2012). Gait spectral analysis: An easy fast quantitative method for diagnosing Parkinson’s disease. Journal of Mechanics in Medicine and Biology, 12(03), 1250041.
Choe, B.-Y., et al. (1998). Neuronal laterality in Parkinson’s disease with unilateral symptom by in vivo 1h magnetic resonance spectroscopy. Investigative Radiology, 33(8), 450–455.
Uitti, R. J., Baba, Y., Whaley, N., Wszolek, Z. K., & Putzke, J. (2005). Parkinson disease: Handedness predicts asymmetry. Neurology, 64(11), 1925–1930.
Kavanagh, J., Barrett, R., & Morrison, S. (2006). The role of the neck and trunk in facilitating head stability during walking. Experimental Brain Research, 172(4), 454.
Alp Güler, R., Neverova, N. & Kokkinos, I. (2018). Densepose: Dense human pose estimation in the wild, pp. 7297–7306.
Marsden, C. (1989). Slowness of movement in Parkinson’s disease. Movement Disorders: Official Journal of the Movement Disorder Society, 4(S1), S26–S37.
Wang, H. & Schmid, C. (2013). Action recognition with improved trajectories, (Sydney, Australia) http://hal.inria.fr/hal-00873267.
Wang, H., Kläser, A., Schmid, C. & Liu, C.-L. (2011). Action recognition by dense trajectories, 3169–3176 (IEEE).
Farnebäck, G. (2003). Two-frame motion estimation based on polynomial expansion, 363–370 (Springer).
Chang, C.-C., & Lin, C.-J. (2011). LIBSVM: A library for support vector machines. ACM Transactions on Intelligent Systems and Technology (TIST), 2(3), 27.
Breiman, L. (2001). Random forests. Machine Learning, 45(1), 5–32.
Lin, T.-Y. et al. (2014). Microsoft coco: Common objects in context, 740–755 (Springer).
Raptis, M., Kokkinos, I. & Soatto, S. (2012). Discovering discriminative action parts from mid-level video representations, 1242–1249 (IEEE).
Latt, M. D., Menz, H. B., Fung, V. S., & Lord, S. R. (2009). Acceleration patterns of the head and pelvis during gait in older people with Parkinson’s disease: A comparison of fallers and nonfallers. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences, 64(6), 700–706.
Balestrino, R., & Schapira, A. (2020). Parkinson disease. European Journal of Neurology, 27(1), 27–42.
Acknowledgements
The authors thank the FAMPAS Foundation (Fundación del Adulto Mayor y Parkinson Santander) and the nursing home Asilo San Rafael for their willingness to collaborate in this research by opening their doors. Additionally, support of the Vicerrectoría de Investigación y Extensión of the Universidad Industrial de Santander is gratefully acknowledged, through the funding of the project: Cuantificación de patrones locomotores para el diagnóstico y seguimiento remoto en zonas de dificil acceso, with SIVIE code 2697.
Funding
The Vicerrectoría de Investigación y Extensión of the Universidad Industrial de Santander supported this research registered by the project: “Cuantificación de patrones locomotores para el diagnóstico y seguimiento remoto en zonas de difícil acceso”, with SIVIE code 2697.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical Approval
This study was approved by the Ethical Committee of the Universidad Industrial de Santander (session held on July 19th, 2019) and counts on a written informed consent.
Employment
None.
Financial interests
None.
Non-financial interests
None.
Rights and permissions
About this article
Cite this article
Guayacán, L.C., Manzanera, A. & Martínez, F. Quantification of Parkinsonian Kinematic Patterns in Body-Segment Regions During Locomotion. J. Med. Biol. Eng. 42, 204–215 (2022). https://doi.org/10.1007/s40846-022-00691-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40846-022-00691-x