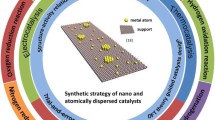
Abstract
The electrochemical nitrogen reduction reaction (NRR) technique has great potential for alleviating the high fossil fuel consumption and carbon emissions of the industrial Haber-Bosch method for ammonia (NH3) synthesis. Moreover, the NRR provides great prospects for fully exploiting renewable energy since NH3 is a promising energy carrier without carbon emissions. However, the development of the NRR technique is limited by the lack of efficient catalysts. Great efforts have been made to develop high-efficiency catalysts thus far, in which density functional theory (DFT) calculations have played an important role in assisting catalyst design. Herein, we summarize the recent catalyst design strategies to boost the NRR performance, i.e., the activity and selectivity. Additionally, representative computational studies are reviewed, accompanied by insights into further improving the catalytic behavior. Finally, we briefly discuss the challenges and opportunities in catalyst design via DFT calculations. The purpose of this review is to motivate more intelligent design strategies for high-efficiency NRR.

摘要
发展电化学氮气还原反应(NRR)的合成氨技术可以有效缓解工 业上的哈伯-博什法合成氨的化石燃料消耗与碳排放问题. 同时, 氨是 一种无碳的能源载体, NRR可以实现可再生能源的转换, 因此具有广阔 的发展前景. 然而, 高效催化剂的缺乏限制了NRR技术的发展. 为此, 人 们对开发高效催化剂进行了广泛的探索, 其中密度泛函理论(DFT)计算 在辅助催化剂设计方面发挥了重要作用. 在本综述中, 我们总结了最近 的催化剂设计策略, 这些策略的目的是提高NRR的催化活性和选择性. 此外, 本综述还回顾了具有代表性的计算工作, 并对进一步改善催化性 能提出了见解. 最后, 本综述简要讨论了通过DFT计算进行催化剂设计 所面临的挑战和机遇. 目的在于指导人们采用更有效的设计策略来实 现高效的NRR过程.
Article PDF
Similar content being viewed by others
References
MacFarlane DR, Cherepanov PV, Choi J, et al. A roadmap to the ammonia economy. Joule, 2020, 4: 1186–1205
Tang C, Qiao SZ. How to explore ambient electrocatalytic nitrogen reduction reliably and insightfully. Chem Soc Rev, 2019, 48: 3166–3180
Smil V. Detonator of the population explosion. Nature, 1999, 400: 415
Erisman JW, Sutton MA, Galloway J, et al. How a century of ammonia synthesis changed the world. Nat Geosci, 2008, 1: 636–639
Foster SL, Bakovic SIP, Duda RD, et al. Catalysts for nitrogen reduction to ammonia. Nat Catal, 2018, 1: 490–500
Deng J, Iñiguez JA, Liu C. Electrocatalytic nitrogen reduction at low temperature. Joule, 2018, 2: 846–856
Guo J, Chen P. Catalyst: NH3 as an Energy Carrier. Chem, 2017, 3: 709–712
Zhao Y, Setzler BP, Wang J, et al. An efficient direct ammonia fuel cell for affordable carbon-neutral transportation. Joule, 2019, 3: 2472–2484
Klerke A, Christensen CH, Nørskov JK, et al. Ammonia for hydrogen storage: Challenges and opportunities. J Mater Chem, 2008, 18: 2304
Chen JG, Crooks RM, Seefeldt LC, et al. Beyond fossil fuel-driven nitrogen transformations. Science, 2018, 360: eaar6611
Martín AJ, Shinagawa T, Pérez-Ramírez J. Electrocatalytic reduction of nitrogen: From Haber-Bosch to ammonia artificial leaf. Chem, 2019, 5: 263–283
Cheng H, Ding LX, Chen GF, et al. Molybdenum carbide nanodots enable efficient electrocatalytic nitrogen fixation under ambient conditions. Adv Mater, 2018, 30: e1803694
Qing G, Ghazfar R, Jackowski ST, et al. Recent advances and challenges of electrocatalytic N2 reduction to ammonia. Chem Rev, 2020, 120: 5437–5516
Giddey S, Badwal SPS, Munnings C, et al. Ammonia as a renewable energy transportation media. ACS Sustain Chem Eng, 2017, 5: 10231–10239
Olabi AG, bahri A, Abdelghafar AA, et al. Large-vscale hydrogen production and storage technologies: Current status and future directions. Int J Hydrogen Energy, 2021, 46: 23498–23528
Singstock NR, Musgrave CB. How the bioinspired Fe2Mo6S8 Chevrel breaks electrocatalytic nitrogen reduction scaling relations. J Am Chem Soc, 2022, 144: 12800–12806
Yao Y, Zhu S, Wang H, et al. A spectroscopic study on the nitrogen electrochemical reduction reaction on gold and platinum surfaces. J Am Chem Soc, 2018, 140: 1496–1501
Chen A, Xia BY. Ambient dinitrogen electrocatalytic reduction for ammonia synthesis. J Mater Chem A, 2019, 7: 23416–23431
Deutch J. Is net zero carbon 2050 possible? Joule, 2020, 4: 2237–2240
Soloveichik G. Electrochemical synthesis of ammonia as a potential alternative to the Haber-Bosch process. Nat Catal, 2019, 2: 377–380
Wang L, Xia M, Wang H, et al. Greening ammonia toward the solar ammonia refinery. Joule, 2018, 2: 1055–1074
Dinh CT, Jain A, de Arquer FPG, et al. Multi-site electrocatalysts for hydrogen evolution in neutral media by destabilization of water molecules. Nat Energy, 2018, 4: 107–114
Cheng H, Cui P, Wang F, et al. High efficiency electrochemical nitrogen fixation achieved with a lower pressure reaction system by changing the chemical equilibrium. Angew Chem Int Ed, 2019, 58: 15541–15547
Chen BWJ, Xu L, Mavrikakis M. Computational methods in heterogeneous catalysis. Chem Rev, 2021, 121: 1007–1048
Daw P, Ben-David Y, Milstein D. Acceptorless dehydrogenative coupling using ammonia: Direct synthesis of N-heteroaromatics from diols catalyzed by ruthenium. J Am Chem Soc, 2018, 140: 11931–11934
Chen ZW, Chen LX, Wen Z, et al. Understanding electro-catalysis by using density functional theory. Phys Chem Chem Phys, 2019, 21: 23782–23802
Zhao X, Levell ZH, Yu S, et al. Atomistic understanding of two-dimensional electrocatalysts from first principles. Chem Rev, 2022, 122: 10675–10709
Tian D, Denny SR, Li K, et al. Density functional theory studies of transition metal carbides and nitrides as electrocatalysts. Chem Soc Rev, 2021, 50: 12338–12376
Bhandari S, Rangarajan S, Mavrikakis M. Combining computational modeling with reaction kinetics experiments for elucidating the in situ nature of the active site in catalysis. Acc Chem Res, 2020, 53: 1893–1904
Liu B, Liu J, Xin L, et al. Unraveling reactivity descriptors and structure sensitivity in low-temperature NH3-SCR reaction over Ce-TiOx catalysts: A combined computational and experimental study. ACS Catal, 2021, 11: 7613–7636
Ziemba M, Schilling C, Ganduglia-Pirovano MV, et al. Toward an atomic-level understanding of ceria-based catalysts: When experiment and theory go hand in hand. Acc Chem Res, 2021, 54: 2884–2893
Zhang L, Ji X, Ren X, et al. Electrochemical ammonia synthesis via nitrogen reduction reaction on a MoS2 catalyst: Theoretical and experimental studies. Adv Mater, 2018, 30: e1800191
Li XF, Li QK, Cheng J, et al. Conversion of dinitrogen to ammonia by FeN3-embedded graphene. J Am Chem Soc, 2016, 138: 8706–8709
An Q, Shen Y, Fortunelli A, et al. QM-mechanism-based hierarchical high-throughput in silico screening catalyst design for ammonia synthesis. J Am Chem Soc, 2018, 140: 17702–17710
Liu JC, Ma XL, Li Y, et al. Heterogeneous Fe3 single-cluster catalyst for ammonia synthesis via an associative mechanism. Nat Commun, 2018, 9: 1610
Ohki Y, Munakata K, Matsuoka Y, et al. Nitrogen reduction by the Fe sites of synthetic [Mo3S4Fe] cubes. Nature, 2022, 607: 86–90
Rod TH, Logadottir A, Nørskov JK. Ammonia synthesis at low temperatures. J Chem Phys, 2000, 112: 5343–5347
Kordali V, Kyriacou G, Lambrou C. Electrochemical synthesis of ammonia at atmospheric pressure and low temperature in a solid polymer electrolyte cell. Chem Commun, 2000, 17: 1673–1674
Logadóttir Á, Nørskov JK. Ammonia synthesis over a Ru(0001) surface studied by density functional calculations. J Catal, 2003, 220: 273–279
Skúlason E, Bligaard T, Gudmundsdóttir S, et al. A theoretical evaluation of possible transition metal electro-catalysts for N2 reduction. Phys Chem Chem Phys, 2012, 14: 1235–1245
Zhou HY, Qu YB, Li JC, et al. Effectively boosting selective ammonia synthesis on electron-deficient surface of MoB2. Appl Catal B-Environ, 2022, 305: 121023
Shi L, Yin Y, Wang S, et al. Rational catalyst design for N2 reduction under ambient conditions: Strategies toward enhanced conversion efficiency. ACS Catal, 2020, 10: 6870–6899
van der Ham CJM, Koper MTM, Hetterscheid DGH. Challenges in reduction of dinitrogen by proton and electron transfer. Chem Soc Rev, 2014, 43: 5183–5191
Singh AR, Rohr BA, Schwalbe JA, et al. Electrochemical ammonia synthesis—The selectivity challenge. ACS Catal, 2016, 7: 706–709
Zhao X, Hu G, Chen GF, et al. Comprehensive understanding of the thriving ambient electrochemical nitrogen reduction reaction. Adv Mater, 2021, 33: e2007650
Ellingsson V, Iqbal A, Skúlason E, et al. Nitrogen reduction reaction to ammonia on transition metal carbide catalysts. ChemSusChem, 2023, 16: e202300947
Abghoui Y, Garden AL, Howalt JG, et al. Electroreduction of N2 to ammonia at ambient conditions on mononitrides of Zr, Nb, Cr, and V: A DFT guide for experiments. ACS Catal, 2015, 6: 635–646
Ling C, Zhang Y, Li Q, et al. New mechanism for N2 reduction: The essential role of surface hydrogenation. J Am Chem Soc, 2019, 141: 18264–18270
Cui X, Tang C, Zhang Q. A review of electrocatalytic reduction of dinitrogen to ammonia under ambient conditions. Adv Energy Mater, 2018, 8: 1800369
Ma XL, Liu JC, Xiao H, et al. Surface single-cluster catalyst for N2-to-NH3 thermal conversion. J Am Chem Soc, 2018, 140: 46–49
Jia HP, Quadrelli EA. Mechanistic aspects of dinitrogen cleavage and hydrogenation to produce ammonia in catalysis and organometallic chemistry: Relevance of metal hydride bonds and dihydrogen. Chem Soc Rev, 2014, 43: 547–564
Chen Z, Liu C, Sun L, et al. Progress of experimental and computational catalyst design for electrochemical nitrogen fixation. ACS Catal, 2022, 12: 8936–8975
Anderson JS, Rittle J, Peters JC. Catalytic conversion of nitrogen to ammonia by an iron model complex. Nature, 2013, 501: 84–87
Tayyebi E, Abghoui Y, Skúlason E. Elucidating the mechanism of electrochemical N2 reduction at the Ru(0001) electrode. ACS Catal, 2019, 9: 11137–11145
Légaré MA, Bélanger-Chabot G, Dewhurst RD, et al. Nitrogen fixation and reduction at boron. Science, 2018, 359: 896–900
Ling C, Niu X, Li Q, et al. Metal-free single atom catalyst for N2 fixation driven by visible light. J Am Chem Soc, 2018, 140: 14161–14168
Fan Y, Yu J, Song X, et al. The role of sp-hybridized boron atoms in the highly efficient photocatalytic N2 reduction activity of boron-doped triphenylene-graphdiyne. J Mater Chem A, 2021, 9: 26077–26085
Dai T, Lang X, Wang Z, et al. Rational design of an Fe cluster catalyst for robust nitrogen activation. J Mater Chem A, 2021, 9: 21219–21227
Chen ZW, Yan J, Jiang Q. Single or double: Which is the altar of atomic catalysts for nitrogen reduction reaction? Small Methods, 2019, 3: 1800291
Montoya JH, Tsai C, Vojvodic A, et al. The challenge of electrochemical ammonia synthesis: A new perspective on the role of nitrogen scaling relations. ChemSusChem, 2015, 8: 2180–2186
Suryanto BHR, Wang D, Azofra LM, et al. MoS2 polymorphic engineering enhances selectivity in the electrochemical reduction of nitrogen to ammonia. ACS Energy Lett, 2019, 4: 430–435
Li L, Martirez JMP, Carter EA. Prediction of highly selective electrocatalytic nitrogen reduction at low overpotential on a Mo-doped g-GaN monolayer. ACS Catal, 2020, 10: 12841–12857
Pang Y, Su C, Jia G, et al. Emerging two-dimensional nanomaterials for electrochemical nitrogen reduction. Chem Soc Rev, 2021, 50: 12744–12787
Liu S, Qian T, Wang M, et al. Proton-filtering covalent organic frameworks with superior nitrogen penetration flux promote ambient ammonia synthesis. Nat Catal, 2021, 4: 322–331
Hao YC, Guo Y, Chen LW, et al. Promoting nitrogen electroreduction to ammonia with bismuth nanocrystals and potassium cations in water. Nat Catal, 2019, 2: 448–456
Liu X, Jiao Y, Zheng Y, et al. Building up a picture of the electrocatalytic nitrogen reduction activity of transition metal single-atom catalysts. J Am Chem Soc, 2019, 141: 9664–9672
Jiao S, Fu X, Huang H. Descriptors for the evaluation of electrocatalytic reactions: d-band theory and beyond. Adv Funct Mater, 2021, 32: 2107651
Chen ZW, Chen LX, Yang CC, et al. Atomic (single, double, and triple atoms) catalysis: Frontiers, opportunities, and challenges. J Mater Chem A, 2019, 7: 3492–3515
Jin H, Guo C, Liu X, et al. Emerging two-dimensional nanomaterials for electrocatalysis. Chem Rev, 2018, 118: 6337–6408
Chen ZW, Chen LX, Jiang M, et al. A triple atom catalyst with ultrahigh loading potential for nitrogen electrochemical reduction. J Mater Chem A, 2020, 8: 15086–15093
Mounet N, Gibertini M, Schwaller P, et al. Two-dimensional materials from high-throughput computational exfoliation of experimentally known compounds. Nat Nanotech, 2018, 13: 246–252
Fei H, Dong J, Chen D, et al. Single atom electrocatalysts supported on graphene or graphene-like carbons. Chem Soc Rev, 2019, 48: 5207–5241
Mahmood J, Lee EK, Jung M, et al. Nitrogenated holey two-dimensional structures. Nat Commun, 2015, 6: 6486
Sun CN, Wang ZL, Lang XY, et al. Synergistic effect of active sites of double-atom catalysts for nitrogen reduction reaction. ChemSusChem, 2021, 14: 4593–4600
Guo X, Gu J, Lin S, et al. Tackling the activity and selectivity challenges of electrocatalysts toward the nitrogen reduction reaction via atomically dispersed biatom catalysts. J Am Chem Soc, 2020, 142: 5709–5721
Hui L, Xue Y, Yu H, et al. Highly efficient and selective generation of ammonia and hydrogen on a graphdiyne-based catalyst. J Am Chem Soc, 2019, 141: 10677–10683
Deng J, Deng D, Bao X. Robust catalysis on 2D materials encapsulating metals: Concept, application, and perspective. Adv Mater, 2017, 29: 1606967
Tang S, Dang Q, Liu T, et al. Realizing a not-strong-not-weak polarization electric field in single-atom catalysts sandwiched by boron nitride and graphene sheets for efficient nitrogen fixation. J Am Chem Soc, 2020, 142: 19308–19315
Lin G, Ju Q, Guo X, et al. Intrinsic electron localization of metastable MoS2 boosts electrocatalytic nitrogen reduction to ammonia. Adv Mater, 2021, 33: e2007509
Azofra LM, Sun C, Cavallo L, et al. Feasibility of N2 binding and reduction to ammonia on Fe-deposited MoS2 2D sheets: A DFT study. Chem Eur J, 2017, 23: 8275–8279
Luo Y, Chen GF, Ding L, et al. Efficient electrocatalytic N2 fixation with MXene under ambient conditions. Joule, 2019, 3: 279–289
Yao X, Chen Z, Wang Y, et al. Activated basal planes of WS2 by intrinsic defects as catalysts for the electrocatalytic nitrogen reduction reaction. J Mater Chem A, 2019, 7: 25961–25968
Gao Y, Yang Y, Hao L, et al. Single Nb atom modified anatase TiO2 (110) for efficient electrocatalytic nitrogen reduction reaction. Chem Catal, 2022, 2: 2275–2288
Huang T, Liu Z, Zhang Y, et al. Promoting electrocatalytic nitrogen reduction to ammonia via Fe-boosted nitrogen activation on MnO2 surfaces. J Mater Chem A, 2020, 8: 13679–13684
Jin F, Yin H, Feng R, et al. Charge transfer and vacancy engineering of Fe2O3 nanoparticle catalysts for highly selective N2 reduction towards NH3 synthesis. J Colloid Interface Sci, 2023, 647: 354–363
Zafari M, Kumar D, Umer M, et al. Machine learning-based high throughput screening for nitrogen fixation on boron-doped single atom catalysts. J Mater Chem A, 2020, 8: 5209–5216
Chen ZW, Lu Z, Chen LX, et al. Machine-learning-accelerated discovery of single-atom catalysts based on bidirectional activation mechanism. Chem Catal, 2021, 1: 183–195
Zhang J, Tian X, Liu M, et al. Cobalt-modulated molybdenum-dinitrogen interaction in MoS2 for catalyzing ammonia synthesis. J Am Chem Soc, 2019, 141: 19269–19275
Kong Y, He T, Puente Santiago AR, et al. Unravelling the reaction mechanisms of N2 fixation on molybdenum nitride: A full DFT study from the pristine surface to heteroatom anchoring. ChemSusChem, 2021, 14: 3257–3266
Jin H, Li L, Liu X, et al. Nitrogen vacancies on 2D layered W2N3: A stable and efficient active site for nitrogen reduction reaction. Adv Mater, 2019, 31: 1902709
Li P, Jin Z, Fang Z, et al. A surface-strained and geometry-tailored nanoreactor that promotes ammonia electrosynthesis. Angew Chem Int Ed, 2020, 59: 22610–22616
Zhang M, Yin H, Jin F, et al. Vacancy engineering of oxidized Nb2CTx MXenes for a biased nitrogen fixation. Green Energy Environ, 2023, 8: 1185–1194
Xia J, Guo H, Cheng M, et al. Electrospun zirconia nanofibers for enhancing the electrochemical synthesis of ammonia by artificial nitrogen fixation. J Mater Chem A, 2021, 9: 2145–2151
Wang S, Li B, Li L, et al. Highly efficient N2 fixation catalysts: Transition-metal carbides M2C (MXenes). Nanoscale, 2020, 12: 538–547
Huang B, Li N, Ong WJ, et al. Single atom-supported MXene: How single-atomic-site catalysts tune the high activity and selectivity of electrochemical nitrogen fixation. J Mater Chem A, 2019, 7: 27620–27631
Liu R, Guo T, Fei H, et al. Highly efficient electrocatalytic N2 reduction to ammonia over metallic 1T phase of MoS2 enabled by active sites separation mechanism. Adv Sci, 2022, 9: 2103583
Yin H, Du A. Boosting nitrogen reduction activity by defect engineering in 2D iron monochalcogenides FeX (X = S, Se). Small Struct, 2022, 3: 2200107
Zhao W, Zhang L, Luo Q, et al. Single Mo1(Cr1) atom on nitrogen-doped graphene enables highly selective electroreduction of nitrogen into ammonia. ACS Catal, 2019, 9: 3419–3425
Feng R, Yin H, Jin F, et al. Highly selective N2 electroreduction to NH3 using a boron-vacancy-rich diatomic NbB catalyst. Small, 2023, 19: 2301627
Bao D, Zhang Q, Meng FL, et al. Electrochemical reduction of N2 under ambient conditions for artificial N2 fixation and renewable energy storage using N2/NH3 cycle. Adv Mater, 2017, 29: 1604799
Wang J, Yu L, Hu L, et al. Ambient ammonia synthesis via palladium-catalyzed electrohydrogenation of dinitrogen at low overpotential. Nat Commun, 2018, 9: 1795
Zhou HY, Qu YB, Fan YC, et al. Multi-site intermetallic Ni3Mo effectively boosts selective ammonia synthesis. Appl Catal B-Environ, 2023, 339: 123133
Yin H, Bell S, Golberg D, et al. Enhancing the nitrogen reduction activity of iron with inactive group-IVA elements at optimized stoichiometry. Inorg Chem Front, 2023, 10: 4442–4449
Yin H, Du A. Revealing the potential of ternary medium-entropy alloys as exceptional electrocatalysts toward nitrogen reduction: An example of Heusler alloys. ACS Appl Mater Interfaces, 2022, 14: 15235–15242
Hannagan RT, Giannakakis G, Flytzani-Stephanopoulos M, et al. Single-atom alloy catalysis. Chem Rev, 2020, 120: 12044–12088
Greiner MT, Jones TE, Beeg S, et al. Free-atom-like d states in single-atom alloy catalysts. Nat Chem, 2018, 10: 1008–1015
Dai T, Wang Z, Lang X, et al. “Sabatier principle” of d electron number for describing the nitrogen reduction reaction performance of single-atom alloy catalysts. J Mater Chem A, 2022, 10: 16900–16907
Zhang Y, Li S, Sun C, et al. Understanding and modifying the scaling relations for ammonia synthesis on dilute metal alloys: From singleatom alloys to dimer alloys. ACS Catal, 2022, 12: 9201–9212
Yu X, Han P, Wei Z, et al. Boron-doped graphene for electrocatalytic N2 reduction. Joule, 2018, 2: 1610–1622
Liu C, Li Q, Wu C, et al. Single-boron catalysts for nitrogen reduction reaction. J Am Chem Soc, 2019, 141: 2884–2888
Li H, Yang L, Wang Z, et al. N-heterocyclic carbene as a promising metal-free electrocatalyst with high efficiency for nitrogen reduction to ammonia. J Energy Chem, 2020, 46: 78–86
Wen L, Sun K, Liu X, et al. Electronic state and microenvironment modulation of metal nanoparticles stabilized by MOFs for boosting electrocatalytic nitrogen reduction. Adv Mater, 2023, 35: 2210669
Ling C, Ouyang Y, Li Q, et al. A general two-step strategy-based high-throughput screening of single atom catalysts for nitrogen fixation. Small Methods, 2019, 3: 1800376
Liu D, Chen M, Du X, et al. Development of electrocatalysts for efficient nitrogen reduction reaction under ambient condition. Adv Funct Mater, 2021, 31: 2008983
Ren Y, Yu C, Tan X, et al. Strategies to suppress hydrogen evolution for highly selective electrocatalytic nitrogen reduction: Challenges and perspectives. Energy Environ Sci, 2021, 14: 1176–1193
Sun CN, Qu YB, Wang ZL, et al. Hydrogen spillover in alkaline solutions for effective nitrogen fixation. Chem Eng J, 2023, 471: 144589
Lv X, Kou L, Frauenheim T. Hydroxyl-boosted nitrogen reduction reaction: The essential role of surface hydrogen in functionalized MXenes. ACS Appl Mater Interfaces, 2021, 13: 14283–14290
Wang T, Abild-Pedersen F. Achieving industrial ammonia synthesis rates at near-ambient conditions through modified scaling relations on a confined dual site. Proc Natl Acad Sci USA, 2021, 118: e2106527118
Xue ZH, Zhang SN, Lin YX, et al. Electrochemical reduction of N2 into NH3 by donor-acceptor couples of Ni and Au nanoparticles with a 67.8% faradaic efficiency. J Am Chem Soc, 2019, 141: 14976–14980
Shi MM, Bao D, Wulan BR, et al. Au sub-nanoclusters on TiO2 toward highly efficient and selective electrocatalyst for N2 conversion to NH3 at ambient conditions. Adv Mater, 2017, 29: 1606550
Han M, Guo M, Yun Y, et al. Effect of heteroatom and charge reconstruction in atomically precise metal nanoclusters on electrochemical synthesis of ammonia. Adv Funct Mater, 2022, 32: 2202820
Dai T, Wang Z, Wen Z, et al. Tailoring electronic structure of copper twin boundaries toward highly efficient nitrogen reduction reaction. ChemSusChem, 2022, 15: e202201189
Fan G, Xu W, Li J, et al. Nanoporous NiSb to enhance nitrogen electroreduction via tailoring competitive adsorption sites. Adv Mater, 2021, 33: e2101126
Wu T, Melander MM, Honkala K. Coadsorption of NRR and HER intermediates determines the performance of Ru-N4 toward electro-catalytic N2 reduction. ACS Catal, 2022, 12: 2505–2512
Guo Y, Gu J, Zhang R, et al. Molecular crowding effect in aqueous electrolytes to suppress hydrogen reduction reaction and enhance electrochemical nitrogen reduction. Adv Energy Mater, 2021, 11: 2101699
Li L, Tang C, Xia B, et al. Two-dimensional mosaic bismuth nanosheets for highly selective ambient electrocatalytic nitrogen reduction. ACS Catal, 2019, 9: 2902–2908
Li Z, Fu JY, Feng Y, et al. A silver catalyst activated by stacking faults for the hydrogen evolution reaction. Nat Catal, 2019, 2: 1107–1114
Chen D, Pu Z, Wang P, et al. Mapping hydrogen evolution activity trends of intermetallic Pt-group silicides. ACS Catal, 2022, 12: 2623–2631
Kong Y, Li Y, Sang X, et al. Atomically dispersed zinc(I) active sites to accelerate nitrogen reduction kinetics for ammonia electrosynthesis. Adv Mater, 2021, 34: e2103548
Choi C, Gu GH, Noh J, et al. Understanding potential-dependent competition between electrocatalytic dinitrogen and proton reduction reactions. Nat Commun, 2021, 12: 4353
McCrum IT, Koper MTM. The role of adsorbed hydroxide in hydrogen evolution reaction kinetics on modified platinum. Nat Energy, 2020, 5: 891–899
Kim K, Yoo CY, Kim JN, et al. Electrochemical synthesis of ammonia from water and nitrogen in ethylenediamine under ambient temperature and pressure. J Electrochem Soc, 2016, 163: F1523–F1526
Lazouski N, Steinberg KJ, Gala ML, et al. Proton donors induce a differential transport effect for selectivity toward ammonia in lithiummediated nitrogen reduction. ACS Catal, 2022, 12: 5197–5208
Steinberg K, Yuan X, Klein CK, et al. Imaging of nitrogen fixation at lithium solid electrolyte interphases via cryo-electron microscopy. Nat Energy, 2022, 8: 138–148
Cai X, Li X, You J, et al. Lithium-mediated ammonia electrosynthesis with ether-based electrolytes. J Am Chem Soc, 2023, 145: 25716–25725
Li K, Andersen SZ, Statt MJ, et al. Enhancement of lithium-mediated ammonia synthesis by addition of oxygen. Science, 2021, 374: 1593–1597
Fu X, Pedersen JB, Zhou Y, et al. Continuous-flow electrosynthesis of ammonia by nitrogen reduction and hydrogen oxidation. Science, 2023, 379: 707–712
Chen G, Ren S, Zhang L, et al. Advances in electrocatalytic N2 reduction—Strategies to tackle the selectivity challenge. Small Methods, 2019, 3: 1800337
Renner JN, Greenlee LF, Ayres KE, et al. Electrochemical synthesis of ammonia: A low pressure, low temperature approach. Interface Mag, 2015, 24: 51–57
Liu H, Zhang Y, Luo J. The removal of inevitable NO species in catalysts and the selection of appropriate membrane for measuring electrocatalytic ammonia synthesis accurately. J Energy Chem, 2020, 49: 51–58
Ren Y, Yu C, Tan X, et al. Is it appropriate to use the nafion membrane in electrocatalytic N2 reduction? Small Methods, 2019, 3: 1900474
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21673095).
Author information
Authors and Affiliations
Contributions
Author contributions Jiang Q supervised the project and conceived the idea. Dai TY wrote the draft. Yang CC revised the manuscript and provided constructive suggestions. All authors discussed and commented on the manuscript.
Corresponding authors
Additional information
Tian-Yi Dai received his BE and PhD degrees from Jilin University in 2018 and 2023, respectively. He is currently working as a postdoctoral fellow at the School of Materials Science and Engineering, Jilin University. His current research focuses on the catalyst design through density functional theory calculations. Some of his studies are carried out in close collaboration with experiments.
Chun-Cheng Yang received his BSc and PhD degrees from Jilin University in 2001 and 2006, respectively. He is currently working as a professor at the School of Materials Science and Engineering, Jilin University. He had worked as a postdoctoral fellow at The University of New South Wales and The University of Sydney for 7 years. His research interests focus on micro- and nano-scale energy materials.
Qing Jiang obtained his PhD degree in chemistry from the University of Stuttgart, Germany in 1990. Since 1992, he has been a professor at the School of Materials Science and Engineering, Jilin University, China. His research interests focus on the synthesis of nanomaterials as well as their applications in catalysis, energy storage and conversion, and interface thermodynamics and kinetics of nanomaterials.
Rights and permissions
About this article
Cite this article
Dai, TY., Yang, CC. & Jiang, Q. Recent progress on catalyst design of nitrogen reduction reaction by density functional theory. Sci. China Mater. 67, 1101–1123 (2024). https://doi.org/10.1007/s40843-023-2847-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-023-2847-1