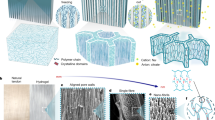
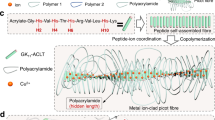
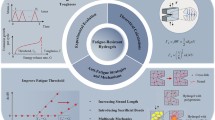
Abstract
Biological tissues (such as tendons and cartilages) are not only well known for their excellent strength, modulus, and toughness, but also have long-term stability which can withstand millions of high-stress cycles without fracture, and show a fatigue threshold of more than 1000 J m−2. In contrast, although synthetic hydrogels are comparable to natural soft tissues in terms of strength, modulus, toughness, and other properties, these toughened gels will still be subjected to fatigue fracture under repeated cyclic loading, exhibiting a fatigue threshold usually below 100 J m−2. Here, we report a simple strategy for the development of tough and fatigue-resistant anisotropic hydrogels with a fatigue threshold more than 100 times that of conventional hydrogels. Our two-step process mainly includes the formation of an arranged polydopamine-Fe3O4-carbon fiber structure, followed by freezing-thawing and annealing, synergistically contributing to the ultrahigh strength and toughness, excellent tribological properties, and extraordinary fatigue resistance. The tensile strength, compressive strength, and fatigue threshold of the anisotropic hydrogel were up to 11.82 ± 0.85 MPa, 5.95 ± 0.35 MPa, and 1845 J m−2, respectively, which were significantly higher than those of most biogels and synthetic hydrogels. Therefore, this research provides a feasible method for manufacturing soft materials with excellent properties and expands the application of soft materials in load-bearing materials, soft robots, flexible electronics, etc.

摘要
生物组织(如肌腱、软骨等)不仅具有优异的强度、模量和韧性, 而且具有长期稳定性, 可承受数百万次高应力循环而不断裂, 其疲劳阈 值超过1000 J m−2. 相比之下, 尽管合成水凝胶在强度、模量、韧性和 其他性能方面与天然软组织相当, 但这些增韧水凝胶仍然会在反复循 环载荷下遭受疲劳断裂, 其疲劳阈值通常低于100 J m−2. 在本工作中, 我们报道了一种简单的策略, 用于开发韧性和抗疲劳的各向异性水凝 胶, 其疲劳阈值超过常规水凝胶的100倍. 各向异性水凝胶通过两步工 艺合成, 包括磁场定向工艺形成优先排列的PDA-Fe3O4-CF纤维结构, 以及冷冻解冻-退火工艺处理, 优先排列纤维结构和高结晶度的协同作 用具有超高的强度和韧性、优异的摩擦学性能和非凡的抗疲劳性能. 各向异性水凝胶的抗拉强度、抗压强度和疲劳阈值分别高达11.82 ± 0.85 MPa、5.95 ±0.35 MPa和1845 J m−2, 显著高于大多数生物凝胶和 合成水凝胶. 因此, 本研究为制造性能优异的软材料提供了一种可行的 方法, 拓展了软质材料在承重材料(如人工软组织)、软机器人、柔性电 子等领域的应用.
Similar content being viewed by others
References
Mredha MTI, Jeon I. Biomimetic anisotropic hydrogels: Advanced fabrication strategies, extraordinary functionalities, and broad applications. Prog Mater Sci, 2021, 124: 100870
Zhao Z, Wang Z, Li G, et al. Injectable microfluidic hydrogel microspheres for cell and drug delivery. Adv Funct Mater, 2021, 31: 2103339
Ilami M, Bagheri H, Ahmed R, et al. Materials, actuators, and sensors for soft bioinspired robots. Adv Mater, 2021, 33: 2003139
Jiang L, Lu X. Functional hydrogel-based supercapacitors for wearable bioelectronic devices. Mater Chem Front, 2021, 5: 7479–7498
Gu Z, Huang K, Luo Y, et al. Double network hydrogel for tissue engineering. WIREs Nanomed Nanobiotechnol, 2018, 10: e1520
Zhao H, Liu M, Zhang Y, et al. Nanocomposite hydrogels for tissue engineering applications. Nanoscale, 2020, 12: 14976–14995
Beckett LE, Lewis JT, Tonge TK, et al. Enhancement of the mechanical properties of hydrogels with continuous fibrous reinforcement. ACS BioMater Sci Eng, 2020, 6: 5453–5473
Zhang G, Chen S, Peng Z, et al. Topologically enhanced dual-network hydrogels with rapid recovery for low-hysteresis, self-adhesive epidemic electronics. ACS Appl Mater Interfaces, 2021, 13: 12531–12540
Zhao Z, Fang R, Rong Q, et al. Bioinspired nanocomposite hydrogels with highly ordered structures. Adv Mater, 2017, 29: 1703045
Choi S, Choi Y, Kim J. Anisotropic hybrid hydrogels with superior mechanical properties reminiscent of tendons or ligaments. Adv Funct Mater, 2019, 29: 1904342
Yang F, Zhao J, Koshut WJ, et al. A synthetic hydrogel composite with the mechanical behavior and durability of cartilage. Adv Funct Mater, 2020, 30: 2003451
Sun JY, Zhao X, Illeperuma WRK, et al. Highly stretchable and tough hydrogels. Nature, 2012, 489: 133–136
Gong JP, Katsuyama Y, Kurokawa T, et al. Double-network hydrogels with extremely high mechanical strength. Adv Mater, 2003, 15: 11551158
Qiao Z, Cao M, Michels K, et al. Design and fabrication of highly stretchable and tough hydrogels. Polym Rev, 2020, 60: 420–441
Bai R, Yang Q, Tang J, et al. Fatigue fracture of tough hydrogels. Extreme Mech Lett, 2017, 15: 91–96
Lin S, Liu X, Liu J, et al. Anti-fatigue-fracture hydrogels. Sci Adv, 2019, 5: eaau8528
Bai R, Yang J, Morelle XP, et al. Fatigue fracture of self-recovery hydrogels. ACS Macro Lett, 2018, 7: 312–317
Zhang W, Liu X, Wang J, et al. Fatigue of double-network hydrogels. Eng Fract Mech, 2018, 187: 74–93
Bircher K, Zündel M, Pensalfini M, et al. Tear resistance of soft collagenous tissues. Nat Commun, 2019, 10: 792
Taylor D, O’Mara N, Ryan E, et al. The fracture toughness of soft tissues. J Mech Behav BioMed Mater, 2012, 6: 139–147
Liang X, Chen G, Lin S, et al. Anisotropically fatigue-resistant hydrogels. Adv Mater, 2021, 33: 2102011
Lin S, Liu J, Liu X, et al. Muscle-like fatigue-resistant hydrogels by mechanical training. Proc Natl Acad Sci USA, 2019, 116: 10244–10249
Hu D, Cui Y, Mo K, et al. Ultrahigh strength nanocomposite hydrogels designed by locking oriented tunicate cellulose nanocrystals in polymeric networks. Compos Part B-Eng, 2020, 197: 108118
Chau M, De France KJ, Kopera B, et al. Composite hydrogels with tunable anisotropic morphologies and mechanical properties. Chem Mater, 2016, 28: 3406–3415
Hua M, Wu S, Ma Y, et al. Strong tough hydrogels via the synergy of freeze-casting and salting out. Nature, 2021, 590: 594–599
Liu K, Han L, Tang P, et al. An anisotropic hydrogel based on mussel-inspired conductive ferrofluid composed of electromagnetic nanohybrids. Nano Lett, 2019, 19: 8343–8356
Liu M, Ishida Y, Ebina Y, et al. An anisotropic hydrogel with electrostatic repulsion between cofacially aligned nanosheets. Nature, 2015, 517: 68–72
Lu Q, Bai S, Ding Z, et al. Hydrogel assembly with hierarchical alignment by balancing electrostatic forces. Adv Mater Inter, 2016, 3: 1500687
Shi W, Huang J, Fang R, et al. Imparting functionality to the hydrogel by magnetic-field-induced nano-assembly and macro-response. ACS Appl Mater Interfaces, 2020, 12: 5177–5194
Hua M, He X. Soft-fiber-reinforced tough and fatigue resistant hydrogels. Matter, 2021, 4: 1755–1757
Ni J, Lin S, Qin Z, et al. Strong fatigue-resistant nanofibrous hydrogels inspired by lobster underbelly. Matter, 2021, 4: 1919–1934
Yang H, Ji M, Yang M, et al. Fabricating hydrogels to mimic biological tissues of complex shapes and high fatigue resistance. Matter, 2021, 4: 1935–1946
Li X, Cui K, Kurokawa T, et al. Effect of mesoscale phase contrast on fatigue-delaying behavior of self-healing hydrogels. Sci Adv, 2021, 7: eabe8210
Rong M, Liu H, Scaraggi M, et al. High lubricity meets load capacity: Cartilage mimicking bilayer structure by brushing up stiff hydrogels from subsurface. Adv Funct Mater, 2020, 30: 2004062
Chen Q, Zhang X, Chen K, et al. Anisotropic hydrogels with enhanced mechanical and tribological performance by magnetically oriented nanohybrids. Chem Eng J, 2022, 430: 133036
Frank E, Steudle LM, Ingildeev D, et al. Carbon fibers: Precursor systems, processing, structure, and properties. Angew Chem Int Ed, 2014, 53: 5262–5298
Luo H, Xiong G, Yang Z, et al. Preparation of three-dimensional braided carbon fiber-reinforced PEEK composites for potential load-bearing bone fixations. Part I. Mechanical properties and cyto-compatibility. J Mech Behav BioMed Mater, 2014, 29: 103–113
Gupta AK, Gupta M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials, 2005, 26: 3995–4021
Liu Y, Ai K, Lu L. Polydopamine and its derivative materials: Synthesis and promising applications in energy, environmental, and biomedical fields. Chem Rev, 2014, 114: 5057–5115
Lee H, Dellatore SM, Miller WM, et al. Mussel-inspired surface chemistry for multifunctional coatings. Science, 2007, 318: 426–430
Fu Y, Han C, Ni Q. Effects of surface modification on the dispersion property of VGC. Chin J Chem, 2009, 27: 1110–1116
Holloway JL, Lowman AM, Palmese GR. The role of crystallization and phase separation in the formation of physically cross-linked PVA hydrogels. Soft Matter, 2013, 9: 826–833
Bodugoz-Senturk H, Choi J, Oral E, et al. The effect of polyethylene glycol on the stability of pores in polyvinyl alcohol hydrogels during annealing. Biomaterials, 2008, 29: 141–149
Bodugoz-Senturk H, Macias CE, Kung JH, et al. Poly(vinyl alcohol)-acrylamide hydrogels as load-bearing cartilage substitute. Biomaterials, 2009, 30: 589–596
Chen K, Chen G, Wei S, et al. Preparation and property of high strength and low friction PVA-HA/PAA composite hydrogel using annealing treatment. Mater Sci Eng-C, 2018, 91: 579–588
Gan S, Lin W, Zou Y, et al. Nano-hydroxyapatite enhanced double network hydrogels with excellent mechanical properties for potential application in cartilage repair. Carbohydrate Polyms, 2020, 229: 115523
Yu Y, Zhu C, Liu Y, et al. Synthesis and characterization of N-maleyl chitosan-cross-linked poly(acrylamide)/montmorillonite nanocomposite hydrogels. Polym-Plasts Tech Eng, 2011, 50: 525–529
Bai R, Yang J, Morelle XP, et al. Flaw-insensitive hydrogels under static and cyclic loads. Macromol Rapid Commun, 2019, 40: 1800883
Yang W, Sherman VR, Gludovatz B, et al. On the tear resistance of skin. Nat Commun, 2015, 6: 6649
Gillies AR, Lieber RL. Structure and function of the skeletal muscle extracellular matrix. Muscle Nerve, 2011, 44: 318–331
Little CJ, Bawolin NK, Chen X. Mechanical properties of natural cartilage and tissue-engineered constructs. Tissue Eng Part B-Rev, 2011, 17: 213–227
Chen Y, Jiao C, Peng X, et al. Biomimetic anisotropic poly(vinyl alcohol) hydrogels with significantly enhanced mechanical properties by freezing-thawing under drawing. J Mater Chem B, 2019, 7: 3243–3249
Wang X, Fang J, Zhu W, et al. Bioinspired highly anisotropic, ultra-strong and stiff, and osteoconductive mineralized wood hydrogel composites for bone repair. Adv Funct Mater, 2021, 31: 2010068
Sun Q, Ma S, Lin P, et al. Anisotropic hydrogels with high mechanical strength by stretching-induced oriented crystallization and drying. ACS Appl Polym Mater, 2020, 2: 2142–2150
Acknowledgements
This work was financially supported by the Natural Science Foundation of Jiangsu Province (BK20211243), Jiangsu Provincial Key Research and Development Program (BE2022708), the Tribology Science Fund of State Key Laboratory of Tribology in Advanced Equipment (SKLTKF21B15), and the Open Fund of State Key Laboratory of Solid Lubrication, Lanzhou Institute of Chemical Physics (LSL-2107).
Author information
Authors and Affiliations
Contributions
Author contributions Chen K and Zhang D guided the project. Chen Q carried out the experimental section and wrote the paper with support from Chen K. All authors contributed to the general discussion.
Corresponding authors
Ethics declarations
Conflict of interest The authors declare that they have no conflict of interest.
Additional information
Supplementary information Experimental details and supporting data are available in the online version of this paper.
Qin Chen is studying for her PhD degree at China University of Mining and Technology. Her current research interests mainly focus on the bionic design of functional materials, the tribology of soft materials, and the intelligent fault diagnosis of mechanical equipment.
Kai Chen received his PhD degree from China University of Mining and Technology in 2015. He is currently an associate professor at China University of Mining and Technology. His current research interests cover the bio-nic design of functional materials, tribology of soft materials, bio-tribology of artificial joints, and wear-resistant materials.
Dekun Zhang received his PhD degree from China University of Mining and Technology in 2003. He has been a professor at China University of Mining and Technology since 2008. His current research interests cover fretting, friction reliability of mining machinery, bio-tribology of artificial joints, bionic design of functional materials, and wear-resistant materials.
Rights and permissions
About this article
Cite this article
Chen, Q., Chen, K., Wu, M. et al. Tough and fatigue-resistant anisotropic hydrogels via fiber reinforcement and magnetic field induction. Sci. China Mater. 66, 4841–4852 (2023). https://doi.org/10.1007/s40843-023-2639-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-023-2639-0