Abstract
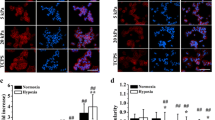
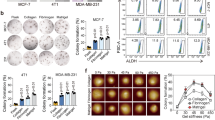
It has long been hypothesized that an increase in the extracellular matrix (ECM) stiffness mechanoactivates malignant phenotypes of breast tumor cells by regulating an array of processes underlying cancer biology. Although the contribution of substrate stiffening to drive malignant phenotype traits and other biological functions of a tumor is increasingly understood, the functional role of substrate softening on breast cancer cellular responses has rarely been investigated. Herein, we employed matrix metalloproteinase (MMP)-sensitive film to perform assays to explore the consequences of lowering stiffness on the biological behaviors of breast cancer cell MDA-MB-231. We demonstrated that cells underwent dramatic changes in migration, cellular conjunction, and expression of malignance-associated proteins and genes when the substrate stiffness decreased. Based on RNA sequencing and analysis, we found that hub genes including TP53, CCND1, MYC, CTNNB1, and YAP were validated to play central parts in regulating stiffness-dependent cellular manner change. Moreover, through visualization of differentially expressed genes (DEGs), cells on dynamically softened substrate appeared less influenced by transfer to tissue culture polystyrene (TCPS). These results suggest substrates with decreasing stiffness could normalize breast tumor malignant phenotype and help cells store the mechanical memory of the consequential weaker malignance.
摘要
肿瘤的发生发展伴随着细胞外基质的重塑和硬化. 目前, 尽 管细胞外基质的硬度已经成为一种强有力的肿瘤细胞行为调节器, 但基底硬度动态下降对乳腺癌细胞行为的影响却鲜有研究. 在此, 我们采用基质金属蛋白酶敏感的聚电解质多层膜进行分析, 探讨 硬度的降低对乳腺癌细胞MDA-MB-231生物学行为的影响. 结果 表明, 与在静态高硬度基底上培养的细胞相比, 在硬度动态降低的 基底上培养时, 细胞的迁移、粘附及恶性相关蛋白和基因表达水 平发生了显著变化, 肿瘤恶性降低. 根据蛋白质相互作用PPI网络 分析结果, 中枢节点基因TP53, CCND1, MYC, CTNNB1和YAP被 认为在硬度依赖性细胞行为变化中起着核心作用. 此外, 将在动态 软化基底上培养的细胞转移到高硬度材料上之后, 细胞仍保持较 弱的恶性, 表现出一定的记忆性.
Article PDF
Similar content being viewed by others
Reference
Chaudhuri O, Koshy ST, Branco da Cunha C, et al. Extracellular matrix stiffness and composition jointly regulate the induction of malignant phenotypes in mammary epithelium. Nat Mater, 2014, 13: 970–978
Yu H, Mouw JK, Weaver VM. Forcing form and function: Biomechanical regulation of tumor evolution. Trends Cell Biol, 2011, 21: 47–56
Acerbi I, Cassereau L, Dean I, et al. Human breast cancer invasion and aggression correlates with ECM stiffening and immune cell infiltration. Integrative Biol, 2015, 7: 1120–1134
Ishihara S, Inman DR, Li WJ, et al. Mechano-signal transduction in mesenchymal stem cells induces prosaposin secretion to drive the proliferation of breast cancer cells. Cancer Res, 2017, 77: 6179–6189
Lu P, Weaver VM, Werb Z. The extracellular matrix: A dynamic niche in cancer progression. J Cell Biol, 2012, 196: 395–406
Wei SC, Yang J. Forcing through tumor metastasis: the interplay between tissue rigidity and epithelial-mesenchymal transition. Trends Cell Biol, 2016, 26: 111–120
Wei SC, Fattet L, Tsai JH, et al. Matrix stiffness drives epithelial-mesenchymal transition and tumour metastasis through a TWIST1-G3BP2 mechanotransduction pathway. Nat Cell Biol, 2015, 17: 678–688
Ondeck MG, Kumar A, Placone JK, et al. Dynamically stiffened matrix promotes malignant transformation of mammary epithelial cells via collective mechanical signaling. Proc Natl Acad Sci USA, 2019, 116: 3502–3507
Medina SH, Bush B, Cam M, et al. Identification of a mechan-ogenetic link between substrate stiffness and chemotherapeutic response in breast cancer. Biomaterials, 2019, 202: 1–11
Mah EJ, Lefebvre AEYT, McGahey GE, et al. Collagen density modulates triple-negative breast cancer cell metabolism through adhesion-mediated contractility. Sci Rep, 2018, 8: 17094
Paszek MJ, Zahir N, Johnson KR, et al. Tensional homeostasis and the malignant phenotype. Cancer Cell, 2005, 8: 241–254
Barcus CE, Keely PJ, Eliceiri KW, et al. Stiff collagen matrices increase tumorigenic prolactin signaling in breast cancer cells. J Biol Chem, 2013, 288: 12722–12732
DeCosse JJ, Gossens CL, Kuzma JF, et al. Breast cancer: Induction of differentiation by embryonic tissue. Science, 1973, 181: 1057–1058
Ingber DE. Can cancer be reversed by engineering the tumor microenvironment? Seminars Cancer Biol, 2008, 18: 356–364
Bischof AG, Yüksel D, Mammoto T, et al. Breast cancer normalization induced by embryonic mesenchyme is mediated by extracellular matrix biglycan. Integrative Biol, 2013, 5: 1045–1056
Abu-Tayeh H, Weidenfeld K, Zhilin-Roth A, et al. ‘Normalizing’ the malignant phenotype of luminal breast cancer cells via alpha(v) beta(3)-integrin. Cell Death Dis, 2016, 7: e2491
Levental KR, Yu H, Kass L, et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell, 2009, 139: 891–906
Hu M, Chang H, Zhang H, et al. Mechanical adaptability of the MMP-responsive film improves the functionality of endothelial cell monolayer. Adv Healthcare Mater, 2017, 6: 1601410
Zhang H, Chang H, Wang L, et al. Effect of polyelectrolyte film stiffness on endothelial cells during endothelial-to-mesenchymal transition. Biomacromolecules, 2015, 16: 3584–3593
Audic S, Claverie JM. The significance of digital gene expression profiles. Genome Res, 1997, 7: 986–995
von Mering C, Huynen M, Jaeggi D, et al. STRING: A database of predicted functional associations between proteins. Nucleic Acids Res, 2003, 31: 258–261
Gkretsi V, Stylianopoulos T. Cell adhesion and matrix stiffness: Coordinating cancer cell invasion and metastasis. Front Oncol, 2018, 8: 145
Deng J, Miller SA, Wang HY, et al. β-catenin interacts with and inhibits NF-κB in human colon and breast cancer. Cancer Cell, 2002, 2: 323–334
van Kempen LCL, Coussens LM. MMP9 potentiates pulmonary metastasis formation. Cancer Cell, 2002, 2: 251–252
Jezierska A, Motyl T. Matrix metalloproteinase-2 involvement in breast cancer progression: A mini-review. Med Sci Monitor, 2009, 15: RA32–40
Mehner C, Hockla A, Miller E, et al. Tumor cell-produced matrix metalloproteinase 9 (MMP-9) drives malignant progression and metastasis of basal-like triple negative breast cancer. Oncotarget, 2014, 5: 2736–2749
Roskoski Jr. R. The ErbB/HER family of protein-tyrosine kinases and cancer. Pharmacol Res, 2014, 79: 34–74
Rau A, Lieb WS, Seifert O, et al. Inhibition of tumor cell growth and cancer stem cell expansion by a bispecific antibody targeting EGFR and HER3. Mol Cancer Ther, 2020, 19: 1474–1485
Klaus A, Birchmeier W. Wnt signalling and its impact on development and cancer. Nat Rev Cancer, 2008, 8: 387–398
Lehmann BD, Bauer JA, Chen X, et al. Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest, 2011, 121: 2750–2767
Lee H, Goodarzi H, Tavazoie SF, et al. TMEM2 is a SOX4-regulated gene that mediates metastatic migration and invasion in breast cancer. Cancer Res, 2016, 76: 4994–5005
Bilir B, Kucuk O, Moreno CS. Wnt signaling blockage inhibits cell proliferation and migration, and induces apoptosis in triple-negative breast cancer cells. J Transl Med, 2013, 11: 280
Sun Z, Schwenzer A, Rupp T, et al. Tenascin-C promotes tumor cell migration and metastasis through integrin α9β1-mediated YAP inhibition. Cancer Res, 2018, 78: 950–961
Oskarsson T, Acharyya S, Zhang XHF, et al. Breast cancer cells produce tenascin C as a metastatic niche component to colonize the lungs. Nat Med, 2011, 17: 867–874
Gilbert PM, Havenstrite KL, Magnusson KEG, et al. Substrate elasticity regulates skeletal muscle stem cell self-renewal in culture. Science, 2010, 329: 1078–1081
Yang C, Tibbitt MW, Basta L, et al. Mechanical memory and dosing influence stem cell fate. Nat Mater, 2014, 13: 645–652
Nasrollahi S, Walter C, Loza AJ, et al. Past matrix stiffness primes epithelial cells and regulates their future collective migration through a mechanical memory. Biomaterials, 2017, 146: 146–155
Weaver VM, Petersen OW, Wang F, et al. Reversion of the malignant phenotype of human breast cells in three-dimensional culture and in vivo by integrin blocking antibodies. J Cell Biol, 1997, 137: 231–245
Bussard KM, Smith GH. Human breast cancer cells are redirected to mammary epithelial cells upon interaction with the regenerating mammary gland microenvironment in-vivo. PLoS ONE, 2012, 7: e49221
Kenny PA, Bissell MJ. Tumor reversion: Correction of malignant behavior by microenvironmental cues. Int J Cancer, 2003, 107: 688–695
Yu L, Hou Y, Xie W, et al. Self-strengthening adhesive force promotes cell mechanotransduction. Adv Mater, 2020, 32: 2006986
Yu L, Hou Y, Xie W, et al. Ligand diffusion enables force-independent cell adhesion via activating α5β1 integrin and initiating Rac and RhoA signaling. Adv Mater, 2020, 32: 2002566
Cheng Z, Nai S, Song S, et al. Photoinduced directional domain sliding motion in peptide hydrogels promotes ectodermal differentiation of embryonic stem cells. Sci China Mater, 2020, 63: 467–478
Nakayama KI, Nakayama K. Ubiquitin ligases: cell-cycle control and cancer. Nat Rev Cancer, 2006, 6: 369–381
Mo JS, Park HW, Guan KL. The Hippo signaling pathway in stem cell biology and cancer. EMBO Rep, 2014, 15: 642–656
Kruiswijk F, Labuschagne CF, Vousden KH. p53 in survival, death and metabolic health: A lifeguard with a licence to kill. Nat Rev Mol Cell Biol, 2015, 16: 393–405
Kaur RP, Vasudeva K, Kumar R, et al. Role of p53 gene in breast cancer: focus on mutation spectrum and therapeutic strategies. Curr Pharm Des, 2018, 24: 3566–3575
Lundgren K, Brown M, Pineda S, et al. Effects of cyclin D1 gene amplification and protein expression on time to recurrence in postmenopausal breast cancer patients treated with anastrozole or tamoxifen: A TransATAC study. Breast Cancer Res, 2012, 14: R57
Rudas M, Lehnert M, Huynh A, et al. Cyclin D1 expression in breast cancer patients receiving adjuvant tamoxifen-based therapy. Clin Cancer Res, 2008, 14: 1767–1774
Lundberg A, Lindström LS, Li J, et al. The long-term prognostic and predictive capacity of cyclin D1 gene amplification in 2305 breast tumours. Breast Cancer Res, 2019, 21: 34
Fallah Y, Brundage J, Allegakoen P, et al. MYC-driven pathways in breast cancer subtypes. Biomolecules, 2017, 7: 53
Katoh M, Katoh M. Molecular genetics and targeted therapy of WNT-related human diseases (Review). Int J Mol Med, 2017, 40: 587–606
Valenta T, Hausmann G, Basler K. The many faces and functions of β-catenin. EMBO J, 2012, 31: 2714–2736
Feng J, Sharma S, Rao E, et al. Stiffness heterogeneity-induced double-edged sword behaviors of carcinoma-associated fibroblasts in antitumor therapy. Sci China Mater, 2019, 62: 873–884
Elosegui-Artola A, Andreu I, Beedle AEM, et al. Force triggers YAP nuclear entry by regulating transport across nuclear pores. Cell, 2017, 171: 1397–1410.e14
Shiu JY, Aires L, Lin Z, et al. Nanopillar force measurements reveal actin-cap-mediated YAP mechanotransduction. Nat Cell Biol, 2018, 20: 262–271
Lee JY, Chang JK, Dominguez AA, et al. YAP-independent mechanotransduction drives breast cancer progression. Nat Commun, 2019, 10: 1848
Lee JY, Chaudhuri O. Regulation of breast cancer progression by extracellular matrix mechanics: Insights from 3D culture models. ACS Biomater Sci Eng, 2017, 4: 302–313
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21875210), the National Key Research and Development Program of China (2016YFC1102203), the Natural Key Research and Development Project of Zhejiang Province (2018C03015), Zhejiang Provincial Ten Thousand Talents Program (2018R52001), the Fundamental Research Funds for the Central Universities (2020FZZX003-01-03), and the Higher Education Discipline Innovation Project (111 Project) (B16042).
Author information
Authors and Affiliations
Contributions
Ren KF and Ji J conceived and supervised the project; Hu M and Ren KF designed the experiments; Hu M and Li X carried out the experiments; Hu M wrote the manuscript. Ren KF revised the manuscript. All authors contributed to the general discussion.
Corresponding author
Additional information
Conflict of interest
The authors declare that they have no conflict of interest.
Mi Hu is a PhD candidate in polymer material at Zhejiang University. Her research focuses on the construction of biocompatible polyelectrolyte films and hydrogels.
Xu Li is a PhD candidate in material engineering at Zhejiang University. His research focuses on the interaction between material and cells.
Ke-Feng Ren received his PhD degree from Zhejiang University in 2006. After working as a Postdoc with Prof. C. Picart in CNRS, France, he is now a full professor at Zhejiang University. He has co-authored over 100 peer-reviewed publications. His current research focuses on biomaterials interfaces, medical coating, cardiovascular medical devices.
Jian Ji received his PhD degree from Zhejiang University in 1997. In 2010, he received the Distinguished Young Scholars Award of the National Science Foundation of China. In 2015, he was awarded as Cheung Kong Scholars by the Ministry of Education. He has co-authored over 200 peer-reviewed publications. His research focuses on interfacial phenomena for biomedical implant, tissue engineering and nanomedicine.
Supplementary Information
Rights and permissions
About this article
Cite this article
Hu, M., Li, X., Huang, WP. et al. Dynamically softened substrate regulates malignancy of breast tumor cells. Sci. China Mater. 64, 2580–2592 (2021). https://doi.org/10.1007/s40843-021-1631-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-021-1631-3