Abstract
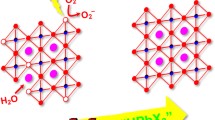
Cesium lead halide perovskite nanocrystals (NCs) have attracted unprecedented attention owing to their compelling properties for optoelectronic applications. Compared with the classical hot-injection method, the room-temperature (RT) synthetic strategy is more facile and tender, but it is hard to obtain stable CsPbI3 NCs and it usually uses polar solvents that sometimes reduce the stability and properties of NCs. Here, we reported a simple approach to synthesize highly efficient and stable CsPbI3 as well as other color-tunable CsPbX3 NCs with high quantum efficiency at room temperature via an anion exchange at the water-oil interface, in which the as-synthesized pristine CsPbBr3 NCs in toluene were treated in aqueous solutions of HX (X=Cl, Br, and I) and protonated oleylamine (OAm) acted as a carrier. The synthesized CsPbI3 NCs had an emission at 680 nm and even showed excellent colloidal stability after being stored for 32 d. The high efficiency and stability of the obtained CsPbX3 NCs were ascribed to the facts that: (i) the polar reagents were almost removed from the surface of NCs; (ii) the defect-related nonradiative recombination was suppressed efficiently by surface passivation.
摘要
全无机CsPbX3钙钛矿纳米晶因其卓越的发光性能而在光电领域引起了前所未有的关注. 相对于传统的热注射法, 室温法是一 种更简单、 温和的合成钙钛矿纳米晶的方法. 然而, 在室温下很难 获得稳定的CsPbI3纳米晶, 并且室温法通常会使用极性溶剂, 这些溶剂有时会降低钙钛矿纳米晶的稳定性和性能. 本文提出了一种 简单的合成方法, 通过在水-油界面处的阴离子交换, 在室温下合成了高效稳定的CsPbI3纳米晶以及其他CsPbX3纳米晶. 在合成过程中, 质子化的油胺充当载体. 测试分析结果表明: 所合成的CsPbI3 纳米晶的发射峰位于680 nm, 且在保存32天后仍然具有极好的发光稳定性. 所合成的CsPbX3纳米晶的高量子效率和稳定性可归因于: (i) 极性试剂几乎从纳米晶溶液中去除; (ii) 通过表面钝化有效地抑制了与缺陷有关的非辐射复合.
Article PDF
Similar content being viewed by others
References
Gao X, Cui Y, Levenson RM, et al. In vivo cancer targeting and imaging with semiconductor quantum dots. Nat Biotechnol, 2004, 22: 969–976
Dai X, Zhang Z, Jin Y, et al. Solution-processed, high-performance light-emitting diodes based on quantum dots. Nature, 2014, 515: 96–99
Sheng X, Chen G, Wang C, et al. Polarized optoelectronics of CsPbX3 (X=Cl, Br, I) perovskite nanoplates with tunable size and thickness. Adv Funct Mater, 2018, 28: 1800283
Zhai W, Lin J, Li Q, et al. Solvothermal synthesis of ultrathin cesium lead halide perovskite nanoplatelets with tunable lateral sizes and their reversible transformation into Cs4PbBr6 nanocrystals. Chem Mater, 2018, 30: 3714–3721
Wu Y, Wei C, Li X, et al. In situ passivation of PbBr64− octahedra toward blue luminescent CsPbBr3 nanoplatelets with near 100% absolute quantum yield. ACS Energy Lett, 2018, 3: 2030–2037
Bekenstein Y, Koscher BA, Eaton SW, et al. Highly luminescent colloidal nanoplates of perovskite cesium lead halide and their oriented assemblies. J Am Chem Soc, 2015, 137: 16008–16011
Wang C, Zhang Y, Wang A, et al. Controlled synthesis of composition tunable formamidinium cesium double cation lead halide perovskite nanowires and nanosheets with improved stability. Chem Mater, 2017, 29: 2157–2166
Swarnkar A, Chulliyil R, Ravi VK, et al. Colloidal CsPbBr3 perovskite nanocrystals: luminescence beyond traditional quantum dots. Angew Chem Int Ed, 2015, 54: 15424–15428
Protesescu L, Yakunin S, Bodnarchuk MI, et al. Nanocrystals of cesium lead halide perovskites (CsPbX3, X=Cl, Br, and I): novel optoelectronic materials showing bright emission with wide color gamut. Nano Lett, 2015, 15: 3692–3696
Yang H, Zhang Y, Pan J, et al. Room-temperature engineering of all-inorganic perovskite nanocrsytals with different dimensionalities. Chem Mater, 2017, 29: 8978–8982
Wei S, Yang Y, Kang X, et al. Room-temperature and gram-scale synthesis of CsPbX3 (X=Cl, Br, I) perovskite nanocrystals with 50%-85% photoluminescence quantum yields. Chem Commun, 2016, 52: 7265–7268
Li X, Wu Y, Zhang S, et al. CsPbX3 quantum dots for lighting and displays: room-temperature synthesis, photoluminescence superiorities, underlying origins and white light-emitting diodes. Adv Funct Mater, 2016, 26: 2435–2445
Akkerman QA, Gandini M, di Stasio F, et al. Strongly emissive perovskite nanocrystal inks for high-voltage solar cells. Nat Energy, 2017, 2: 16194
Sun S, Yuan D, Xu Y, et al. Ligand-mediated synthesis of shape-controlled cesium lead halide perovskite nanocrystals via reprecipitation process at room temperature. ACS Nano, 2016, 10: 3648–3657
Veldhuis SA, Ng YF, Ahmad R, et al. Crown ethers enable room-temperature synthesis of CsPbBr3 quantum dots for light-emitting diodes. ACS Energy Lett, 2018, 3: 526–531
Tong Y, Ehrat F, Vanderlinden W, et al. Dilution-induced formation of hybrid perovskite nanoplatelets. ACS Nano, 2016, 10: 10936–10944
Xie M, Liu H, Chun F, et al. Aqueous phase exfoliating quasi-2D CsPbBr3 nanosheets with ultrahigh intrinsic water stability. Small, 2019, 15: 1901994
Di Stasio F, Christodoulou S, Huo N, et al. Near-unity photoluminescence quantum yield in CsPbBr3 nanocrystal solid-state films via postsynthesis treatment with lead bromide. Chem Mater, 2017, 29: 7663–7667
Creutz SE, Crites EN, De Siena MC, et al. Anion exchange in cesium lead halide perovskite nanocrystals and thin films using trimethylsilyl halide reagents. Chem Mater, 2018, 30: 4887–4891
Imran M, Caligiuri V, Wang M, et al. Benzoyl halides as alternative precursors for the colloidal synthesis of lead-based halide perovskite nanocrystals. J Am Chem Soc, 2018, 140: 2656–2664
Balakrishnan SK, Kamat PV. Ligand assisted transformation of cubic CsPbBr3 nanocrystals into two-dimensional CsPb2Br5 nanosheets. Chem Mater, 2018, 30: 74–78
Udayabhaskararao T, Houben L, Cohen H, et al. A mechanistic study of phase transformation in perovskite nanocrystals driven by ligand passivation. Chem Mater, 2018, 30: 84–93
Liu H, Liu Z, Xu W, et al. Engineering the photoluminescence of CsPbX3 (X=Cl, Br, and I) perovskite nanocrystals across the full visible spectra with the interval of 1 nm. ACS Appl Mater Interfaces, 2019, 11: 14256–14265
Ruan L, Lin J, Shen W, et al. Ligand-mediated synthesis of compositionally related cesium lead halide CsPb2X5 nanowires with improved stability. Nanoscale, 2018, 10: 7658–7665
Ramasamy P, Lim DH, Kim B, et al. All-inorganic cesium lead halide perovskite nanocrystals for photodetector applications. Chem Commun, 2016, 52: 2067–2070
Liu P, Chen W, Wang W, et al. Halide-rich synthesized cesium lead bromide perovskite nanocrystals for light-emitting diodes with improved performance. Chem Mater, 2017, 29: 5168–5173
Li F, Liu Y, Wang H, et al. Postsynthetic surface trap removal of CsPbX3 (X=Cl, Br, or I) quantum dots via a ZnX2/hexane solution toward an enhanced luminescence quantum yield. Chem Mater, 2018, 30: 8546–8554
Tong Y, Bladt E, Aygüler MF, et al. Highly luminescent cesium lead halide perovskite nanocrystals with tunable composition and thickness by ultrasonication. Angew Chem Int Ed, 2016, 55: 13887–13892
Zhong H, Bai Z, Zou B. Tuning the luminescence properties of colloidal I-III-VI semiconductor nanocrystals for optoelectronics and biotechnology applications. J Phys Chem Lett, 2012, 3: 3167–3175
Udayabhaskararao T, Kazes M, Houben L, et al. Nucleation, growth, and structural transformations of perovskite nanocrystals. Chem Mater, 2017, 29: 1302–1308
Zhang M, Li H, Jing Q, et al. Atomic characterization of byproduct nanoparticles on cesium lead halide nanocrystals using highresolution scanning transmission electron microscopy. Crystals, 2018, 8: 2
Gomez L, de Weerd C, Hueso JL, et al. Color-stable water-dispersed cesium lead halide perovskite nanocrystals. Nanoscale, 2017, 9: 631–636
Kang J, Wang LW. High defect tolerance in lead halide perovskite CsPbBr3. J Phys Chem Lett, 2017, 8: 489–493
Liu Y, Xiao H, Goddard William A. I. Two-dimensional halide perovskites: tuning electronic activities of defects. Nano Lett, 2016, 16: 3335–3340
Pan J, Sarmah SP, Murali B, et al. Air-stable surface-passivated perovskite quantum dots for ultra-robust, single- and two-photoninduced amplified spontaneous emission. J Phys Chem Lett, 2015, 6: 5027–5033
Kim Y, Yassitepe E, Voznyy O, et al. Efficient luminescence from perovskite quantum dot solids. ACS Appl Mater Interfaces, 2015, 7: 25007–25013
Koscher BA, Swabeck JK, Bronstein ND, et al. Essentially trap-free CsPbBr3 colloidal nanocrystals by postsynthetic thiocyanate surface treatment. J Am Chem Soc, 2017, 139: 6566–6569
Cai Y, Wang H, Li Y, et al. Trimethylsilyl iodine-mediated synthesis of highly bright red-emitting CsPbI3 perovskite quantum dots with significantly improved stability. Chem Mater, 2019, 31: 881–889
De Roo J, Ibáñez M, Geiregat P, et al. Highly dynamic ligand binding and light absorption coefficient of cesium lead bromide perovskite nanocrystals. ACS Nano, 2016, 10: 2071–2081
Song J, Fang T, Li J, et al. Organic-inorganic hybrid passivation enables perovskite QLEDs with an EQE of 16.48%. Adv Mater, 2018, 30: 1805409
Manser JS, Christians JA, Kamat PV. Intriguing optoelectronic properties of metal halide perovskites. Chem Rev, 2016, 116: 12956–13008
Wang Y, Lü X, Yang W, et al. Pressure-induced phase transformation, reversible amorphization, and anomalous visible light response in organolead bromide perovskite. J Am Chem Soc, 2015, 137: 11144–11149
Zhao Y, Li J, Dong Y, et al. Synthesis of colloidal halide perovskite quantum dots/nanocrystals: progresses and advances. Isr J Chem, 2019, 59: 649–660
Zhang Y, Ding C, Wu G, et al. Air stable PbSe colloidal quantum dot heterojunction solar cells: ligand-dependent exciton dissociation, recombination, photovoltaic property, and stability. J Phys Chem C, 2016, 120: 28509–28518
Kumawat NK, Swarnkar A, Nag A, et al. Ligand engineering to improve the luminance efficiency of CsPbBr3 nanocrystal based light-emitting diodes. J Phys Chem C, 2018, 122: 13767–13773
Zhong Q, Cao M, Hu H, et al. One-pot synthesis of highly stable CsPbBr3@SiO2 core-shell nanoparticles. ACS Nano, 2018, 12: 8579–8587
Gao Y, Peng X. Photogenerated excitons in plain core CdSe nanocrystals with unity radiative decay in single channel: the effects of surface and ligands. J Am Chem Soc, 2015, 137: 4230–4235
Nedelcu G, Protesescu L, Yakunin S, et al. Fast anion-exchange in highly luminescent nanocrystals of cesium lead halide perovskites (CsPbX3, X=Cl, Br, I). Nano Lett, 2015, 15: 5635–5640
Parobek D, Dong Y, Qiao T, et al. Photoinduced anion exchange in cesium lead halide perovskite nanocrystals. J Am Chem Soc, 2017, 139: 4358–4361
Jang DM, Kim DH, Park K, et al. Ultrasound synthesis of lead halide perovskite nanocrystals. J Mater Chem C, 2016, 4: 10625–10629
Long Z, Ren H, Sun J, et al. High-throughput and tunable synthesis of colloidal CsPbX3 perovskite nanocrystals in a heterogeneous system by microwave irradiation. Chem Commun, 2017, 53: 9914–9917
Li H, Brescia R, Povia M, et al. Synthesis of uniform disk-shaped copper telluride nanocrystals and cation exchange to cadmium telluride quantum disks with stable red emission. J Am Chem Soc, 2013, 135: 12270–12278
Hens Z, Martins JC. A solution NMR toolbox for characterizing the surface chemistry of colloidal nanocrystals. Chem Mater, 2013, 25: 1211–1221
Akkerman QA, D’Innocenzo V, Accornero S, et al. Tuning the optical properties of cesium lead halide perovskite nanocrystals by anion exchange reactions. J Am Chem Soc, 2015, 137: 10276–10281
Yassitepe E, Yang Z, Voznyy O, et al. Amine-free synthesis of cesium lead halide perovskite quantum dots for efficient light-emitting diodes. Adv Funct Mater, 2016, 26: 8757–8763
Acknowledgements
This work was supported by the Natural Science Foundation of Fujian Province (2019J05041), the Education Foundation of Fujian Province (JAT170021), and the “Double-First Class” Foundation of Materials and Intelligent Manufacturing Discipline of Xiamen University.
Author information
Authors and Affiliations
Contributions
Author contributions Tang X and Xie RJ conceived and designed the experiments; Fang C and Cai Y performed the experiments; Fang C and Li Y analyzed the data; Zhou TL contributed the materials tools; Fang C and Xie RJ wrote the paper.
Corresponding authors
Ethics declarations
Conflict of interest The authors declare that they have no conflict of interest.
Additional information
Cong Fang is currently a graduate student at the College of Materials, Xiamen University. He obtained his bachelor degree from Fujian University of Technology in 2017. His current research focuses on the synthesis of perovskite nanocrystals.
Rong-Jun Xie received his PhD degree from Shanghai Institute of Ceramics, Chinese Academy of Sciences in 1998. He was Science and Technology Agency (STA) research fellow at the National Institute for Research in Inorganic Materials (Japan, 1998–2000), and Japan Science and Technology Agency (JST) research fellow at the National Institute of Advanced Industrial Science and Technology (Japan, 2001–2002) and Alexander von Humboldt research fellow at Darmstadt University of Technology (Germany, 2002–2003). He joined the National Institute for Materials Science (Japan) as Senior Researcher (2003–2007), Principal Researcher (2007–2014) and Chief Researcher (2014–2017). Since 2018, he has been a full professor in the College of Materials, Xiamen University (China). His research interests include the syntheses and applications of luminescent materials (phosphors, quantum dots (QDs), metal-organic frameworks (MOFs)), solid state lighting and other optoelectronic devices
Supplementary Information for
40843_2020_1379_MOESM1_ESM.pdf
Facial synthesis of highly stable and bright CsPbX3 (X = Cl, Br, I) perovskite nanocrystals via an anion exchange at the water-oil interface
Rights and permissions
About this article
Cite this article
Fang, C., Li, Y., Cai, Y. et al. Facial synthesis of highly stable and bright CsPbX3 (X=Cl, Br, I) perovskite nanocrystals via an anion exchange at the water-oil interface. Sci. China Mater. 64, 158–168 (2021). https://doi.org/10.1007/s40843-020-1379-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-020-1379-4