Abstract
Purpose of Review
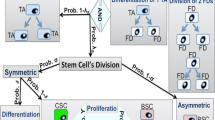
This paper briefly describes recent mathematical models that use stochastic and deterministic approaches to understand stem cell dynamics and how these models are utilized to study the roles of stem cells’ dynamics in cancer and aging.
Recent Findings
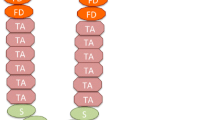
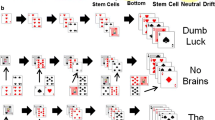
Stochastic compartmental models have been developed to investigate the generalized double-hit mutant production under the influence of different types of stem cell divisions. More specialized examination of the generation, spread, and optimizing structure of 2-hit mutants in the colon crypts has also been conducted. The recent introduction of a hybrid stochastic-deterministic model creates innovative approaches to studying carcinogenesis, while other stochastic models interested in the stem cell renewal process have explored the phenomenon of aging.
Summary
The results of these studies indicate that asymmetric stem cell divisions increase the probability of mutants production and their fixation probability. Moreover, the hybrid stochastic-deterministic model demonstrates how a low rate of dedifferentiation can significantly accelerate carcinogenesis. Finally, a stochastic model for the stem cell renewal process behind aging shows that the fixation probability of an altered stem cell with a longer cell cycle than the rest is higher than other stem cells.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Afify SM, Seno M. Conversion of Stem Cells to Cancer Stem Cells: Undercurrent of Cancer Initiation. Cancers. 2019;11:345.
Ahmed ASI, Sheng MH, Wasnik S, Baylink DJ, Lau KW. Effect of aging on stem cells. World J Exp Med. 2017;7:1–10.
Aragona M, Dekoninck S, Rulands S, Lenglez S, Mascré G, Simons BD, etal. Defining stem cell dynamics and migration during wound healing in mouse skin epidermis. Nat Commun. 2017;8.
Batlle E, Clevers H. Cancer stem cells revisited. Nat Med. 2017;23:1124–34.
Bjerkvig R, Tysnes BB, Aboody KS, Najbauer J, Terzis AJA. The origin of the cancer stem cell: current controversies and new insights. Nat Rev Cancer. 2005;5:899–904.
Dekoninck S, Blanpain C. Stem cell dynamics, migration and plasticity during wound healing. Nat Cell Biol. 2019;21:18–24.
Jiang W, Xu J. Immune modulation by mesenchymal stem cells. Cell Prolif. 2020;53.
Pardal R, Clarke M, Morrison S. Applying the principles of stem-cell biology to cancer. Nat Rev Cancer. 2003;3:895–902.
Sei Y, Feng J, Samsel L, White A, Zhao X, Yun S, etal. Mature enteroendocrine cells contribute to basal and pathological stem cell dynamics in the small intestine. Am J Physiol Gastrointest Liver Physiol.2018;315.
Spangrude GJ, Heimfeld S, Weissman IL. Purification and characterization of mouse hematopoietic stem cells. Science. 1988;241:58–62.
Ayob AZ, Ramasamy TS. Cancer stem cells as key drivers of tumour progression. J Biomed Sci. 2018;25.
Islam F, Gopalan V, Lam AK. Chapter 6 - Cancer Stem Cells: Role in Tumor Progression and Treatment Resistance. In: Dammacco F, Silvestris F, editors. Oncogenomics. Cambridge: Academic Press; 2019. p. 77–87.
Nair N, Calle AS, Zahra MH, Prieto-Vila M, Oo AKK, Hurley L, etal. A cancer stem cell model as the point of origin of cancer-associated fibroblasts in tumor microenvironment. Sci Rep. 2017;7.
Tan BT, Park CY, Ailles LE, Weissman IL. The cancer stem cell hypothesis: a work in progress. Lab Invest. 2006;86:1203–7.
Takebe N, Ivy SP. Controversies in cancer stem cells: targeting embryonic signaling pathways. Clin Cancer Res. 2010;16:3106–12.
Multhoff G, Molls M, Radons J. Chronic Inflammation in Cancer Development. Front Immunol.2012;2. Available from: http://journal.frontiersin.org/article/10.3389/fimmu.2011.00098/abstract.
Grochowski C, Radzikowska E, Maciejewski R. Neural stem cell therapy-Brief review. Clinical Neurology and Neurosurgery. 2018;173:8–14.
Muller P, Lemcke H, David R. Stem Cell Therapy in Heart Diseases - Cell Types, Mechanisms and Improvement Strategies. Cell Physiol Biochem. 2018;48:2607–55.
Yamanaka S. Pluripotent Stem Cell-Based Cell Therapy-Promise and Challenges. Cell Stem Cell. 2020;27:523–31.
Yoon SY. Mesenchymal stem cells for restoration of ovarian function. Clin Exp Reprod Med. 2019;46:1–7.
Zakrzewski W, Dobrzyński M, Szymonowicz M, Rybak Z. Stem cells: past, present, and future. Stem Cell Res Ther. 2019;10.
Shahriyari L, Komarova NL. Symmetric vs. Asymmetric Stem Cell Divisions: An Adaptation against Cancer? PLOS ONE. 2013;8.
Bollas A, Shahriyari L. The role of backward cell migration in two-hit mutants’ production in the stem cell niche. PLOS ONE. 2017;12.
Shahriyari L, Komarova NL. The Role of the Bi-Compartmental Stem Cell Niche in Delaying Cancer. Phys Biol.2015;12.
Shahriyari L, Mahdipour-Shirayeh A. Modeling Dynamics of Mutants in Heterogeneous Stem Cell Niche. Phys Biol.2017;14.
Mahdipour-Shirayeh A, Shahriyari L. Modeling Cell Dynamics in Colon and Intestinal Crypts: The Significance of Central Stem Cells in Tumorigenesis. Bull Math Biol. 2018;80:2273–305.
Alvarado C, Fider AN, Wearing HJ, Komarova NL. Optimizing Homeostatic Cell Renewal in Hierarchical Tissues. PLoS Comput Biol. 2018;14.
Fendrik AJ, Romanelli L, E R. Stochastic Cell Renewal Process and Lengthening of Cell Cycle. Phys Biol. 2019;17.
Jilkine A, Gutenkunst RN. Effect of Dedifferentiation on Time to Mutation Acquisition in Stem Cell-Driven Cancers. PLoS Comput Biol. 2014;10.
Renardy M, Jilkine A, Shahriyari L, Chou CS. Control of cell fraction and population recovery during tissue regeneration in stem cell lineages. J Theor Biol. 2018;445.
Shahriyari L, Komarova NL, Jilkine A. The role of cell location and spatial gradients in the evolutionary dynamics of colon and intestinal crypts. Biol Direct. 2016;11.
• Cheng H, Zheng Z, Cheng T. New paradigms on hematopoietic stem cell differentiation. Protein Cell. 2020;11:34-44. This paper provides a comprehensive review of the recent investigation of the heterogeneity between hematopoietic stem cells and the progenitors and emerging hierarchical models in the mouse system, advancing the current understanding of both hematopoiesis and the hierarchical structure of stem cells.
• Fu NY, Nolan E, Lindeman GJ, Visvader JE. Stem Cells and the Differentiation Hierarchy in Mammary Gland Development. Physiol Rev. 2020;100:489-523. This study reviews recent progress in understanding the cell structure in the mammary gland, which offers a framework for comprehending the cell’s hierarchical collaborative function as well as it’s role in the initiation of cancer.
Gasch C, Ffrench B, O’Leary JJ, Gallagher MF. Catching moving targets: cancer stem cell hierarchies, therapy-resistance and considerations for clinical intervention. Mol Cancer. 2017;16:43.
Yousefi M, Li L, Lengner CJ. Hierarchy and Plasticity in the Intestinal Stem Cell Compartmentt. Trends Cell Biol. 2017;27:753–64.
Knudson AG. Mutation and cancer: statistical study of retinoblastoma. Proc Natl Acad Sci USA. 1971;68:820–3.
Knudson AG. Two genetic hits (more or less) to cancer. Nat Rev Cancer. 2001;1:157–62.
Chaicharoenaudomrung N, Kunhorm P, Noisa P. Three-dimensional cell culture systems as an in vitro platform for cancer and stem cell modeling. World J Stem Cells. 2019;11:1065–83.
Metzcar J, Wang Y, Heiland R, Macklin P. A Review of Cell-Based Computational Modeling in Cancer Biology. JCO Clin Cancer Inform. 2019;3:1–13.
Sfakianakis N, Kolbe N, Hellmann N, Lukácová-Medvid’ová M. A Multiscale Approach to the Migration of Cancer Stem Cells: Math Model Simul Bull Math Biol. 2017;79:209-235.
Stiehl T, Marciniak-Czochra A. Mathematical Modeling of Leukemogenesis and Cancer Stem Cell Dynamics. Math Model Nat Phenom. 2012;7:166–202.
Author information
Authors and Affiliations
Contributions
SG carried out the study and drafted the manuscript; LS designed and supervised the study. All authors gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of Interest
Authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gong, S., Shahriyari, L. A Review of Stochastic and Deterministic Modeling of Stem Cell Dynamics. Curr Stem Cell Rep 9, 1–8 (2023). https://doi.org/10.1007/s40778-023-00225-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40778-023-00225-4