Abstract
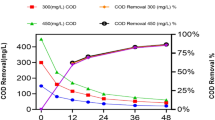
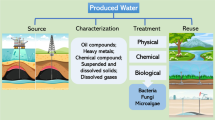
This study focuses on biological removals of petroleum hydrocarbons from an oilfield-produced water generated in Naft Shahr resource by natural attenuation, biostimulation, and bioaugmentation processes. The results of the natural attenuation experiments indicated low population of existing environmental bacterial cells which could not remove total petroleum hydrocarbon (TPH) and initial chemical oxygen demand (COD) at 30 °C and 180 rpm. However, biostimulation by adding nitrogen, sulfur, and phosphorus sources resulted to 19.9% removal of TPH, mainly corresponding to the low chain n-alkanes, and 15.9% removal of COD in 6 days. Degradation of hydrocarbons with longer chain than C20 was limited mainly due to the low solubility of the hydrophobic compounds in the process. Bioaugmentation with the locally-isolated biosurfactant-producing Candida catenulata provided a companionship of the yeast with existing bacterial environmental cells and resulted in increase of the biodegradability of TPH by 16.1% even in the absence of the additional nutrient. A uniform degradation for all hydrocarbon fractions was only observed in the combination of biostimulation with bioaugmentation processes. The study on initial pH yielded the highest removals of 93.39% of TPH and 66.85% of COD in the combined process when initial pH was set at 5.0.








Similar content being viewed by others
References
APHA (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, American Water Works Association, and Water Environment Federation, Washington DC
Bezza FA, Beukes M, Nkhalambayausi Chirwa EM (2015) Application of biosurfactant produced by Ochrobactrum intermedium CN3 for enhancing petroleum sludge bioremediation. Process Biochem 50:1911–1922
Binazadeh M, Karimi IA, Li Z (2009) Fast biodegradation of long chain n-alkanes and crude oil at high concentrations with Rhodococcus sp. Moj-3449. Enzym Microb Technol 45:195–202
Chandran P, Das N (2011) Characterization of sophorolipid biosurfactant produced by yeast species grown on diesel oil. Int J Sci Nature 2(1):63–71
Chemlal R, Tassist A, Drouiche M, Lounici H, Drouiche N, Mameri N (2012) Microbiological aspects study of bioremediation of diesel-contaminated soils by biopile technique. Int Biodeter Biodegr 75:201–206
Chemlal R, Abdi N, Lounici H, Drouiche N, Paussc A, Mameri N (2013) Modeling and qualitative study of diesel biodegradation using biopile process in sandy soil. Int Biodeter Biodegr 78:43–48
Cunningham JA, Rahme H, Hopkins GD, Lebron C, Reinhard M (2001) Enhanced in situ bioremediation of BTEX-contaminated groundwater by combined injection of nitrate and sulfate. Environ Sci Technol 35(8):1663–1670
de Luna JM, Sarubbo L, de Campos-Takaki GM (2009) A new biosurfactant produced by Candida glabrata UCP 1002: characteristics of stability and application in oil recovery. Brazil Arch Biol Technol 52(4):785–793
El Fantroussi S, Agathos SN (2005) Is bioaugmentation a feasible strategy for pollutant removal and site remediation? Curr Opin Biotech 8(3):268–275
Hassanshahian M, Tebyanian H, Cappello S (2012) Isolation and characterization of two crude oil-degrading yeast strains, Yarrowia lipolytica PG-20 and PG-32, from the Persian Gulf. Mar Pollut Bull 64:1386–1391
Huang L, Xie J, Lv B, Shi X, Li G, Liang F, Lian J (2013) Optimization of nutrient component for diesel oil degradation by Acinetobacter beijerinckii ZRS. Mar Pollut Bull 76:325–332
Jamali S, Gharaei M, Abbasi S (2016) Identification of yeast species from uncultivated soils by sequence analysis of the hypervariable D1/D2 domain of LSU-rDNA gene in Kermanshah province, Iran. Mycologia Iranica 3(2):87–98
Jiang Y, Brassington KJ, Prpich G, Paton GI, Semple KT, Pollard SJT, Coulon F (2016) Insights into the biodegradation of weathered hydrocarbons in contaminated soils by bioaugmentation and nutrient stimulation. Chemosphere 161:300–307
Jiménez-Penalver P, Gea T, Sánchez A, Font A (2016) Production of sophorolipids from winterization of oil cake by solid-state fermentation: optimization, monitoring and effect of mixing. Biochem Eng J 115:93–100
Joo HS, Ndegwa PM, Shoda M, Phae C (2008) Bioremediation of oil-contaminated soil using Candida catenulata and food waste. Environ Pollut 156:891–896
Lottermoser K, Schunck WH, Asperger O (1996) Cytochromes P450 of the sophorose lipid-producing yeast Candida apicola: heterogeneity and polymerase chain reaction-mediated cloning of two genes. Yeast 12:565–575
Lu M, Zhang Z, YFu W, Zhu W (2009) Biological treatment of oilfield-produced water: a field pilot study. Int Biodeter Biodegr 63:316–321
Malina G, Zawierucha I (2007) Potential of bioaugmentation and biostimulation for enhancing intrinsic biodegradation in oil hydrocarbon–contaminated soil. Biorem J 11(3):141–147
Manchola L, Dussan J (2014) Lysinibacillus sphaericus and Geobacillus sp biodegradation of petroleum hydrocarbons and biosurfactant production. Remed J 25(1):85–100
Mariaalalraj SK, Pasumarthi R, Achary A, Mutnuri S (2016) Effect of rhamnolipid on biodegradation of hydrocarbons in non-aqueous phase liquid (NAPL). Biorem J 20(3):183–193
Masneuf-Pomarede I, Le Jeune C, Durrens P, Lollier M, Aigle M, Dubourdieu D (2007) Molecular typing of wine yeast strains Saccharomyces bayanus var. uvarum using microsatellite markers. Syst Appl Microbiol 30(1):75–82
Perelo LW (2010) Review: in situ and bioremediation of organic pollutants in aquatic sediments. J Hazard Mater 177:81–89
Płaza GA, Łukasik K, Wypych J, Nałęcz-Jawecki G, Berry C, Brigmon RL (2008) Biodegradation of crude oil and distillation products by biosurfactant-producing bacteria. Pol J Environ Stud 17(1):87–94
Rahman KSM, Rahman TJ, Kourkoutas Y, Petsas I, Marchant R, Banat IM (2003) Enhanced bioremediation of n-alkane in petroleum sludge using bacterial consortium amended with rhamnolipid and micronutrients. Bioresour Technol 90(2):159–168
Ron EZ, Rosenberg E (2002) Biosurfactants and oil bioremediation. Curr Opin Biotechnol 13(3):249–252
Rousk J, Brookes PC, Baath E (2009) Contrasting soil pH effects on fungal and bacterial growth suggest functional redundancy in carbon mineralization. Appl Environ Microb 75(6):1589–1596
Saborimanesh N, Mulligan CN (2015) Effect of sophorolipid biosurfactant on oil biodegradation by the natural oil-degrading bacteria on the weathered biodiesel, diesel and light crude oil. J Bioremed Biodeg 6:314. https://doi.org/10.4172/2155-6199.1000314
Scheller U, Zimmer T, Becher D, Schauer F, Schunck WH (1998) Oxygenation cascade in conversion of n-Alkanes to α,ω-dioic acids catalyzed by cytochrome P450 52A3. J Biol Chem 273(49):32528–32534
Siriverdin T, Dallbauman L (2004) Organic matrix in produced water from the osage-skiatook petroleum environmental research site, Osage County, Oklahoma. Chemosphere 57(6):463–469
Sood N, Lal B (2009) Isolation of a novel yeast strain Candida digboiensis TERI ASN6 capable of degrading petroleum hydrocarbons in acidic conditions. J Environ Manag 90:1728–1736
Tellez GT, Nirmalakhandan N, Gardea-Torresdey LG (2002) Performance evaluation of an activated sludge system for removing petroleum hydrocarbons from oilfield produced water. Adv Environ Res 6(4):455–470
Thomas JM, Yordy P, Jerry R, Amador JA, Alexander M (1986) Rates of dissolution and biodegradation of water-insoluble organic compounds. Appl Environ Microbiol 52(2):290–296
van Bogaert IN, Saerens K, De Muynck C, Develter D, Soetaert W, Vandamme EJ (2007) Microbial production and application of sophorolipids. Appl Microbiol Biotechnol 76(1):23–34
van Bogaert IN, Demey M, Develter D, Soetaert W, Vandamme EJ (2009) Importance of the cytochromeP450monooxygenaseCYP52 family for the sophorolipid-producing yeast Candida bombicola. FEMS Yeast Res 9:87–94
Zahed MA, Aziz HA, Hasnian Isa M, Mohajeri L, Mohajeri S (2010) Optimal conditions for bioremediation of oily seawater. Bioresour Technol 101:9455–9460
Acknowledgements
The authors would like to thank Dr. Saeed Abbasi, Associate Professor at Plant Protection Group of Agricultural Engineering in Razi University, for giving a culture of the isolated C. catenulata yeast. This work was supported by a grant from Research and Development of West Oil and Gas Production Co., Kermanshah, Iran (Grant no. 95-12690).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Habibi, A., Babaei, F. Biological Treatment of Real Oilfield-Produced Water by Bioaugmentation with Sophorolipid-Producing Candida catenulata . Environ. Process. 4, 891–906 (2017). https://doi.org/10.1007/s40710-017-0268-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40710-017-0268-1