Abstract
Background
Many plant food allergens have been implicated in cross-reactivities and may cause repetitive pan-allergenic exposures, which increase the severity of patients’ allergic symptoms. Identification of allergenic protein levels in food allows for more precise dietary advice. This study aimed to determine LTP, profilin, and PR-10 in the 15 most popular fruits in Taiwan.
Methods
cDNA encoding profilin (Sola l 1), nonspecific lipid transfer protein (nsLTP, Sola l 3), and pathogenesis-related protein (PR-10, Sola l 4) were cloned into the pQE30, expressed in Escherichia coli, and purified. Extracts of the most popular fruits, including apple, banana, carrot, celery, dragon fruit, hami melon, jujube, kiwi, mango, melon, orange, papaya, peach, persimmon, and tomato, in Taiwan were prepared in PBS. The allergenic protein levels in these extracts were determined by immuno-dot blotting using rabbit anti-rSola l 1, 3, and 4 polyclonal antibodies.
Results
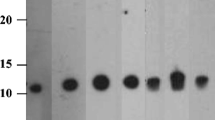
Among all 15 fruit extracts tested, peach and mango contained the highest levels of profilin, nsLTP, and PR-10. Papaya, melon, and tomato contained low but detectable levels of the three pan-allergens.
Conclusion
Our data suggest that peach and mango contain the highest levels of important pan-allergens among the 15 common fruits tested, which agrees with epidemiological data.


Similar content being viewed by others
Abbreviations
- AEC:
-
3-amino-9-ethylcarbazole
- LTP:
-
Lipid transfer proteins
- MW:
-
Molecular weight
- NC:
-
Nitrocellulose membrane
- nsLTP:
-
Nonspecific lipid transfer protein
- PMSF:
-
Phenylmethyl sulfonyl fluoride
- PR:
-
Pathogenesis-related
- SDS-PAGE:
-
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis
References
Aksoy AG, Boran P, Karakoc-Aydiner E, Gokcay G, Tamay ZU, Devecioglu E, et al. Prevalence of allergic disorders and risk factors associated with food allergy in Turkish preschoolers. Allergol Immunopathol. 2021;49(1):11–6. https://doi.org/10.15586/aei.v49i1.23.
Sicherer SH, Sampson HA. Food allergy: a review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J Allergy Clin Immunol. 2018;141(1):41–58. https://doi.org/10.1016/j.jaci.2017.11.003.
Xie Q, Xue W. IgE-Mediated food allergy: Current diagnostic modalities and novel biomarkers with robust potential. Crit Rev Food Sci Nutr. 2022; https://doi.org/10.1080/10408398.2022.2075312.
Hu Y, Chen J, Li H. Comparison of food allergy prevalence among Chinese infants in Chongqing, 2009 versus 1999. Pediatr Int. 2010;52(5):820–4. https://doi.org/10.1111/j.1442-200X.2010.03166.x.
Wu TC, Tsai TC, Huang CF, Chang FY, Lin CC, Huang IF, et al. Prevalence of food allergy in Taiwan: a questionnaire-based survey. Intern Med J. 2012;42(12):1310–5. https://doi.org/10.1111/j.1445-5994.2012.02820.x.
Grimshaw KE, Maskell J, Oliver EM, Morris RC, Foote KD, Mills EN, et al. Diet and food allergy development during infancy: birth cohort study findings using prospective food diary data. J Allergy Clin Immunol. 2014;133(2):511–9. https://doi.org/10.1016/j.jaci.2013.05.035.
Berin MC. Pathogenesis of IgE-mediated food allergy. Clin Exp Allergy. 2015;45(10):1483–96. https://doi.org/10.1111/cea.12598.
Lee MF, Chen YH, Lin HC, Wang HL, Hwang GY, Wu CH. Identification of hevamine and hev B 1 as major latex allergens in Taiwan. Int Arch Allergy Immunol. 2006;139(1):38–44. https://doi.org/10.1159/000089521.
Lee MF, Hwang GY, Chen YH, Lin HC, Wu CH. Molecular cloning of Indian jujube (Zizyphus mauritiana) allergen Ziz m 1 with sequence similarity to plant class III chitinases. Mol Immunol. 2006;43(8):1144–51. https://doi.org/10.1016/j.molimm.2005.07.021.
Sirvent S, Palomares O, Cuesta-Herranz J, Villalba M, Rodriguez R. Analysis of the structural and immunological stability of 2S albumin, nonspecific lipid transfer protein, and profilin allergens from mustard seeds. J Agric Food Chem. 2012;60(23):6011–8. https://doi.org/10.1021/jf300555h.
Breiteneder H, Radauer C. A classification of plant food allergens. J Allergy Clin Immunol. 2004;113(5):821–30. https://doi.org/10.1016/j.jaci.2004.01.779. quiz 31.
Yagami T, Osuna H, Kouno M, Haishima Y, Nakamura A, Ikezawa Z. Significance of carbohydrate epitopes in a latex allergen with beta‑1,3‑glucanase activity. Int Arch Allergy Immunol. 2002;129(1):27–37. https://doi.org/10.1159/000065180.
O’Riordain G, Radauer C, Hoffmann-Sommergruber K, Adhami F, Peterbauer CK, Blanco C, et al. Cloning and molecular characterization of the Hevea brasiliensis allergen Hev b 11, a class I chitinase. Clin Exp Allergy. 2002;32(3):455–62. https://doi.org/10.1046/j.1365-2222.2002.01312.x.
Palacin A, Tordesillas L, Gamboa P, Sanchez-Monge R, Cuesta-Herranz J, Sanz ML, et al. Characterization of peach thaumatin-like proteins and their identification as major peach allergens. Clin Exp Allergy. 2010;40(9):1422–30. https://doi.org/10.1111/j.1365-2222.2010.03578.x.
Smole U, Wagner S, Balazs N, Radauer C, Bublin M, Allmaier G, et al. Bet v 1 and its homologous food allergen Api g 1 stimulate dendritic cells from birch pollen-allergic individuals to induce different Th-cell polarization. Allergy. 2010;65(11):1388–96. https://doi.org/10.1111/j.1398-9995.2010.02407.x.
Gandolfo-Cano M, Gonzalez-Mancebo E, Gonzalez-de-Olano D, Mohedano-Vicente E, Munoz-Garcia E, Bartolome B, et al. Lipid transfer proteins and thaumatins as relevant allergens in melon peel allergy. Ann Allergy Asthma Immunol. 2012;109(3):224–5. https://doi.org/10.1016/j.anai.2012.06.023.
van Esse HP, Reuber TL, van der Does D. Genetic modification to improve disease resistance in crops. New Phytol. 2020;225(1):70–86. https://doi.org/10.1111/nph.15967.
Patrawala S, Ramsey A, Capucilli P, Tuong LA, Vadamalai K, Mustafa SS. Real-world adoption of FDA-approved peanut oral immunotherapy with palforzia. J Allergy Clin Immunol Pract. 2022;10(4):1120–1122.e1. https://doi.org/10.1016/j.jaip.2021.12.022.
Oriel RC, Wang J. Diagnosis and management of food allergy. Immunol Allergy Clin North Am. 2021;41(4):571–85. https://doi.org/10.1016/j.iac.2021.07.012.
Lee MF, Lin SJ, Wang NM, Wu HJ, Chen YH. Plant chitinase III Ziz m 1 stimulates multiple cytokines, most predominantly interleukin-13, from peripheral blood mononuclear cells of latex-fruit allergic patients. Ann Allergy Asthma Immunol. 2012;108(2):113–6. https://doi.org/10.1016/j.anai.2011.10.011.
Lee MF, Chen YH, Lan JL, Tseng CY, Wu CH. Allergenic components of Indian jujube (Zizyphus mauritiana) show IgE cross-reactivity with latex allergen. Int Arch Allergy Immunol. 2004;133(3):211–6. https://doi.org/10.1159/000076826.
Asero R. Tomato allergy: clinical features and usefulness of current routinely available diagnostic methods. J Investig Allergol Clin Immunol. 2013;23(1):37–42.
Pravettoni V, Primavesi L, Farioli L, Brenna OV, Pompei C, Conti A, et al. Tomato allergy: detection of IgE-binding lipid transfer proteins in tomato derivatives and in fresh tomato peel, pulp, and seeds. J Agric Food Chem. 2009;57(22):10749–54. https://doi.org/10.1021/jf9022367.
Wlodarczyk K, Smolinska B, Tomato Allergy MI. The characterization of the selected allergens and antioxidants of tomato (Solanum lycopersicum)—A review. Antioxidants. 2022; https://doi.org/10.3390/antiox11040644.
Lee MF, Tsai JJ, Hwang GY, Lin SJ, Chen YH. Identification of immunoglobulin E (IgE)-binding epitopes and recombinant IgE reactivities of a latex cross-reacting Indian jujube Ziz m 1 allergen. Clin Exp Immunol. 2008;152(3):464–71. https://doi.org/10.1111/j.1365-2249.2008.03661.x.
Sareen R, Shah A. Hypersensitivity manifestations to the fruit mango. Asia Pac Allergy. 2011;1(1):43–9. https://doi.org/10.5415/apallergy.2011.1.1.43.
He FJ, Nowson CA, MacGregor GA. Fruit and vegetable consumption and stroke: meta-analysis of cohort studies. Lancet. 2006;367(9507):320–6. https://doi.org/10.1016/S0140-6736(06)68069-0.
He FJ, Nowson CA, Lucas M, MacGregor GA. Increased consumption of fruit and vegetables is related to a reduced risk of coronary heart disease: meta-analysis of cohort studies. J Hum Hypertens. 2007;21(9):717–28. https://doi.org/10.1038/sj.jhh.1002212.
Reche M, Pascual CY, Vicente J, Caballero T, Martin-Munoz F, Sanchez S, et al. Tomato allergy in children and young adults: cross-reactivity with latex and potato. Allergy. 2001;56(12):1197–201. https://doi.org/10.1034/j.1398-9995.2001.00279.x.
Salcedo G, Sanchez-Monge R, Barber D, Diaz-Perales A. Plant non-specific lipid transfer proteins: an interface between plant defence and human allergy. Biochim Biophys Acta. 2007;1771(6):781–91. https://doi.org/10.1016/j.bbalip.2007.01.001.
Midoro-Horiuti T, Brooks EG, Goldblum RM. Pathogenesis-related proteins of plants as allergens. Ann Allergy Asthma Immunol. 2001;87(4):261–71. https://doi.org/10.1016/S1081-1206(10)62238-7.
Hofmann A, Burks AW. Pollen food syndrome: update on the allergens. Curr Allergy Asthma Rep. 2008;8(5):413–7. https://doi.org/10.1007/s11882-008-0080-0.
Asero R, Mistrello G, Roncarolo D, Amato S, Falagiani P. Analysis of the heat stability of lipid transfer protein from apple. J Allergy Clin Immunol. 2003;112(5):1009–11. https://doi.org/10.1016/s0091-6749(03)02006-2.
Asero R, Mistrello G, Amato S, Roncarolo D, Martinelli A, Zaccarini M. Peach fuzz contains large amounts of lipid transfer protein: is this the cause of the high prevalence of sensitization to LTP in Mediterranean countries? Eur Ann Allergy Clin Immunol. 2006;38(4):118–21.
Fernandez-Rivas M, Cuevas M. Peels of Rosaceae fruits have a higher allergenicity than pulps. Clin Exp Allergy. 1999;29(9):1239–47. https://doi.org/10.1046/j.1365-2222.1999.00628.x.
Lopez-Matas MA, Larramendi CH, Ferrer A, Huertas AJ, Pagan JA, Garcia-Abujeta JL, et al. Identification and quantification of tomato allergens: in vitro characterization of six different varieties. Ann Allergy Asthma Immunol. 2011;106(3):230–8. https://doi.org/10.1016/j.anai.2010.11.022.
Acknowledgements
This research work was supported by grants from Taichung Veterans General Hospital (TCVGH-1057318C and TCVGH-1067317C).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M.F. Lee, Y.H. Chen, Y.-W. Chu, P.C. Hsieh and N.M. Wang declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Lee, M.F., Chen, Y.H., Chu, YW. et al. High pan-allergen content in mango and peach in Taiwan. Allergo J Int 32, 97–103 (2023). https://doi.org/10.1007/s40629-022-00240-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40629-022-00240-7