Abstract
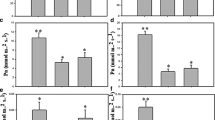
Water deficit presents new challenges for fruit production, among which, obtaining and selecting tolerant genotypes has become relevant. The aim of this work was to characterize novel Prunus rootstocks in regard to tolerance to water deficit as a key trait to cope the challenges for agriculture imposed by the climate change expected in the near future. Here, we report the assessment of physiological parameters and growth responses to water deficit of four interspecific Prunus rootstock hybrids. Two commercially available rootstocks, ROOTPAC®40 and ROOTPAC®20, and two experimental genotypes, AGAF 0301-07 and ADAGAF 0403 were evaluated. Plants were subjected to two irrigation treatments (well-watered and water deficit) during 33 days. During this period, stomatal conductance (gs) and photosynthetic CO2 assimilation (Pn) were measured. At the end of the experiment, biomass accumulation was registered for all the genotypes, while the expression of seven aquaporins belonging to plasma membrane intrinsic proteins subfamily (PIPs) was assessed in ROOTPAC®40 and ROOTPAC®20. ROOTPAC®20 had a decrease in the aquaporin gene expression in roots and this could be associated with its early decline in transpiration. Biomass accumulation was lower in ROOTPAC®20, AGAF 0301-07 and ADAGAF 0403 due to the water deficit. However, ROOTPAC®40 showed a larger whole-plant water use efficiency under water deficit because of a later reduction in relative transpiration under low fractions of transpirable soil water. This response resulted in higher biomass accumulation in ROOTPAC®40 than the other genotypes. We identified ROOTPAC®40 as a tolerant genotype to water deficit, which is majorly explained by a later reduction in relative transpiration under water deficit.




Similar content being viewed by others
References
Bae E-K, Lee H, Lee J-S, Noh E-W (2011) Drought, salt and wounding stress induce the expression of the plasma membrane intrinsic protein 1 gene in poplar (Populus alba × P. tremula var. glandulosa). Gene. https://doi.org/10.1016/j.gene.2011.05.015
Belko N, Zaman-Allah M, Cisse N, Diop NN, Zombre G, Ehlers JD, Vadez V (2012) Lower soil moisture threshold for transpiration decline under water deficit correlates with lower canopy conductance and higher transpiration efficiency in drought-tolerant cowpea. Funct Plant Biol 39:306–322. https://doi.org/10.1071/FP11282
Ben Yahmed J, Ghrab M, Ben Mimoun M (2016a) Eco-physiological evaluation of different scion-rootstock combinations of almond grown in Mediterranean conditions. Fruits 71:185–193. https://doi.org/10.1051/fruits/2016003
Ben Yahmed J, Ghrab M, Moreno MÁ, Pinochet J, Ben Mimoun M (2016b) Performance of ‘Subirana’ flat peach cultivar budded on different Prunus rootstocks in a warm production area in North Africa. Sci Hortic 206:24–32. https://doi.org/10.1016/j.scienta.2016.04.031
Berger J, Palta J, Vadez V (2016) Review: an integrated framework for crop adaptation to dry environments: responses to transient and terminal drought. Plant Sci 253:58–67. https://doi.org/10.1016/j.plantsci.2016.09.007
Bindi M, Bellesi S, Orlandini S, Fibbi L, Moriondo M, Sinclair T (2005) Influence of water deficit stress on leaf area development and transpiration of Sangiovese grapevines grown in pots. Am J Enol Viticult 56:68–72
Brunner I, Herzog C, Dawes M, Arend M, Sperisen C (2015) How tree roots respond to drought. Front Plant Sci 6:1–16. https://doi.org/10.3389/fpls.2015.00547
Camposeo S, Palasciano M, Vivaldi GA, Godini A (2011) Effect of increasing climatic water deficit on some leaf and stomatal parameters of wild and cultivated almonds under Mediterranean conditions. Sci Hort 127:234–241. https://doi.org/10.1016/j.scienta.2010.09.022
Cantero-Navarro E, Romero-Aranda R, Fernández-Muñoz R, Martínez-Andújar C, Pérez-Alfocea F, Albacete A (2016) Improving agronomic water use efficiency in tomato by rootstock-mediated hormonal regulation of leaf biomass. Plant Sci 251:90–100. https://doi.org/10.1016/j.plantsci.2016.03.001
Chang S, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Rep 11:113–116. https://doi.org/10.1007/bf02670468
Chaumont F, Moshelion M, Daniels MJ (2005) Regulation of plant aquaporin activity. Biol Cell 97:749–764. https://doi.org/10.1042/BC20040133
Chaves MM, Flexas J, Pinheiro C (2009) Photosynthesis under drought and salt stress: regulation mechanisms from whole plant to cell. Ann Bot 103:551–560. https://doi.org/10.1093/aob/mcn125
Di Rienzo J, Casanoves F, Balzarini M, Gonzalez L, Tablada M, Robledo C (2011) InfoStat versión 2011 Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. http://www.infostatcomar
Duval H (2015) Use of Prunus genetic diversity for peach rootstocks. Acta Hortic 1084:277–282. https://doi.org/10.17660/ActaHortic.2015.1084.39
FAOSTAT (2017) Crops production in 2017. http://faostat.fao.org/. Accessed 28 Dec 2018
Font i Forcada C, Reig G, Giménez R, Mignard P, Mestre L, Moreno MÁ (2019) Sugars and organic acids profile and antioxidant compounds of nectarine fruits influenced by different rootstocks. Sci Hortic 248:145–153. https://doi.org/10.1016/j.scienta.2018.12.010
Fußeder A, Wartinger A, Hartung W, Schulze ED, Heilmeier H (1992) Cytokinins in the xylem sap of desert-grown almond (Prunus dulcis) trees: daily courses and their possible interactions with abscisic acid and leaf conductance. New Phytol 122:45–52. https://doi.org/10.1111/j.1469-8137.1992.tb00051.x
Gambetta GA et al (2012) The relationship between root hydraulics and scion vigour across Vitis rootstocks: what role do root aquaporins play? J Exp Bot 63:6445–6455. https://doi.org/10.1093/jxb/ers312
Gewin V (2010) Food: an underground revolution. Nature 466:552–553. https://doi.org/10.1038/466552a
Grondin A, Mauleon R, Vadez V, Henry A (2016) Root aquaporins contribute to whole plant water fluxes under drought stress in rice (Oryza sativa L.). Plant, Cell Environ 39:347–365. https://doi.org/10.1111/pce.12616
Ingram J, Bartels D (1996) The molecular basis of dehydration tolerance in plants. Annu Rev Plant Physiol Plant Mol Biol 47:377–403. https://doi.org/10.1146/annurev.arplant.47.1.377
Isaakidis A, Sotiropoulos T, Almaliotis D, Therios I, Stylianidis D (2004) Response to severe water stress of the almond (Prunus amygdalus) ‘Ferragnès’ grafted on eight rootstocks. New Z J Crop Hortic 32:355–362. https://doi.org/10.1080/01140671.2004.9514316
Jiménez S, Dridi J, Gutiérrez D, Moret D, Irigoyen JJ, Moreno MA, Gogorcena Y (2013) Physiological, biochemical and molecular responses in four Prunus rootstocks submitted to drought stress. Tree Physiol 33:1061–1075. https://doi.org/10.1093/treephys/tpt074
Kaldenhoff R, Fischer M (2006) Aquaporins in plants. Acta Physiol 187:169–176. https://doi.org/10.1111/j.1748-1716.2006.01563.x
Lawlor DW (2013) Genetic engineering to improve plant performance under drought: physiological evaluation of achievements, limitations, and possibilities. J Exp Bot 64:83–108. https://doi.org/10.1093/jxb/ers326
Li Y, Ye W, Wang M, Yan X (2009) Climate change and drought: a risk assessment of crop-yield impacts. Clim Res 39:31–46
Li J et al (2015) An aquaporin protein is associated with drought stress tolerance. Biochem Biophys Res Commun 459:208–213. https://doi.org/10.1016/j.bbrc.2015.02.052
Li R et al (2016) Plasma membrane intrinsic proteins SlPIP2;1, SlPIP2;7 and SlPIP2;5 conferring enhanced drought stress tolerance in Tomato. Sci Rep 6:31814. https://doi.org/10.1038/srep31814
Marguerit E, Brendel O, Lebon E, Van Leeuwen C, Ollat N (2012) Rootstock control of scion transpiration and its acclimation to water deficit are controlled by different genes. New Phytol 194:416–429
Maurel C, Verdoucq L, Luu D-T, Santoni V (2008) Plant aquaporins: membrane channels with multiple integrated functions. Annu Rev Plant Biol 59:595–624. https://doi.org/10.1146/annurev.arplant.59.032607.092734
Maurel C, Boursiac Y, Luu D-T, Santoni V, Shahzad Z, Verdoucq L (2015) Aquaporins in plants. Physiol Rev 95:1321–1358. https://doi.org/10.1152/physrev.00008.2015
ODEPA (2018) Frutales superficie y producción. http://icet.odepa.cl/. Accessed 30 Dec 2018
O’Neal ME, Landis DA, Isaacs R (2002) An inexpensive, accurate method for measuring leaf area and defoliation through digital image analysis. J Econ Entomol 95(6):1190–1194
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT–PCR. Nucleic Acids Res 29:e45. https://doi.org/10.1093/nar/29.9.e45
Pou A, Medrano H, Flexas J, Tyerman SD (2013) A putative role for TIP and PIP aquaporins in dynamics of leaf hydraulic and stomatal conductances in grapevine under water stress and re-watering. Plant, Cell Environ 36:828–843. https://doi.org/10.1111/pce.12019
Reddy AR, Chaitanya KV, Vivekanandan M (2004) Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J Plant Physiol 161:1189–1202. https://doi.org/10.1016/j.jplph.2004.01.013
Reddy KS, Sekhar KM, Reddy AR (2017) Genotypic variation in tolerance to drought stress is highly coordinated with hydraulic conductivity–photosynthesis interplay and aquaporin expression in field-grown mulberry (Morus spp.). Tree Physiol 37:926–937. https://doi.org/10.1093/treephys/tpx051
Rieger M, Duemmel MJ (1992) Comparison of drought resistance among Prunus species from divergent habitats. Tree Physiol 11:369–380. https://doi.org/10.1093/treephys/11.4.369
Rieger M, Lo Bianco R, Okie WR (2003) Responses of Prunus ferganensis, Prunus persica and two interspecific hybrids to moderate drought stress. Tree Physiol 23:51–58. https://doi.org/10.1093/treephys/23.1.51
Sakurai-Ishikawa J, Hayashi H, Ahamed A, Fukushi K, Matsumoto T, Kitagawa Y (2011) Transpiration from shoots triggers diurnal changes in root aquaporin expression. Plant, Cell Environ 34:1150–1163. https://doi.org/10.1111/j.1365-3040.2011.02313.x
Shekoofa A, Sinclair TR (2018) Aquaporin activity to improve crop drought tolerance. Cells 7:123. https://doi.org/10.3390/cells7090123
Sinclair T, Ludlow M (1986) Influence of soil water supply on the plant water balance of four tropical grain legumes. Funct Plant Biol 13:329–341. https://doi.org/10.1071/PP9860329
Sinclair TR, Holbrook NM, Zwieniecki MA (2005) Daily transpiration rates of woody species on drying soil. Tree Physiol 25:1469–1472. https://doi.org/10.1093/treephys/25.11.1469
Sofo A, Tuzio AC, Dichio B, Xiloyannis C (2005) Influence of water deficit and rewatering on the components of the ascorbate–glutathione cycle in four interspecific Prunus hybrids. Plant Sci 169:403–412. https://doi.org/10.1016/j.plantsci.2005.04.004
Solari LI, Johnson S, DeJong TM (2006) Relationship of water status to vegetative growth and leaf gas exchange of peach (Prunus persica) trees on different rootstocks. Tree Physiol 26:1333–1341. https://doi.org/10.1093/treephys/26.10.1333
Tomás M, Medrano H, Pou A, Escalona JM, Martorell S, Ribas-CarbÓ M, Flexas J (2012) Water-use efficiency in grapevine cultivars grown under controlled conditions: effects of water stress at the leaf and whole-plant level. Aust J Grape Wine Res 18:164–172. https://doi.org/10.1111/j.1755-0238.2012.00184.x
Tong Z, Gao Z, Wang F, Zhou J, Zhang Z (2009) Selection of reliable reference genes for gene expression studies in peach using real-time PCR. BMC Mol Biol 10:1–13. https://doi.org/10.1186/1471-2199-10-71
Vadez V, Kholova J, Zaman-Allah M, Belko N (2013) Water: the most important ‘molecular’ component of water stress tolerance research. Funct Plant Biol 40:1310–1322. https://doi.org/10.1071/fp13149
Valliyodan B, Nguyen HT (2006) Understanding regulatory networks and engineering for enhanced drought tolerance in plants. Curr Opin Plant Biol 9:189–195. https://doi.org/10.1016/j.pbi.2006.01.019
Vandeleur RK, Mayo G, Shelden MC, Gilliham M, Kaiser BN, Tyerman SD (2009) The role of plasma membrane intrinsic protein aquaporins in water transport through roots: diurnal and drought stress responses reveal different strategies between isohydric and anisohydric cultivars of grapevine. Plant Physiol 149:445–460. https://doi.org/10.1104/pp.108.128645
Vartanian N, Marcotte L, Giraudat J (1994) Drought rhizogenesis in Arabidopsis thaliana (differential responses of hormonal mutants). Plant Physiol 104:761–767. https://doi.org/10.1104/pp.104.2.761
Wang J, Zheng R, Bai S, Gao X, Liu M, Yan W (2015) Mongolian almond (Prunus mongolica Maxim): the morpho-physiological, biochemical and transcriptomic response to drought stress. PLoS ONE 10:1–19
Webster AD (1995) Rootstock and interstock effects on deciduous fruit tree vigour, precocity, and yield productivity. N Z J Crop Hortic 23:373–382. https://doi.org/10.1080/01140671.1995.9513913
Xoconostle-Cazares B, Ramirez-Ortega FA, Flores-Elenes L, Ruiz-Medrano R (2010) Drought tolerance in crop plants Am. J Plant Physiol 5:1–16
Xu Y et al (2014) A banana aquaporin gene, MaPIP1;1, is involved in tolerance to drought and salt stresses. BMC Plant Biol 14:1–14. https://doi.org/10.1186/1471-2229-14-59
Yordanov I, Velikova V, Tsonev T (2000) Plant responses to drought, acclimation, and stress tolerance. Photosynthetica 38:171–186. https://doi.org/10.1023/a:1007201411474
Zaman-Allah M, Jenkinson DM, Vadez V (2011) A conservative pattern of water use, rather than deep or profuse rooting, is critical for the terminal drought tolerance of chickpea. J Exp Bot 62:4239–4252. https://doi.org/10.1093/jxb/err139
Zhu J-K (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53:247–273. https://doi.org/10.1146/annurev.arplant.53.091401.143329
Funding
This work was supported by Grant from project FONDECYT No. 1150853, CONICYT Regional/CEAF/R08I1001, CONICYT scholarship PFCHA/Doctorado Nacional 2018/Folio 21180766 and Programa de Doctorado en Ciencias Silvoagropecuarias y Veterinarias de la Universidad de Chile.
Author information
Authors and Affiliations
Corresponding author
Additional information
In Memoriam: This paper is dedicated to the memory of Nicolás Franck.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
40626_2019_162_MOESM1_ESM.tif
Supplementary material 1 (TIFF 531 kb) Fig. S1 Plant available water of well-watered (WW, black symbols) and water deficit (WD, white symbols) plants throughout the assay of (a) ‘AD0403’ (triangles), (b) ‘AG030107’ (circles), (c) ‘R20’ (diamonds) and (d) ‘R40’ (squares). n=4
Rights and permissions
About this article
Cite this article
Opazo, I., Toro, G., Solis, S. et al. Late reduction on transpiration is an important trait for water deficit tolerance in interspecific Prunus rootstock hybrids. Theor. Exp. Plant Physiol. 31, 493–506 (2019). https://doi.org/10.1007/s40626-019-00162-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40626-019-00162-w