Abstract
Background
Fatty acid-binding protein 4 (FABP4) is secreted from adipocytes and macrophages in adipose tissue and acts as an adipokine. It has recently been reported that FABP4, but not liver-type FABP (L-FABP/FABP1), is also expressed in injured glomerular endothelial cells and infiltrating macrophages in the glomerulus and that urinary FABP4 (U-FABP4) is associated with proteinuria and kidney function impairment in nephrotic patients. However, the link between glomerular FABP4 and U-FABP4 has not been fully addressed in IgA nephropathy (IgAN).
Methods
We investigated the involvement of FABP4 in human and mouse IgAN.
Results
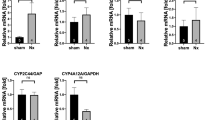
In patients with IgAN (n = 23), the ratio of FABP4-positive area to total area within glomeruli (G-FABP4-Area) and U-FABP4 were positively correlated with proteinuria and were negatively correlated with eGFR. In 4–28-week-old male grouped ddY mice, a spontaneous IgAN-prone mouse model, FABP4 was detected in glomerular endothelial cells and macrophages, and G-FABP4-Area was positively correlated with urinary albumin-to-creatinine ratio (r = 0.957, P < 0.001). Endoplasmic reticulum stress markers were detected in glomeruli of human and mouse IgAN. In human renal glomerular endothelial cells, FABP4 was induced by treatment with vascular endothelial growth factor and was secreted from the cells. Treatment of human renal glomerular endothelial cells or mouse podocytes with palmitate-bound recombinant FABP4 significantly increased gene expression of inflammatory cytokines and endoplasmic reticulum stress markers, and the effects of FABP4 in podocytes were attenuated in the presence of an anti-FABP4 antibody.
Conclusion
FABP4 in the glomerulus contributes to proteinuria in IgAN, and U-FABP4 level is a useful surrogate biomarker for glomerular damage in IgAN.




Similar content being viewed by others
Data availability
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
References
Rodrigues JC, Haas M, Reich HN (2017) IgA nephropathy. Clin J Am Soc Nephrol 12:677–686
Pattrapornpisut P, Avila-Casado C, Reich HN (2021) IgA nephropathy: core curriculum 2021. Am J Kidney Dis 78:429–441
Koyama A, Igarashi M, Kobayashi M (1997) Natural history and risk factors for immunoglobulin A nephropathy in Japan. Research Group on Progressive Renal Diseases. Am J Kidney Dis 29:526–532
Ruan XZ, Varghese Z, Moorhead JF (2009) An update on the lipid nephrotoxicity hypothesis. Nat Rev Nephrol 5:713–721
Izquierdo-Lahuerta A, Martinez-Garcia C, Medina-Gomez G (2016) Lipotoxicity as a trigger factor of renal disease. J Nephrol 29:603–610
Osanami A, Tanaka M, Furuhashi M et al (2022) Increased LDL cholesterol level is associated with deterioration of renal function in males. Clin Kidney J 15:1888–1895
Gregor MF, Hotamisligil GS (2011) Inflammatory mechanisms in obesity. Annu Rev Immunol 29:415–445
Hotamisligil GS (2017) Inflammation, metaflammation and immunometabolic disorders. Nature 542:177–185
Urano F, Wang X, Bertolotti A et al (2000) Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science 287:664–666
Deng J, Lu PD, Zhang Y et al (2004) Translational repression mediates activation of nuclear factor kappa B by phosphorylated translation initiation factor 2. Mol Cell Biol 24:10161–10168
Hu P, Han Z, Couvillon AD et al (2006) Autocrine tumor necrosis factor alpha links endoplasmic reticulum stress to the membrane death receptor pathway through IRE1alpha-mediated NF-kappaB activation and down-regulation of TRAF2 expression. Mol Cell Biol 26:3071–3084
Ozcan U, Yilmaz E, Ozcan L et al (2006) Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science 313:1137–1140
Ishimura S, Furuhashi M, Mita T et al (2014) Reduction of endoplasmic reticulum stress inhibits neointima formation after vascular injury. Sci Rep 4:6943
Koyama M, Furuhashi M, Ishimura S et al (2014) Reduction of endoplasmic reticulum stress by 4-phenylbutyric acid prevents the development of hypoxia-induced pulmonary arterial hypertension. Am J Physiol Heart Circ Physiol 306:H1314-1323
Furuhashi M, Hotamisligil GS (2008) Fatty acid-binding proteins: role in metabolic diseases and potential as drug targets. Nat Rev Drug Discov 7:489–503
Furuhashi M (2019) Fatty acid-binding protein 4 in cardiovascular and metabolic diseases. J Atheroscler Thromb 26:216–232
Kamijo A, Kimura K, Sugaya T et al (2002) Urinary free fatty acids bound to albumin aggravate tubulointerstitial damage. Kidney Int 62:1628–1637
Kamijo A, Kimura K, Sugaya T et al (2004) Urinary fatty acid-binding protein as a new clinical marker of the progression of chronic renal disease. J Lab Clin Med 143:23–30
Kamijo-Ikemori A, Sugaya T, Kimura K (2014) Novel urinary biomarkers in early diabetic kidney disease. Curr Diab Rep 14:513
Furuhashi M, Tuncman G, Gorgun CZ et al (2007) Treatment of diabetes and atherosclerosis by inhibiting fatty-acid-binding protein aP2. Nature 447:959–965
Ishimura S, Furuhashi M, Watanabe Y et al (2013) Circulating levels of fatty acid-binding protein family and metabolic phenotype in the general population. PLoS ONE 8:e81318
Fuseya T, Furuhashi M, Matsumoto M et al (2017) Ectopic fatty acid-binding protein 4 expression in the vascular endothelium is involved in neointima formation after vascular injury. J Am Heart Assoc 6:e006377
Tanaka M, Furuhashi M, Okazaki Y et al (2014) Ectopic expression of fatty acid-binding protein 4 in the glomerulus is associated with proteinuria and renal dysfunction. Nephron Clin Pract 128:345–351
Okazaki Y, Furuhashi M, Tanaka M et al (2014) Urinary excretion of fatty acid-binding protein 4 is associated with albuminuria and renal dysfunction. PLoS ONE 9:e115429
Tanaka M, Furuhashi M, Moniwa N et al (2020) Significance of urinary fatty acid-binding protein 4 level as a possible biomarker for the identification of minimal change disease in patents with nephrotic-range proteinuria. BMC Nephrol 21:459
KDIGO (2012) Chapter 10: Immunoglobulin A nephropathy. Kidney Int Suppl 2:209–217
Yuzawa Y, Yamamoto R, Takahashi K et al (2016) Evidence-based clinical practice guidelines for IgA nephropathy 2014. Clin Exp Nephrol 20:511–535
Matsuo S, Imai E, Horio M et al (2009) Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis 53:982–992
Xu A, Wang Y, Xu JY et al (2006) Adipocyte fatty acid-binding protein is a plasma biomarker closely associated with obesity and metabolic syndrome. Clin Chem 52:405–413
Suzuki H, Suzuki Y (2018) Murine models of human IgA nephropathy. Semin Nephrol 38:513–520
Okazaki K, Suzuki Y, Otsuji M et al (2012) Development of a model of early-onset IgA nephropathy. J Am Soc Nephrol 23:1364–1374
Mundel P, Reiser J, Zuniga Mejia Borja A et al (1997) Rearrangements of the cytoskeleton and cell contacts induce process formation during differentiation of conditionally immortalized mouse podocyte cell lines. Exp Cell Res 236:248–258
Asanuma K, Shirato I, Ishidoh K et al (2002) Selective modulation of the secretion of proteinases and their inhibitors by growth factors in cultured differentiated podocytes. Kidney Int 62:822–831
Furuhashi M, Mita T, Moniwa N et al (2015) Angiotensin II receptor blockers decrease serum concentration of fatty acid-binding protein 4 in patients with hypertension. Hypertens Res 38:252–259
Mita T, Furuhashi M, Hiramitsu S et al (2015) FABP4 is secreted from adipocytes by adenyl cyclase-PKA- and guanylyl cyclase-PKG-dependent lipolytic mechanisms. Obesity (Silver Spring) 23:359–367
Kanda Y (2013) Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant 48:452–458
Elmasri H, Karaaslan C, Teper Y et al (2009) Fatty acid binding protein 4 is a target of VEGF and a regulator of cell proliferation in endothelial cells. FASEB J 23:3865–3873
Elmasri H, Ghelfi E, Yu CW et al (2012) Endothelial cell-fatty acid binding protein 4 promotes angiogenesis: role of stem cell factor/c-kit pathway. Angiogenesis 15:457–468
Furuhashi M, Fuseya T, Murata M et al (2016) Local production of fatty acid-binding protein 4 in epicardial/perivascular fat and macrophages is linked to coronary atherosclerosis. Arterioscler Thromb Vasc Biol 36:825–834
Yao F, Li Z, Ehara T et al (2015) Fatty Acid-Binding Protein 4 mediates apoptosis via endoplasmic reticulum stress in mesangial cells of diabetic nephropathy. Mol Cell Endocrinol 411:232–242
Prentice KJ, Saksi J, Robertson LT et al (2021) A hormone complex of FABP4 and nucleoside kinases regulates islet function. Nature 600:720–726
D’Amico G (2004) Natural history of idiopathic IgA nephropathy and factors predictive of disease outcome. Semin Nephrol 24:179–196
Hotta O, Miyazaki M, Furuta T et al (2001) Tonsillectomy and steroid pulse therapy significantly impact on clinical remission in patients with IgA nephropathy. Am J Kidney Dis 38:736–743
Pozzi C, Andrulli S, Del Vecchio L et al (2004) Corticosteroid effectiveness in IgA nephropathy: long-term results of a randomized, controlled trial. J Am Soc Nephrol 15:157–163
Kawamura T, Yoshimura M, Miyazaki Y et al (2014) A multicenter randomized controlled trial of tonsillectomy combined with steroid pulse therapy in patients with immunoglobulin A nephropathy. Nephrol Dial Transplant 29:1546–1553
Hirano K, Matsuzaki K, Yasuda T et al (2019) Association between tonsillectomy and outcomes in patients with immunoglobulin A nephropathy. JAMA Netw Open 2:e194772
Medjeral-Thomas NR, O’Shaughnessy MM (2020) Complement in IgA nephropathy: the role of complement in the pathogenesis, diagnosis, and future management of IgA nephropathy. Adv Chronic Kidney Dis 27:111–119
Zhao N, Hou P, Lv J et al (2012) The level of galactose-deficient IgA1 in the sera of patients with IgA nephropathy is associated with disease progression. Kidney Int 82:790–796
Suzuki H, Allegri L, Suzuki Y et al (2016) Galactose-deficient IgA1 as a candidate urinary polypeptide marker of IgA nephropathy? Dis Markers 2016:7806438
Irabu H, Shimizu M, Kaneko S et al (2021) Apoptosis inhibitor of macrophage as a biomarker for disease activity in Japanese children with IgA nephropathy and Henoch-Schonlein purpura nephritis. Pediatr Res 89:667–672
Takahata A, Arai S, Hiramoto E et al (2020) Crucial role of AIM/CD5L in the development of glomerular inflammation in IgA nephropathy. J Am Soc Nephrol 31:2013–2024
Tomino Y (2016) Diagnosis and treatment of patients with IgA nephropathy in Japan. Kidney Res Clin Pract 35:197–203
Obajdin J, Cotter M, Snelling S et al (2018) Fatty-acid binding protein 4 (FABP4) as a potential preclinical biomarker of drug-induced kidney injury. Toxicol Sci 166:441–450
Shrestha S, Sunaga H, Hanaoka H et al (2018) Circulating FABP4 is eliminated by the kidney via glomerular filtration followed by megalin-mediated reabsorption. Sci Rep 8:16451
Funding
M.T and M.F were supported by JSPS KAKENHI (19K08708, 20K08913, 22K08313).
Author information
Authors and Affiliations
Contributions
MT, and MF designed the study, performed data analyses and wrote the paper. NM, CN, TK, TM, and HT performed data collection. MM and AK performed experiments. YO, KA, and YS supervised the analyses. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The human study strictly conformed to the principles in the Declaration of Helsinki and was approved by the Ethical Committee of Sapporo Medical University (number: 20-2-58) and the Ethical Committee of Teine Keijinkai Hospital (number: 2017-031). All experimental protocols were approved by the Ethics Review Committee for Animal Experimentation of Juntendo University Faculty of Medicine and the Animal Care and Experiment Committee of Sapporo Medical University.
Informed consent
Written informed consent was received from all of the study subjects.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tanaka, M., Moniwa, N., Nogi, C. et al. Glomerular expression and urinary excretion of fatty acid-binding protein 4 in IgA nephropathy. J Nephrol 36, 385–395 (2023). https://doi.org/10.1007/s40620-022-01551-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-022-01551-2