Abstract
Background
The underlying mechanisms of nephrotic syndrome (NS) are still under debate and the need for more effective and less toxic treatment is challenging. We aimed to evaluate the efficacy of montelukast, a leukotriene receptor antagonist as an add-on therapy, and to explore the leukotriene (LT) biology in steroid-dependent nephrotic syndrome (SDNS).
Methods
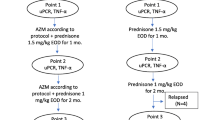
A randomized controlled trial was conducted including 32 patients with SDNS who were randomly assigned to receive standard steroid treatment only [low-dose steroid (LDS) group] or standard steroid therapy plus montelukast (Montelukast group). Urine protein/creatinine ratio, serum albumin, creatinine, cholesterol, and plasma LTs (LTB4/C4/D4/E4) were evaluated in all patients before and after treatment.
Results
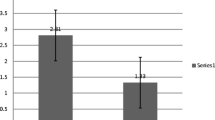
After treatment, both groups showed a significant decrease of LTB4 and LTC4/D4/E4. Further, the Montelukast group showed a significant decrease in serum creatinine and a significant increase in diastolic blood pressure and protein/creatinine ratio. It also showed a marked decrease in plasma LTC4/D4/E4 compared to the LDS group, although not statistically significant.
Conclusions
Our findings highlight the effect of montelukast on renal function, but suggest that the clinical and laboratory efficacy of the addition of montelukast to steroids in maintenance treatment of SDNS is debatable.


Similar content being viewed by others
References
Wang X, Xu H (2013) New insights into treatment of nephrotic syndrome in children. Contrib Nephrol 181:119–130
Bakr A, Eid R, Sarhan A, Hammad A, El-Refaey AM, El-Mougy A, Zedan MM, Moustafa F, Abdelrahman A (2014) Pathological profile of biopsied Egyptian children with primary nephrotic syndrome: 15-year single center experience. J Nephrol 27(4):419–423
Swierczewska M, Ostalska-Nowicka D, Kempisty B, Nowicki M, Zabel M (2013) Molecular basis of mechanisms of steroid resistance in children with nephrotic syndrome. Acta Biochim Pol 60(3):339–344
Schönenberger E, Ehrich JH, Haller H, Schiffer M (2011) Thepodocyte as a direct target of immunosuppressive agents. Nephrol Dial Transplant 26(1):18–24
Ruggenenti P, Ruggiero B, Cravedi P, Vivarelli M, Massella L, Marasà M, Chianca A, Rubis N, Ene-Iordache B, Rudnicki M, Pollastro RM, Capasso G, Pisani A, Pennesi M, Emma F, Remuzzi G (2014) Rituximab in nephrotic syndrome of steroid-dependent or frequently relapsing minimal change disease or segmental glomerulosclerosis (NEMO) Study Group. Rituximab in steroid-dependent or frequently relapsing idiopathic nephrotic syndrome. J Am Soc Nephrol 25(4):850–863
Uwaezuoke SN (2015) Steroid-sensitive nephrotic syndrome in children: triggers of relapse and evolving hypotheses on pathogenesis. Ital J Pediatr 41:19
Ravani P, Rossi R, Bonanni A, Quinn RR, Sica F, Bodria M, Pasini A, Montini G, Edefonti A, Belingheri M, De Giovanni D, Barbano G, Degl’Innocenti L, Scolari F, Murer L, Reiser J, Fornoni A, Ghiggeri GM (2015) Rituximab in children with steroid-dependent nephrotic syndrome: a multicenter, open-label, noninferiority, randomized controlled trial. J Am Soc Nephrol. doi:10.1681/ASN.2014080799
Bakr A, Hawas S, Slem S, Moniem AA, Ghatab T, Tawfik M (2004) 5-Lipoxygenase and leukotriene A4 hydrolase expression in primary nephrotic syndrome. Pediatr Nephrol 19(4):396–399
Mir S, Huseyinov A, Kantar M, Kabasakal C, Cura A, Çoker I (1998) The role of leukotrienes in the pathogenesis of steroid-sensitive nephrotic syndrome. Turk J Med Sci 28:625–629
Rinaldo-Matthis A, Haeggström JZ (2010) Structures and mechanisms of enzymes in the leukotriene cascade. Biochimie 92(6):676–681
Menegatti E, Roccatello D, Fadden K, Piccoli G, De Rosa G, Sena LM, Rifai A (1999) Gene expression of 5-lipoxygenase and LTA4 hydrolase in renal tissue of nephrotic syndrome patients. Clin Exp Immunol 116(2):347–353
KDIGO (2012) Clinical practice guideline for glomerulonephritis. Kidney Int Suppl 2:163–171. Available at http://kdigo.org/home/glomerulonephritis-gn/. Accessed 19 Mar 2016
Report of the Second Task Force on Blood Pressure Control in Children (1987) Task force on blood pressure control in children. National Heart, Lung, and Blood Institute, Bethesda, Maryland. Pediatrics 79(1):1–25
Farid FA, Mohammed AA, Afifi HM, Beltagi RS (2011) Tissue factor pathway inhibitor in paediatric patients with nephrotic syndrome. SAJCH. 5(4):107–111
Lehmann E (1998) Nonparametrics: statistical methods based on ranks, revised. Pearson education, Upper Saddle River, pp 76–81
Imig JD, Ryan MJ (2013) Immune and inflammatory role in renal disease. Compr Physiol 3(2):957–976
Mathieson PW (2003) Immune dysregulation in minimal change nephropathy. Nephrol Dial Transplant 18:26–29
Souto MF, Teixeira AL, Russo RC, Penido MG, Silveira KD, Teixeira MM, Simões E Silva AC (2008) Immune mediators in idiopathic nephrotic syndrome: evidence for a relation between interleukin 8 and proteinuria. Pediatr Res 64(6):637–642
Golan DE, Tashjian AH, Armstrong EJ (2011) Principles of pharmacology: the pathophysiologic basis of drug therapy, 3rd edn. Lippincott Williams & Wilkins, Philadelphia, p 753
Otunctemur A, Ozbek E, Cekmen M, Cakir SS, Dursun M, Polat EC, Somay A, Ozbay N (2013) Protective effect of montelukast which is cysteinyl-leukotriene receptor antagonist on gentamicin-induced nephrotoxicity and oxidative damage in rat kidney. Ren Fail 35(3):403–410
Park SJ, Saleem MA, Nam JA, Ha TS, Shin JI (2015) Effects of interleukin-13 and montelukast on the expression of zonula occludens-1 in human podocytes. Yonsei Med J 56(2):426–432
Jackson SE, Holloway JW, Warner JA, Sampson AP (2012) Interleukin-13, but not indomethacin, increases cysteinyl-leukotriene synthesis in human lung macrophages. J Allergy (Cairo) 2012:348741
Petric R, Ford-Hutchinson A (1995) Inhibition of leukotriene biosynthesis improves renal function in experimental glomerulonephritis. J Lipid Mediat Cell Signal 11(3):231–240
Al-Saedi HF, Al-Zubaidy AA, Khattab YI (2014) The possible effects of montelukast against doxorubicin-induced nephrotoxicity in rabbits. Int J Adv Res 2(11):723–729
Suddek GM (2013) Montelukast ameliorates kidney function and urinary bladder sensitivity in experimentally induced renal dysfunction in rats. Fundam Clin Pharmacol 27(2):186–191
Kose E, Beytur A, Dogan Z, Ekincioglu Z, Vardi N, Cinar K, Turkoz Y, Soysal H, Ekinci N (2012) The effects of montelukast against amikacin-induced acute renal damage. Eur Rev Med Pharmacol Sci 16(4):503–511
Helmy MM, El-Gowelli HM (2012) Montelukast abrogates rhabdomyolysis-induced acute renal failure via rectifying detrimental changes in antioxidant profile and systemic cytokines and apoptotic factors production. Eur J Pharmacol 683(1–3):294–300
Calapai G, Casciaro M, Miroddi M, Calapai F, Navarra M, Gangemi S (2014) Montelukast-induced adverse drug reactions: a review of case reports in the literature. Pharmacology 94(1–2):60–70
Kumagai T, Hori Y, Kishida Y, Yakumaru K, Takahashi T, Itou T (2003) Acute renal failure and nephrotic syndrome associated with zafirlukast therapy. Nephrol Dial Transplant 18(10):2202–2203
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was not funded.
Conflict of interest
The authors declared that they have no conflict of interests.
Ethical statement
All procedures performed in the study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Zedan, M.M., El-Refaey, A., Zaghloul, H. et al. Montelukast as an add-on treatment in steroid dependant nephrotic syndrome, randomised-controlled trial. J Nephrol 29, 585–592 (2016). https://doi.org/10.1007/s40620-016-0297-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-016-0297-2