Abstract
Background
Poorly differentiated thyroid cancer (PDTC) and anaplastic thyroid cancer (ATC) can be developed from differentiated thyroid cancer, and this dedifferentiated transformation leads to poor prognosis and high mortality. The role of Nrf2 in the dedifferentiation of differentiated thyroid cancer (DTC) induced by KRAS remains unclear.
Methods and materials
In this study, two DTC cell lines, BCPAP and WRO, were used to evaluate the function of Nrf2 in the dedifferentiation caused by wild-type KRAS (KRAS-WT) and G12V point mutation KRAS (KRAS-G12V).
Results
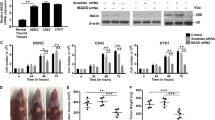
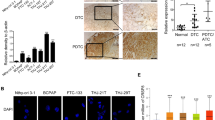
The overexpression of KRAS-WT and KRAS-G12V increased the proliferative and invasive ability of BCPAP and WRO cells. Aggressive morphology was observed in KRAS-WT and KRAS-G12V overexpressed WRO cells. These results suggested that overexpression of KRAS-WT or KRAS-G12V may induce dedifferentiation in DTC cells. The expression of Nrf2 was increased by KRAS-WT and KRAS-G12V in DTC cells. In addition, compared with normal thyroid tissues, the expression of Nrf2 protein was considerably higher in thyroid cancer tissues on immunohistochemistry (IHC) staining, and the increased expression of Nrf2 indicated a poor prognosis of thyroid cancer. These results indicated that Nrf2 is the KRAS downstream molecule in thyroid cancer. Functional studies showed that the Nrf2 inhibitor Brusatol counteracted the proliferative and invasive abilities induced by KRAS-WT and KRAS-G12V in BCPAP and WRO cells. In addition, the xenograft assay further confirmed that Brusatol inhibits tumor growth induced by KRAS-WT and KRAS-G12V.
Conclusion
Collectively, this study suggests that Nrf2 could be a promising therapeutic target in KRAS-mediated dedifferentiation of thyroid cancer.







Similar content being viewed by others
Data availability
Not applicable.
Abbreviations
- ATC:
-
Anaplastic thyroid cancer
- DTC:
-
Differentiated thyroid cancer
- FTC:
-
Follicular thyroid cancer
- IHC:
-
Immunohistochemistry
- IRS:
-
Immunoreactive score
- KRAS-G12V:
-
G12V point mutation KRAS
- KRAS-WT:
-
Wild-type KRAS
- PDTC:
-
Poorly differentiated thyroid cancer
- PTC:
-
Papillary thyroid cancer
References
Cabanillas ME, McFadden DG, Durante C (2016) Thyroid cancer. Lancet 388(10061):2783–2795
Volante M, Lam AK, Papotti M, Tallini G (2021) Molecular pathology of poorly differentiated and anaplastic thyroid cancer: what do pathologists need to know? Endocr Pathol 32(1):63–76
Radkay LA, Chiosea SI, Seethala RR, Hodak SP, LeBeau SO, Yip L, McCoy KL, Carty SE, Schoedel KE, Nikiforova MN, Nikiforov YE, Ohori NP (2014) Thyroid nodules with KRAS mutations are different from nodules with NRAS and HRAS mutations with regard to cytopathologic and histopathologic outcome characteristics. Cancer Cytopathol 122(12):873–882
Cabanillas ME, Dadu R, Iyer P, Wanland KB, Busaidy NL, Ying A, Gule-Monroe M, Wang JR, Zafereo M, Hofmann MC (2020) Acquired secondary RAS mutation in BRAF(V600E)-mutated thyroid cancer patients treated with BRAF inhibitors. Thyroid 30(9):1288–1296
Owen DH, Konda B, Sipos J, Liu T, Webb A, Ringel MD, Timmers CD, Shah MH (2019) KRAS G12V mutation in acquired resistance to combined BRAF and MEK inhibition in papillary thyroid cancer. J Natl Compr Canc Netw 17(5):409–413
Tao S, Wang S, Moghaddam SJ, Ooi A, Chapman E, Wong PK, Zhang DD (2014) Oncogenic KRAS confers chemoresistance by upregulating NRF2. Cancer Res 74(24):7430–7441
Gong Z, Xue L, Wei M, Liu Z, Vlantis AC, van Hasselt CA, Chan JYK, Li D, Zeng X, Tong MCF, Chen GG (2021) The knockdown of Nrf2 suppressed tumor growth and increased the sensitivity to lenvatinib in anaplastic thyroid cancer. Oxid Med Cell Longev 2021:3900330
Mouron S, Manso L, Caleiras E, Rodriguez-Peralto JL, Rueda OM, Caldas C, Colomer R, Quintela-Fandino M, Bueno MJ (2021) FGFR1 amplification or overexpression and hormonal resistance in luminal breast cancer: rationale for a triple blockade of ER, CDK4/6, and FGFR1. Breast Cancer Res 23(1):21
Kaemmerer D, Peter L, Lupp A, Schulz S, Sänger J, Baum RP, Prasad V, Hommann M (2012) Comparing of IRS and Her2 as immunohistochemical scoring schemes in gastroenteropancreatic neuroendocrine tumors. Int J Clin Exp Pathol 5(3):187–194
Cai SJ, Liu Y, Han S, Yang C (2019) Brusatol, an NRF2 inhibitor for future cancer therapeutic. Cell Biosci 9:45
Pozdeyev N, Rose MM, Bowles DW, Schweppe RE (2020) Molecular therapeutics for anaplastic thyroid cancer. Semin Cancer Biol 61:23–29
Xing M (2013) Molecular pathogenesis and mechanisms of thyroid cancer. Nat Rev Cancer 13(3):184–199
Nikiforova MN, Wald AI, Roy S, Durso MB, Nikiforov YE (2013) Targeted next-generation sequencing panel (ThyroSeq) for detection of mutations in thyroid cancer. J Clin Endocrinol Metab 98(11):E1852–E1860
Han Luo XX, Kim GD, Liu Y, Xue Z, Zhang L, Shu Y, Yang T, Chen Y, Zhang S, Chen H, Zhang W, Li R, Tang H, Dong B, Xianghui F, Cheng W, Zhang W, Yang L, Peng Y, Dai L, Hongbo H, Jiang Y, Gong C, Yiguo H, Zhu J, Li Z, Caulin C, Wei T, Park J, Heng X (2021) Characterizing dedifferentiation of thyroid cancer by integrated analysis. Sci Adv 7:eabf3657
Saiselet M, Floor S, Tarabichi M, Dom G, Hebrant A, van Staveren WC, Maenhaut C (2012) Thyroid cancer cell lines: an overview. Front Endocrinol (Lausanne) 3:133
Schweppe RE, Klopper JP, Korch C, Pugazhenthi U, Benezra M, Knauf JA, Fagin JA, Marlow LA, Copland JA, Smallridge RC, Haugen BR (2008) Deoxyribonucleic acid profiling analysis of 40 human thyroid cancer cell lines reveals cross-contamination resulting in cell line redundancy and misidentification. J Clin Endocrinol Metab 93(11):4331–4341
Landa I, Pozdeyev N, Korch C, Marlow LA, Smallridge RC, Copland JA, Henderson YC, Lai SY, Clayman GL, Onoda N, Tan AC, Garcia-Rendueles MER, Knauf JA, Haugen BR, Fagin JA, Schweppe RE (2019) Comprehensive genetic characterization of human thyroid cancer cell lines: a validated panel for preclinical studies. Clin Cancer Res 25(10):3141–3151
Pan LN, Ma YF, Li Z, Hu JA, Xu ZH (2021) KRAS G12V mutation upregulates PD-L1 expression via TGF-beta/EMT signaling pathway in human non-small-cell lung cancer. Cell Biol Int 45(4):795–803
Monticone M, Biollo E, Maffei M, Donadini A, Romeo F, Storlazzi CT, Giaretti W, Castagnola P (2008) Gene expression deregulation by KRAS G12D and G12V in a BRAF V600E context. Mol Cancer 7:92
McCormick F (2015) KRAS as a therapeutic target. Clin Cancer Res 21(8):1797–1801
Liu P, Wang Y, Li X (2019) Targeting the untargetable KRAS in cancer therapy. Acta Pharm Sin B 9(5):871–879
Uprety D, Adjei AA (2020) KRAS: from undruggable to a druggable cancer target. Cancer Treat Rev 89:102070
Canon J, Rex K, Saiki AY, Mohr C, Cooke K, Bagal D, Gaida K, Holt T, Knutson CG, Koppada N, Lanman BA, Werner J, Rapaport AS, San Miguel T, Ortiz R, Osgood T, Sun JR, Zhu X, McCarter JD, Volak LP, Houk BE, Fakih MG, O’Neil BH, Price TJ, Falchook GS, Desai J, Kuo J, Govindan R, Hong DS, Ouyang W, Henary H, Arvedson T, Cee VJ, Lipford JR (2019) The clinical KRAS(G12C) inhibitor AMG 510 drives anti-tumour immunity. Nature 575(7781):217–223
Robledinos-Antón N, Fernández-Ginés R, Manda G, Cuadrado A (2019) Activators and inhibitors of NRF2: a review of their potential for clinical development. Oxid Med Cell Longev 2019:1–20
Acknowledgements
We thank Dr. Jia Peng and Ms. SY Chun for their technical assistance.
Funding
This study was supported by grants from the National Natural Science Foundation of China (n. 81972493) and the Research Grants Council of the Hong Kong Special Administrative Region CUHK 14108921.
Author information
Authors and Affiliations
Contributions
Study design and concept: ZG, MT, GC. Data acquisition: ZG, LX, AV, CV, JC. Data analysis and interpretation: ZG, JF, RW. Collection of clinical data and sample disposal: ZG, YY, DL, XZ, Manuscript preparation: ZG, GC. Manuscript review: GC, MT. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that there is no conflict of interest.
Ethical approval and consent to participate
Human studies were approved by the Joint CUHK-NTEC Clinical Research Ethics Committee, which is accordance with the Declaration of Helsinki.
Animal ethic approval
All procedures of mouse experiments were approved by the Animal Ethics Committee of the Chinese University of Hong Kong.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gong, Z., Xue, L., Vlantis, A.C. et al. Brusatol attenuated proliferation and invasion induced by KRAS in differentiated thyroid cancer through inhibiting Nrf2. J Endocrinol Invest 47, 1271–1280 (2024). https://doi.org/10.1007/s40618-023-02248-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-023-02248-4