Abstract
Purpose
To investigate the relationship between fibrinogen-like protein 1 (FGL-1) concentrations and various metabolic characteristics in patients with polycystic ovary syndrome (PCOS) and explore whether FGL-1 could be a predictive biomarker for PCOS.
Methods
This case–control study included 136 patients with PCOS and 34 normal controls recruited in the Department of Endocrinology and Metabolism, Shanghai Tenth People’s Hospital between May 2017 and June 2021. Anthropometric characteristics, metabolic parameters, and reproductive hormones were collected. Serum FGL-1 measurement was conducted using enzyme-linked immunosorbent assay (ELISA) kits.
Results
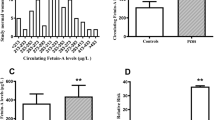
Serum FGL-1 concentrations were higher in patients with PCOS than in control subjects in body mass index (BMI) subgroups, insulin resistance (IR) subgroups, and hepatic function subgroups, respectively. Serum FGL-1 concentrations were significantly associated with BMI, glycosylated hemoglobin A1c (HbA1c), fasting plasma glucose (FPG), homeostasis model assessment of insulin resistance (HOMA-IR), alanine aminotransferase (ALT), aspartate aminotransferase (AST), high-density lipoprotein cholesterol (HDL-c), and serum uric acid (SUA) in all individuals. The receiver operating characteristic (ROC) curve analysis revealed that the best cutoff value for FGL-1 levels to predict PCOS was 21.02 ng/ml with a sensitivity of 74.3% and a specificity of 70.6%. Both univariate and multiple logistic regressions indicated that the odds ratio (OR) for PCOS significantly increased in the subjects with high levels of FGL-1.
Conclusion
In our study, FGL-1 was associated with serum aminotransferase and various metabolic indexes. Moreover, the high risk of PCOS was independently associated with the increased FGL-1 levels, which suggested that FGL-1 could be a predictive biomarker for PCOS.



Similar content being viewed by others
Data availability statement
All data generated or analyzed during this study are available from the corresponding authors on reasonable request.
References
Escobar-Morreale HF (2018) Polycystic ovary syndrome: Definition, aetiology, diagnosis and treatment. Nat Rev Endocrinol 14:270–284. https://doi.org/10.1038/nrendo.2018.24
Fauser BCJM, Tarlatzis F et al (2004) Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Hum Reprod 19:41–47. https://doi.org/10.1093/humrep/deh098
Dumesic DA, Oberfield SE, Stener-Victorin E et al (2015) Scientific statement on the diagnostic criteria, epidemiology, pathophysiology, and molecular genetics of polycystic ovary syndrome. Endocr Rev 36:487–525. https://doi.org/10.1210/er.2015-1018
Moran LJ, Norman RJ, Teede HJ (2015) Metabolic risk in PCOS: Phenotype and adiposity impact. Trends Endocrinol Metab 26:136–143. https://doi.org/10.1016/j.tem.2014.12.003
Shorakae S, Boyle J, Teede H (2014) Polycystic ovary syndrome: a common hormonal condition with major metabolic sequelae that physicians should know about. Intern Med J 44:720–726. https://doi.org/10.1111/imj.12495
Hara H, Yoshimura H, Uchida S et al (2001) Molecular cloning and functional expression analysis of a cDNA for human hepassocin, a liver-specific protein with hepatocyte mitogenic activity. Biochimica et biophysica acta 1520(1):45–53. https://doi.org/10.1016/s0167-4781(01)00249-4
Demchev V, Malana G, Vangala D et al (2013) Targeted deletion of fibrinogen like protein 1 reveals a novel role in energy substrate utilization. PLoS ONE. https://doi.org/10.1371/journal.pone.0058084
Huang RL, Li CH, Du YF et al (2020) Discovery of a role of the novel hepatokine, hepassocin, in obesity. BioFactors 46:100–105. https://doi.org/10.1002/biof.1574
Wu HT, Chen SC, Fan KC et al (2020) Targeting fibrinogen-like protein 1 is a novel therapeutic strategy to combat obesity. FASEB J 34:2958–2967. https://doi.org/10.1096/fj.201901925R
Ketenci Gencer F, Yuksel S, Goksever Celik H (2021) Do serum hepassocin levels change in women with polycystic ovary syndrome? Eur J Obstetr Gynecol Reprod Biol 267:137–141. https://doi.org/10.1016/j.ejogrb.2021.10.034
Wu HT, Ou HY, Hung HC et al (2016) A novel hepatokine, HFREP1, plays a crucial role in the development of insulin resistance and type 2 diabetes. Diabetologia 59:1732–1742. https://doi.org/10.1007/s00125-016-3991-7
Chen C, Lu FC, Department of Disease Control Ministry of Health PC (2004) The guidelines for prevention and control of overweight and obesity in Chinese adults. Biomed Environ Sci 17(Suppl):1–36
Matthews DR, Hosker JP, Rudenski AS et al (1985) Homeostasis model assessment: insulin resistance and ?-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419. https://doi.org/10.1007/BF00280883
Yamada C, Mitsuhashi T, Hiratsuka N et al (2011) Optimal reference interval for homeostasis model assessment of insulin resistance in a Japanese population. J Diabetes Investig 2:373–376. https://doi.org/10.1111/j.2040-1124.2011.00113.x
Wu HT, Lu FH, Ou HY et al (2013) The role of Hepassocin in the development of non-alcoholic fatty liver disease. J Hepatol 59:1065–1072. https://doi.org/10.1016/j.jhep.2013.06.004
Abdelmoemen G, Khodeir SA, Zaki AN et al (2019) Overexpression of hepassocin in diabetic patients with nonalcoholic fatty liver disease may facilitate increased hepatic lipid accumulation. Endocr Metabol Immune Disord Drug Targets 19:185–188. https://doi.org/10.2174/1871530318666180716100543
Kuba VM, Cavalieri PM, Christóforo AC et al (2006) Insulin resistance and metabolic profile in lean and overweight/obese polycystic ovary syndrome patients. Arq Bras Endocrinol Metabol 50:1026–1033. https://doi.org/10.1590/s0004-27302006000600008
Durmus U, Duran C, Ecirli S (2017) Visceral adiposity index levels in overweight and/or obese, and non-obese patients with polycystic ovary syndrome and its relationship with metabolic and inflammatory parameters. J Endocrinol Invest 40:487–497. https://doi.org/10.1007/s40618-016-0582-x
Chen M-J, Chiu H-M, Chen C-L et al (2010) Hyperandrogenemia is independently associated with elevated alanine aminotransferase activity in young women with polycystic ovary syndrome. J Clin Endocrinol Metab 95:3332–3341. https://doi.org/10.1210/jc.2009-2698
Zeng J, Lawrence WR, Yang J et al (2021) Association between serum uric acid and obesity in Chinese adults: a 9-year longitudinal data analysis. BMJ Open 11:e041919. https://doi.org/10.1136/bmjopen-2020-041919
Mu L, Pan J, Yang L et al (2018) Association between the prevalence of hyperuricemia and reproductive hormones in polycystic ovary syndrome. Reprod Biol Endocrinol 16:104. https://doi.org/10.1186/s12958-018-0419-x
Yarali H, Yildirir A, Aybar F et al (2001) Diastolic dysfunction and increased serum homocysteine concentrations may contribute to increased cardiovascular risk in patients with polycystic ovary syndrome. Fertil Steril 76:511–516. https://doi.org/10.1016/s0015-0282(01)01937-9
Macut D, Simic T, Lissounov A et al (2011) Insulin resistance in non-obese women with polycystic ovary syndrome: relation to byproducts of oxidative stress. Exp Clin Endocrinol 119:451–455. https://doi.org/10.1055/s-0031-1279740
Belmonte N, Phillips BW, Massiera F et al (2001) Activation of extracellular signal-regulated kinases and CREB/ATF-1 mediate the expression of CCAAT/enhancer binding proteins β and -δ in preadipocytes. Mol Endocrinol 15:2037–2049. https://doi.org/10.1210/mend.15.11.0721
Jung TW, Chung YH, Kim H-C et al (2018) Hyperlipidemia-induced hepassocin in the liver contributes to insulin resistance in skeletal muscle. Mol Cell Endocrinol 470:26–33. https://doi.org/10.1016/j.mce.2017.10.014
Yang Y, Liu X, Chen H et al (2022) HPS protects the liver against steatosis, cell death, inflammation, and fibrosis in mice with steatohepatitis. FEBS J. https://doi.org/10.1111/febs.16430
Li CY, Cao CZ, Xu WX et al (2010) Recombinant human hepassocin stimulates proliferation of hepatocytes in vivo and improves survival in rats with fulminant hepatic failure. Gut 59:817–826. https://doi.org/10.1136/gut.2008.171124
Kang L, Li H-Y, Ou H-Y et al (2020) Role of placental fibrinogen-like protein 1 in gestational diabetes. Transl Res 218:73–80. https://doi.org/10.1016/j.trsl.2020.01.001
Tsai I-T, Hung W-C, Lu Y-C et al (2021) Circulating hepassocin level in patients with stable angina is associated with fatty liver and renal function. Int J Med Sci 18:1–7. https://doi.org/10.7150/ijms.50646
Ou H-Y, Wu H-T, Lin C-H et al (2017) The hepatic protection effects of hepassocin in hyperglycemic crisis. J Clin Endocrinol Metab 102:2407–2415. https://doi.org/10.1210/jc.2016-3287
Acknowledgements
This study would not have been possible without the participation of the patients.
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. YZ, DD, and DC: collected the data, performed ELISA experiments, performed the statistical analysis, and interpreted the data. MC, HY, HS, and XG: prepared material and participated in recruiting subjects. XS and MZ: designed and wrote the manuscript. SQ: edited the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
All procedures performed in our study involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the Ethics Committee of Shanghai Tenth People's Hospital (approval number SHSY-IEC-4.1/21-227/01).
Informed Consent
Written informed consent was obtained from all participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Y., Dilimulati, D., Chen, D. et al. Serum fibrinogen-like protein 1 as a novel biomarker in polycystic ovary syndrome: a case–control study. J Endocrinol Invest 45, 2123–2130 (2022). https://doi.org/10.1007/s40618-022-01844-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-022-01844-0