Abstract
Purpose
It is postulated that patients with different types of pituitary neuroendocrine tumors (PitNETs) may present a higher incidence of cancer. Factors underlying individuals becoming overweight, such as insulin resistance, hyperleptinemia, and low-grade inflammation, may play a role in the risk of differentiated thyroid carcinoma (DTC) in such patients. This study aimed to investigate the frequency of and obesity-related risk factors associated with DTC in patients with PitNETs.
Methods
This cross-sectional study involved 149 patients with nonacromegalic PitNETs (AG group), 71 patients with acromegaly (ACRO group), and 156 controls (CG group). All participants underwent insulin and blood glucose measurements with the determination of the homeostatic model assessment-insulin resistance (HOMA-IR) index, leptin, and high-sensitivity C-reactive protein (hsCRP), and they also underwent thyroid ultrasound. Clinically significant nodules were biopsied for subsequent cytopathological evaluation, and participants were operated on when indicated.
Results
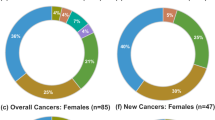
Patients in the AG group had high levels of insulin resistance and significantly higher levels of leptin and hsCRP compared with those of patients in the ACRO group. There were no cases of DTC in the AG group; two findings, one incidental, of DTC occurred in the CG group, and three cases of DTC were present in the ACRO group. Acromegaly was associated with DTC after adjusted analysis.
Conclusions
Our findings in patients with nonacromegalic PitNETs do not indicate a high risk for DTC despite the presence of metabolic and inflammatory risk factors for neoplastic events. In contrast, acromegaly promotes a greater risk of DTC.

Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Ezzat S, Asa SL, Couldwell WT, Barr CE, Dodge WE, Vance ML, McCutcheon IE (2004) The prevalence of pituitary adenomas: a systematic review. Cancer 101:613–619. https://doi.org/10.1002/cncr.20412
Popovic V, Damjanovic S, Micic D, Nesovic M, Djurovic M, Petakov M, Obradovic S, Zoric S, Simic M, Penezic Z, Marinkovic J (1998) Increased incidence of neoplasia in patients with pituitary adenomas. The pituitary study group. Clin Endocrinol (Oxf) 49:441–445. https://doi.org/10.1046/j.1365-2265.1998.00536.x
Olsson DS, Hammarstrand C, Bryngelsson IL, Nilsson AG, Andersson E, Johannsson G, Ragnarsson O (2017) Incidence of malignant tumours in patients with a non-functioning pituitary adenoma. Endocr Relat Cancer 24:227–235. https://doi.org/10.1530/erc-16-0518
Terzolo M, Reimondo G, Berchialla P et al (2017) Acromegaly is associated with increased cancer risk: a survey in Italy. Endocr Relat Cancer 24:495–504. https://doi.org/10.1530/erc-16-0553
Boguszewski CL, Ayuk J (2016) Management of endocrine disease: acromegaly and cancer: an old debate revisited. Eur J Endocrinol 175:R147–R156. https://doi.org/10.1530/eje-16-0178
Dal J, Leisner MZ, Hermansen K, Farkas DK, Bengtsen M, Kistorp C, Nielsen EH, Andersen M, Feldt-Rasmussen U, Dekkers OM, Sørensen HT, Jørgensen JOL (2018) Cancer incidence in patients with acromegaly: a cohort study and meta-analysis of the literature. J Clin Endocrinol Metab 103:2182–2188. https://doi.org/10.1210/jc.2017-02457
Rokkas T, Pistiolas D, Sechopoulos P, Margantinis G, Koukoulis G (2008) Risk of colorectal neoplasm in patients with acromegaly: a meta-analysis. World J Gastroenterol 14:3484–3489. https://doi.org/10.3748/wjg.14.3484
dos Santos MC, Nascimento GC, Nascimento AG, Carvalho VC, Lopes MH, Montenegro R, Montenegro R Jr, Vilar L, Albano MF, Alves AR, Parente CV, dos Santos FM (2013) Thyroid cancer in patients with acromegaly: a case-control study. Pituitary 16:109–114. https://doi.org/10.1007/s11102-012-0383-y
Wolinski K, Czarnywojtek A, Ruchala M (2014) Risk of thyroid nodular disease and thyroid cancer in patients with acromegaly–meta-analysis and systematic review. PLoS ONE 9:e88787. https://doi.org/10.1371/journal.pone.0088787
Roberts DL, Dive C, Renehan AG (2010) Biological mechanisms linking obesity and cancer risk: new perspectives. Annu Rev Med 61:301–316. https://doi.org/10.1146/annurev.med.080708.082713
Pekic S, Popovic V (2013) GH therapy and cancer risk in hypopituitarism: what we know from human studies. Eur J Endocrinol 169:R89–R97. https://doi.org/10.1530/eje-13-0389
Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M (2008) Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet 371:569–578. https://doi.org/10.1016/s0140-6736(08)60269-x
Pappa T, Alevizaki M (2014) Obesity and thyroid cancer: a clinical update. Thyroid 24:190–199. https://doi.org/10.1089/thy.2013.0232
Chanson P, Raverot G, Castinetti F, Cortet-Rudelli C, Galland F, Salenave S (2015) Management of clinically non-functioning pituitary adenoma. Ann Endocrinol (Paris) 76:239–247. https://doi.org/10.1016/j.ando.2015.04.002
Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, Wass JA (2011) Diagnosis and treatment of hyperprolactinemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 96:273–288. https://doi.org/10.1210/jc.2010-1692
Nieman LK, Biller BM, Findling JW, Newell-Price J, Savage MO, Stewart PM, Montori VM (2008) The diagnosis of Cushing's syndrome: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 93:1526–1540. https://doi.org/10.1210/jc.2008-0125
Katznelson L, Laws ER, Melmed S, Molitch ME, Murad MH, Utz A, Wass JA (2014) Acromegaly: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 99:3933–3951. https://doi.org/10.1210/jc.2014-2700
Geloneze B, Repetto EM, Geloneze SR, Tambascia MA, Ermetice MN (2006) The threshold value for insulin resistance (HOMA-IR) in an admixtured population IR in the Brazilian metabolic syndrome study. Diabetes Res Clin Pract 72:219–220. https://doi.org/10.1016/j.diabres.2005.10.017
American Diabetes Association (2019) 2. Classification and diagnosis of diabetes: standards of medical care in diabetes-2019. Diabetes Care 42:S13–S28. https://doi.org/10.2337/dc19-S002
Fleseriu M, Hashim IA, Karavitaki N, Melmed S, Murad MH, Salvatori R, Samuels MH (2016) Hormonal replacement in hypopituitarism in adults: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 101:3888–3921. https://doi.org/10.1210/jc.2016-2118
Haugen BR, Alexander EK, Bible KC et al (2016) 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid 26:1–133. https://doi.org/10.1089/thy.2015.0020
Ali S, Cibas E (2018) The Bethesda system for reporting thyroid cytopathology: definitions, criteria and explanatory notes. Springer International Publishing, Cham
Lloyd RV, Osamura RY, Klöppel G, Rosai J (2017) Tumours of the thyroid gland. In: Lloyd RV, Osamura RY, Klöppel G, Rosai J (eds) WHO classification of tumours of endocrine organs. IARC, Lyon, pp 65–142
Minniti G, Traish D, Ashley S, Gonsalves A, Brada M (2005) Risk of second brain tumor after conservative surgery and radiotherapy for pituitary adenoma: update after an additional 10 years. J Clin Endocrinol Metab 90:800–804. https://doi.org/10.1210/jc.2004-1152
Norberg L, Johansson R, Rasmuson T (2008) Pituitary adenomas in northern Sweden: a study on therapy choices and the risk of second primary tumours. Clin Endocrinol (Oxf) 68:780–785. https://doi.org/10.1111/j.1365-2265.2007.03118.x
Berinder K, Akre O, Granath F, Hulting AL (2011) Cancer risk in hyperprolactinemia patients: a population-based cohort study. Eur J Endocrinol 165:209–215. https://doi.org/10.1530/eje-11-0076
Sattler MG, van Beek AP, Wolffenbuttel BH, van den Berg G, Sluiter WJ, Langendijk JA, van den Bergh AC (2012) The incidence of second tumours and mortality in pituitary adenoma patients treated with postoperative radiotherapy versus surgery alone. Radiother Oncol 104:125–130. https://doi.org/10.1016/j.radonc.2012.04.024
Wolinski K, Stangierski A, Dyrda K, Nowicka K, Pelka M, Iqbal A, Car A, Lazizi M, Bednarek N, Czarnywojtek A, Gurgul E, Ruchala M (2017) Risk of malignant neoplasms in acromegaly: a case–control study. J Endocrinol Invest 40:319–322. https://doi.org/10.1007/s40618-016-0565-y
Tam AA, Kaya C, Aydın C, Ersoy R, Çakır B (2016) Differentiated thyroid cancer in patients with prolactinoma. Turk J Med Sci 46:1360–1365. https://doi.org/10.3906/sag-1501-58
Lauby-Secretan B, Scoccianti C, Loomis D, Grosse Y, Bianchini F, Straif K (2016) Body fatness and cancer–viewpoint of the IARC working group. N Engl J Med 375:794–798. https://doi.org/10.1056/NEJMsr1606602
Schmid D, Ricci C, Behrens G, Leitzmann MF (2015) Adiposity and risk of thyroid cancer: a systematic review and meta-analysis. Obes Rev 16:1042–1054. https://doi.org/10.1111/obr.12321
Yin DT, He H, Yu K, Xie J, Lei M, Ma R, Li H, Wang Y, Liu Z (2018) The association between thyroid cancer and insulin resistance, metabolic syndrome and its components: a systematic review and meta-analysis. Int J Surg 57:66–75. https://doi.org/10.1016/j.ijsu.2018.07.013
He Q, Sun H, Li F, Liang N (2019) Obesity and risk of differentiated thyroid cancer: a large-scale case–control study. Clin Endocrinol (Oxf) 91:869–878. https://doi.org/10.1111/cen.14091
Shin HY, Jee YH, Cho ER (2017) Body mass index and incidence of thyroid cancer in Korea: the Korean cancer prevention study-II. J Cancer Res Clin Oncol 143:143–149. https://doi.org/10.1007/s00432-016-2261-x
Manole D, Schildknecht B, Gosnell B, Adams E, Derwahl M (2001) Estrogen promotes growth of human thyroid tumor cells by different molecular mechanisms. J Clin Endocrinol Metab 86:1072–1077. https://doi.org/10.1210/jcem.86.3.7283
Berger NA (2018) Young adult cancer: influence of the obesity pandemic. Obesity (Silver Spring, Md) 26:641–650. https://doi.org/10.1002/oby.22137
Sung H, Siegel RL, Rosenberg PS, Jemal A (2019) Emerging cancer trends among young adults in the USA: analysis of a population-based cancer registry. Lancet Public Health 4:e137–e147. https://doi.org/10.1016/s2468-2667(18)30267-6
Koroukian SM, Dong W, Berger NA (2019) Changes in age distribution of obesity-associated cancers. JAMA Netw Open 2:e199261. https://doi.org/10.1001/jamanetworkopen.2019.9261
Nyasani E, Munir I, Perez M, Payne K, Khan S (2019) Linking obesity-induced leptin-signaling pathways to common endocrine-related cancers in women. Endocrine 63:3–17. https://doi.org/10.1007/s12020-018-1748-4
Lee S, Choe J-W, Kim H-K, Sung J (2011) High-sensitivity C-reactive protein and cancer. J Epidemiol 21:161–168. https://doi.org/10.2188/jea.je20100128
Dossus L, Franceschi S, Biessy C et al (2018) Adipokines and inflammation markers and risk of differentiated thyroid carcinoma: the EPIC study. Int J Cancer 142:1332–1342. https://doi.org/10.1002/ijc.31172
Kitahara CM, Farkas DKR, Jørgensen JOL, Cronin-Fenton D, Sørensen HT (2018) Benign thyroid diseases and risk of thyroid cancer: a nationwide cohort study. J Clin Endocrinol Metab 103:2216–2224. https://doi.org/10.1210/jc.2017-02599
Li M, Maso LD, Vaccarella S (2020) Global trends in thyroid cancer incidence and the impact of overdiagnosis. Lancet Diabetes Endocrinol 8:468–470. https://doi.org/10.1016/s2213-8587(20)30115-7
Kitahara CM, Sosa JA (2016) The changing incidence of thyroid cancer. Nat Rev Endocrinol 12:646–653. https://doi.org/10.1038/nrendo.2016.110
Cheng S, Gomez K, Serri O, Chik C, Ezzat S (2015) The role of diabetes in acromegaly associated neoplasia. PLoS ONE 10:e0127276. https://doi.org/10.1371/journal.pone.0127276
Acknowledgements
The authors thank the CEPEC staff nurses for their logistical support. We are especially grateful to Adriana Guimarães and Patrícia Azevedo for their assistance with data management.
Funding
No funding was received.
Author information
Authors and Affiliations
Contributions
GCN, MSF and MBG contributed to the study conception and design. Data acquisition, analysis and interpretation were performed by GCN, AGPACN, CMVP, VPR, RSSA, VCCR, SSPD and MM. GCN, VPR, MM and MSF drafted the manuscript. All authors revised the work critically for valuable intellectual content, read and approved the final version to be published, and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethics approval
All procedures performed in studies involving human participants were conducted in accordance with the ethical standards of the institutional and/or national research committee (this study was approved by the Research and Ethics Committee of the Presidente Dutra University Hospital of the Federal University of Maranhão under the number 1.258.417) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Research involving human participants and/or animals
This article does not involve any studies with animals performed by any of the authors.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cortês Nascimento, G., de Araujo Cortês Nascimento, A.G.P., de Maria Ribeiro Veiga Parente, C. et al. Pituitary neuroendocrine tumors and differentiated thyroid cancer: do metabolic and inflammatory risk factors play roles?. J Endocrinol Invest 44, 735–744 (2021). https://doi.org/10.1007/s40618-020-01357-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-020-01357-8