Abstract
Purpose
Thyroid hormones are essential for the normal function of almost all human tissues, and have critical roles in metabolism, differentiation and growth. Free triiodothyronine (fT3), free thyroxine (fT4) and thyroid-stimulating hormone (TSH) levels are under strong genetic influence; however, most of the heritability is yet unexplained.
Methods
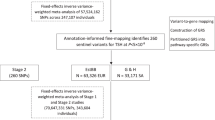
In order to identify novel loci associated with fT3, fT4 and TSH serum levels we performed a genome-wide meta-analysis of 7 411 206 polymorphisms in up to 1731 euthyroid individuals from three Croatian cohorts from Dalmatia region: two genetically isolated island populations and one mainland population. Additionally, we also performed a bivariate analysis of fT3 and fT4 levels.
Results
The EPHB2 gene variant rs67142165 reached genome-wide significance for association with fT3 plasma levels (P = 9.27 × 10−9) and its significance was confirmed in bivariate analysis (P = 9.72 × 10−9). We also found a genome-wide significant association for variant rs13037502 upstream of the PTPN1 gene and TSH plasma levels (P = 1.67 × 10−8).
Conclusion
We identified a first genome-wide significant variant associated with fT3 plasma levels, as well as a novel locus associated with TSH plasma levels. These findings are biologically relevant and enrich our knowledge about the genetic basis of pituitary-thyroid axis function.



Similar content being viewed by others
References
Rousset B, Dupuy C, Miot F, Dumont J (2000) Thyroid hormone synthesis and secretion. In: Feingold KR, Anawalt B, Boyce A et al. (eds) Endotext. MDText.com, Inc., South Dartmouth. https://www.ncbi.nlm.nih.gov/books/NBK285550/. Accessed 20 August 2018
Stockigt J (2003) Assessment of thyroid function: towards an integrated laboratory—clinical approach. Clin Biochem Rev 24(4):109–122
Nussey SWS (2001) Endocrinology: an integrated approach. the thyroid gland. BIOS Scientific Publishers, Oxford. https://www.ncbi.nlm.nih.gov/books/NBK22/. Accessed 20 August 2018
Yen PM (2001) Physiological and molecular basis of thyroid hormone action. Physiol Rev 81(3):1097–1142. https://doi.org/10.1152/physrev.2001.81.3.1097
Visser TJ, Kaptein E, Terpstra OT, Krenning EP (1988) Deiodination of thyroid hormone by human liver. J Clin Endocrinol Metab 67(1):17–24. https://doi.org/10.1210/jcem-67-1-17
Andersen S, Pedersen KM, Bruun NH, Laurberg P (2002) Narrow individual variations in serum T(4) and T(3) in normal subjects: a clue to the understanding of subclinical thyroid disease. J Clin Endocrinol Metab 87(3):1068–1072. https://doi.org/10.1210/jcem.87.3.8165
Meier CA, Maisey MN, Lowry A, Muller J, Smith MA (1993) Interindividual differences in the pituitary-thyroid axis influence the interpretation of thyroid function tests. Clin Endocrinol 39(1):101–107. https://doi.org/10.1111/j.1365-2265.1993.tb01758.x
Biondi B, Cooper DS (2008) The clinical significance of subclinical thyroid dysfunction. Endocr Rev 29(1):76–131. https://doi.org/10.1210/er.2006-0043
Asvold, B.O., Vatten, L.J., Nilsen, T.I., Bjoro, T.: The association between TSH within the reference range and serum lipid concentrations in a population-based study The HUNT Study. Eur J Endocrinol 156(2), 181–186 (2007). https://doi.org/10.1530/eje.1.02333
Wang F, Tan Y, Wang C, Zhang X, Zhao Y, Song X, Zhang B, Guan Q, Xu J, Zhang J, Zhang D, Lin H, Yu C, Zhao J (2012) Thyroid-stimulating hormone levels within the reference range are associated with serum lipid profiles independent of thyroid hormones. J Clin Endocrinol Metab 97(8):2724–2731. https://doi.org/10.1210/jc.2012-1133
van Tienhoven-Wind LJ, Dullaart RP (2017) Increased leptin/adiponectin ratio relates to low-normal thyroid function in metabolic syndrome. Lipids Health Dis 16(1):6. https://doi.org/10.1186/s12944-016-0403-4
Knudsen N, Laurberg P, Rasmussen LB, Bulow I, Perrild H, Ovesen L, Jorgensen T (2005) Small differences in thyroid function may be important for body mass index and the occurrence of obesity in the population. J Clin Endocrinol Metab 90(7):4019–4024. https://doi.org/10.1210/jc.2004-2225
Le TN, Celi FS, Wickham EP (2016) Thyrotropin levels are associated with cardiometabolic risk factors in euthyroid adolescents. Thyroid 26(10):1441–1449. https://doi.org/10.1089/thy.2016.0055
Bielecka-Dabrowa A, Mikhailidis DP, Rysz J, Banach M (2009) The mechanisms of atrial fibrillation in hyperthyroidism. Thyroid Res 2:4–4. https://doi.org/10.1186/1756-6614-2-4
Hansen PS, Brix TH, Sorensen TI, Kyvik KO, Hegedus L (2004) Major genetic influence on the regulation of the pituitary-thyroid axis: a study of healthy Danish twins. J Clin Endocrinol Metab 89(3):1181–1187. https://doi.org/10.1210/jc.2003-031641
Panicker V, Wilson SG, Spector TD, Brown SJ, Falchi M, Richards JB, Surdulescu GL, Lim EM, Fletcher SJ, Walsh JP (2008) Heritability of serum TSH, free T4 and free T3 concentrations: a study of a large UK twin cohort. Clin Endocrinol 68(4):652–659. https://doi.org/10.1111/j.1365-2265.2007.03079.x
Medici M, Visser TJ, Peeters RP (2017) Genetics of thyroid function. Best Pract Res Clin Endocrinol Metab 31(2):129–142. https://doi.org/10.1016/j.beem.2017.04.002
Taylor, P.N., Porcu, E., Chew, S., Campbell, P.J., Traglia, M., Brown, S.J., Mullin, B.H., Shihab, H.A., Min, J., Walter, K., Memari, Y., Huang, J., Barnes, M.R., Beilby, J.P., Charoen, P., Danecek, P., Dudbridge, F., Forgetta, V., Greenwood, C., Grundberg, E., Johnson, A.D., Hui, J., Lim, E.M., McCarthy, S., Muddyman, D., Panicker, V., Perry, J.R., Bell, J.T., Yuan, W., Relton, C., Gaunt, T., Schlessinger, D., Abecasis, G., Cucca, F., Surdulescu, G.L., Woltersdorf, W., Zeggini, E., Zheng, H.F., Toniolo, D., Dayan, C.M., Naitza, S., Walsh, J.P., Spector, T., Davey Smith, G., Durbin, R., Richards, J.B., Sanna, S., Soranzo, N., Timpson, N.J., Wilson, S.G., UK Consortium: Whole-genome sequence-based analysis of thyroid function. Nat Commun 6, 5681 (2015). https://doi.org/10.1038/ncomms6681
Rudan I, Marusic A, Jankovic S, Rotim K, Boban M, Lauc G, Grkovic I, Dogas Z, Zemunik T, Vatavuk Z, Bencic G, Rudan D, Mulic R, Krzelj V, Terzic J, Stojanovic D, Puntaric D, Bilic E, Ropac D, Vorko-Jovic A, Znaor A, Stevanovic R, Biloglav Z, Polasek O (2009) "10 001 Dalmatians:" Croatia launches Its National Biobank. Croat Med J 50(1):4–6. https://doi.org/10.3325/cmj.2009.50.4
Aulchenko YS, Ripke S, Isaacs A, Van Duijn CM (2007) GenABEL: an R library for genome-wide association analysis. Bioinformatics 23(10):1294–1296. https://doi.org/10.1093/bioinformatics/btm108
Marchini J, Howie B, Myers S, McVean G, Donnelly P (2007) A new multipoint method for genome-wide association studies by imputation of genotypes. Nat Genet 39(7):906–913. https://doi.org/10.1038/ng2088
Struchalin MV, Amin N, Eilers PHC, van Duijn CM, Aulchenko YS (2012) An R package "VariABEL" for genome-wide searching of potentially interacting loci by testing genotypic variance heterogeneity. Bmc Genet 13(1):4. https://doi.org/10.1186/1471-2156-13-4
Schwarzer G (2007) meta: an R package for meta-analysis 7(3):40-45. https://cran.r-project.org/doc/Rnews/Rnews_2007-3.pdf. Accessed 15 August 2018
Pruim RJ, Welch RP, Sanna S, Teslovich TM, Chines PS, Gliedt TP, Boehnke M, Abecasis GR, Willer CJ (2010) LocusZoom: regional visualization of genome-wide association scan results. Bioinformatics 26(18):2336–2337. https://doi.org/10.1093/bioinformatics/btq419
Turner, S.D.: qqman: an R package for visualizing GWAS results using Q-Q and manhattan plots. biorXiv (2017). https://doi.org/10.1101/005165
Shen X, Klaric L, Sharapov S, Mangino M, Ning Z, Wu D, Trbojevic-Akmacic I, Pucic-Bakovic M, Rudan I, Polasek O, Hayward C, Spector TD, Wilson JF, Lauc G, Aulchenko YS (2017) Multivariate discovery and replication of five novel loci associated with Immunoglobulin G N-glycosylation. Nat Commun 8(1):447. https://doi.org/10.1038/s41467-017-00453-3
Manning G, Whyte DB, Martinez R, Hunter T, Sudarsanam S (2002) The protein kinase complement of the human genome. Science 298(5600):1912–1934. https://doi.org/10.1126/science.1075762
Cantisani MC, Parascandolo A, Perala M, Allocca C, Fey V, Sahlberg N, Merolla F, Basolo F, Laukkanen MO, Kallioniemi OP, Santoro M, Castellone MD (2016) A loss-of-function genetic screening identifies novel mediators of thyroid cancer cell viability. Oncotarget 7(19):28510–28522. https://doi.org/10.18632/oncotarget.8577
Park I, Lee H-S (2015) EphB/ephrinB signaling in cell adhesion and migration. Mol Cells 38(1):14–19. https://doi.org/10.14348/molcells.2015.2116
Salvucci O, Tosato G (2012) Essential roles of EphB receptors and EphrinB ligands in endothelial cell function and angiogenesis. Adv Cancer Res 114:21–57. https://doi.org/10.1016/b978-0-12-386503-8.00002-8
Westerlund J, Andersson L, Liang S, Carlsson T, Nilsson M, Amendola E, Fagman H (2011) Role of EphA4 receptor signaling in thyroid development: regulation of folliculogenesis and propagation of the C-cell lineage. Endocrinology 152(3):1154–1164. https://doi.org/10.1210/en.2010-0232
Benvenga S, Guarneri F (2019) Thyroid hormone binding motifs and iodination pattern of thyroglobulin. Front Biosci 24:212–230
Sweeney C, Lai C, Riese DJ 2nd, Diamonti AJ, Cantley LC, Carraway KL 3rd (2000) Ligand discrimination in signaling through an ErbB4 receptor homodimer. J Biol Chem 275(26):19803–19807. https://doi.org/10.1074/jbc.C901015199
Sundvall M, Peri L, Maatta JA, Tvorogov D, Paatero I, Savisalo M, Silvennoinen O, Yarden Y, Elenius K (2007) Differential nuclear localization and kinase activity of alternative ErbB4 intracellular domains. Oncogene 26(48):6905–6914. https://doi.org/10.1038/sj.onc.1210501
Haugen DR, Akslen LA, Varhaug JE, Lillehaug JR (1996) Expression of c-erbB-3 and c-erbB-4 proteins in papillary thyroid carcinomas. Can Res 56(6):1184–1188
Chu X, Pan CM, Zhao SX, Liang J, Gao GQ, Zhang XM, Yuan GY, Li CG, Xue LQ, Shen M, Liu W, Xie F, Yang SY, Wang HF, Shi JY, Sun WW, Du WH, Zuo CL, Shi JX, Liu BL, Guo CC, Zhan M, Gu ZH, Zhang XN, Sun F, Wang ZQ, Song ZY, Zou CY, Sun WH, Guo T, Cao HM, Ma JH, Han B, Li P, Jiang H, Huang QH, Liang LM, Liu LB, Chen G, Su Q, Peng YD, Zhao JJ, Ning G, Chen Z, Chen JL, Chen SJ, Huang W, Song HD, Genetics CC (2011) A genome-wide association study identifies two new risk loci for Graves' disease. Nat Genet 43(9):897–901. https://doi.org/10.1038/ng.898
Hansen PS, Brix TH, Iachine I, Sorensen TIA, Kyvik KO, Hegedus L (2007) Genetic and environmental interrelations between measurements of thyroid function in a healthy Danish twin population. AJP Endocrinol Metab 292(3):765–770. https://doi.org/10.1152/ajpendo.00321.2006
Uhlén, M., Fagerberg, L., Hallström, B.M., Lindskog, C., Oksvold, P., Mardinoglu, A., Sivertsson, Å., Kampf, C., Sjöstedt, E., Asplund, A., Olsson, I., Edlund, K., Lundberg, E., Navani, S., Szigyarto, C.A.-K., Odeberg, J., Djureinovic, D., Takanen, J.O., Hober, S., Alm, T., Edqvist, P.-H., Berling, H., Tegel, H., Mulder, J., Rockberg, J., Nilsson, P., Schwenk, J.M., Hamsten, M., von Feilitzen, K., Forsberg, M., Persson, L., Johansson, F., Zwahlen, M., von Heijne, G., Nielsen, J., Pontén, F.: Tissue-based map of the human proteome. Science (2015). https://doi.org/10.1126/science.1260419
Robinson DR, Wu YM, Lin SF (2000) The protein tyrosine kinase family of the human genome. Oncogene 19(49):5548–5557. https://doi.org/10.1038/sj.onc.1203957
Fallahi P, Ferrari SM, Vita R, Di Domenicantonio A, Corrado A, Benvenga S, Antonelli A (2014) Thyroid dysfunctions induced by tyrosine kinase inhibitors. Expert Opin Drug Saf 13(6):723–733. https://doi.org/10.1517/14740338.2014.913021
Ahmadieh H, Salti I (2013) Tyrosine kinase inhibitors induced thyroid dysfunction: a review of its incidence, pathophysiology, clinical relevance, and treatment. Biomed Res Int 2013:9. https://doi.org/10.1155/2013/725410
Tsou RC, Bence KK (2012) The genetics of PTPN1 and obesity: insights from mouse models of tissue-specific PTP1B deficiency. J Obes 2012:926857. https://doi.org/10.1155/2012/926857
Michalaki MA, Vagenakis AG, Leonardou AS, Argentou MN, Habeos LG, Makri MG, Psyrogiannis AI, Kalfarentzos FE, Kyriazopoulou VE (2006) Thyroid function in humans with morbid obesity. Thyroid 16(1):73–78. https://doi.org/10.1089/thy.2006.16.73
Mueller A, Schofl C, Dittrich R, Cupisti S, Oppelt PG, Schild RL, Beckmann MW, Haberle L (2009) Thyroid-stimulating hormone is associated with insulin resistance independently of body mass index and age in women with polycystic ovary syndrome. Hum Reprod 24(11):2924–2930. https://doi.org/10.1093/humrep/dep285
Bakker SJL, ter Maaten JC, Popp-Snijders C, Slaets JPJ, Heine RJ, Gans ROB (2001) The relationship between thyrotropin and low density lipoprotein cholesterol is modified by insulin sensitivity in healthy euthyroid subjects. J Clin Endocr Metab 86(3):1206–1211. https://doi.org/10.1210/Jc.86.3.1206
Chubb SAP, Davis WA, Davis TME (2005) Interactions among thyroid function, insulin sensitivity, and serum lipid concentrations: The Fremantle Diabetes Study. J Clin Endocr Metab 90(9):5317–5320. https://doi.org/10.1210/jc.2005-0298
Duarte GC, Cendoroglo MS, Araujo LM, Almada Filho Cde M (2015R) Association between increased serum thyrotropin concentration and the oldest old: what do we know? Einstein (Sao Paulo) 13(1):117–121. https://doi.org/10.1590/S1679-45082015RW2874
Antunes TT, Gagnon A, Chen B, Pacini F, Smith TJ, Sorisky A (2006) Interleukin-6 release from human abdominal adipose cells is regulated by thyroid-stimulating hormone: effect of adipocyte differentiation and anatomic depot. Am J Physiol Endocrinol Metab 290(6):1140–1144. https://doi.org/10.1152/ajpendo.00516.2005
Li QL, Yang GZ, Wang Y, Zhang XP, Sang Q, Wang HA, Zhao XZ, Xing QH, He L, Wang L (2011) Common genetic variation in the 3'-untranslated region of gonadotropin-releasing hormone receptor regulates gene expression in cella and is associated with thyroid function, insulin secretion as well as insulin sensitivity in polycystic ovary syndrome patients. Hum Genet 129(5):553–561. https://doi.org/10.1007/s00439-011-0954-4
Porcu, E., Medici, M., Pistis, G., Volpato, C.B., Wilson, S.G., Cappola, A.R., Bos, S.D., Deelen, J., den Heijer, M., Freathy, R.M., Lahti, J., Liu, C.Y., Lopez, L.M., Nolte, I.M., O’Connell, J.R., Tanaka, T., Trompet, S., Arnold, A., Bandinelli, S., Beekman, M., Bohringer, S., Brown, S.J., Buckley, B.M., Camaschella, C., de Craen, A.J.M., Davies, G., de Visser, M.C.H., Ford, I., Forsen, T., Frayling, T.M., Fugazzola, L., Gogele, M., Hattersley, A.T., Hermus, A.R., Hofman, A., Houwing-Duistermaat, J.J., Jensen, R.A., Kajantie, E., Kloppenburg, M., Lim, E.M., Masciullo, C., Mariotti, S., Minelli, C., Mitchell, B.D., Nagaraja, R., Netea-Maier, R.T., Palotie, A., Persani, L., Piras, M.G., Psaty, B.M., Raikkonen, K., Richards, J.B., Rivadeneira, F., Sala, C., Sabra, M.M., Sattar, N., Shields, B.M., Soranzo, N., Starr, J.M., Stott, D.J., Sweep, F.C.G.J., Usala, G., van der Klauw, M.M., van Heemst, D., van Mullem, A., Vermeulen, S.H., Visser, W.E., Walsh, J.P., Westendorp, R.G.J., Widen, E., Zhai, G.J., Cucca, F., Deary, I.J., Eriksson, J.G., Ferrucci, L., Fox, C.S., Jukema, J.W., Kiemeney, L.A., Pramstaller, P.P., Schlessinger, D., Shuldiner, A.R., Slagboom, E.P., Uitterlinden, A.G., Vaidya, B., Visser, T.J., Wolffenbuttel, B.H.R., Meulenbelt, I., Rotter, J.I., Spector, T.D., Hicks, A.A., Toniolo, D., Sanna, S., Peeters, R.P., Naitza, S.: A Meta-analysis of thyroid-related traits reveals novel loci and gender-specific differences in the regulation of thyroid function. Plos Genet (2013). https://doi.org/10.1371/journal.pgen.1003266
Acknowledgements
The Croatian Science Foundation funded this work under the project “Identification of new genetic loci implicated in regulation of thyroid and parathyroid function” (Grant No. 1498). The “10 001 Dalmatians” project was funded by grants from the Medical Research Council (UK), European Commission Framework 6 project EUROSPAN (Contract No. LSHG-CT-2006-018947), the Republic of Croatia Ministry of Science, Education and Sports research Grant (216-1080315-0302), the Croatian Science Foundation (Grant 8875), CEKOM (Ministry of Economy, Entrepreneurship and Crafts) and the Research Centre of Excellence in Personalized Medicine (Ministry of Science and Education). We would like to thank all participants of this study and acknowledge invaluable support of the local teams in Zagreb and Split, especially that of the Institute for Anthropological Research, Zagreb, Croatia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Popović, M., Matana, A., Torlak, V. et al. Genome-wide meta-analysis identifies novel loci associated with free triiodothyronine and thyroid-stimulating hormone. J Endocrinol Invest 42, 1171–1180 (2019). https://doi.org/10.1007/s40618-019-01030-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-019-01030-9