Abstract
Purpose
Feto-placental unit represents an important source of activin A, a member of transforming growth factors-β involved in the mechanisms of labor. No evidences are available on activin A in pregnancies beyond 41 weeks of gestation, where induction of labor is often required. The present study aimed to evaluate activin A maternal serum levels and placental mRNA expression in term and late-term pregnancy, with spontaneous or induced labor, and its possible role to predict the response to labor induction.
Methods
Maternal serum samples and placental specimens were collected from women with singleton pregnancy admitted for either term spontaneous labor (n = 23) or induction of labor for late-term pregnancy (n = 41), to evaluate activin A serum levels and placental mRNA expression. Univariate and multivariate analyses on activin A serum levels, maternal clinical parameters, and cervical length were conducted in women undergoing induction of labor.
Results
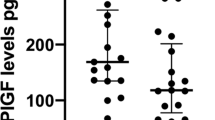
Maternal serum activin A levels and placental activin A mRNA expression in late-term pregnancies were significantly higher than at term. Late-term pregnancies who did not respond to induction of labor showed significantly lower levels of activin A compared to responders. The combination of serum activin A and cervical length achieved a sensitivity of 100% and a specificity of 93.55% for the prediction of successful induction.
Conclusion
Late-term pregnancy is characterized by hyperexpression of placental activin A and increased maternal activin A secretion. By combining maternal serum activin A levels with cervical length, a good predictive model for the response to induction of labor was elaborated.



Similar content being viewed by others
References
Leduc D, Biringer A, Lee L et al (2013) Induction of labour. J Obstet Gynaecol Can 35:840–860
Spong CY (2013) Defining “term” pregnancy: recommendations from the Defining “Term” Pregnancy Workgroup. Jama 309:2445–2446. doi:10.1001/jama.2013.6235
ACOG (2014) Practice bulletin no. 146: management of late-term and postterm pregnancies. Obstet Gynecol 124:390–396. doi:10.1097/01.AOG.0000452744.06088.48
Alexander JM, McIntire DD, Leveno KJ (2000) Forty weeks and beyond: pregnancy outcomes by week of gestation. Obstet Gynecol 96:291–294. doi:10.1016/S0029-7844(00)00862-0
Ramirez M, Ramin S (2009) ACOG practice bulletin No. 107: induction of labor. Obstet Gynecol 114:386–397. doi:10.1097/AOG.0b013e3181b48ef5
Gatward H, Simpson M, Woodhart L, Stainton MC (2010) Women’s experiences of being induced for late-term pregnancy. Women Birth 231:3–9
Crowley P (2006) WITHDRAWN: Interventions for preventing or improving the outcome of delivery at or beyond term. Cochrane Database Syst Rev CD000170. doi:10.1002/14651858.cd000170
Galal M, Symonds I, Murray H, Petraglia F, Smith R (2012) Postterm pregnancy. Facts Views Vis ObGyn. 4:175–187.
Menon R, Bonney EA, Condon J et al (2016) Novel concepts on pregnancy clocks and alarms: redundancy and synergy in human parturition. Hum Reprod Update 22:535–560. doi:10.1093/humupd/dmw022
Reis FM, Gervasi MT, Florio P et al (2003) Prediction of successful induction of labor at term: Role of clinical history, digital examination, ultrasound assessment of the cervix, and fetal fibronectin assay. Am J Obstet Gynecol 189:1361–1367. doi:10.1067/S0002-9378(03)00725-7
Gibson KS, Waters TP (2015) Measures of success: prediction of successful labor induction. Semin Perinatol 39:475–482. doi:10.1053/j.semperi.2015.07.012
Torricelli M, Voltolini C, Conti N et al (2013) Weight gain regardless of pre-pregnancy BMI and influence of fetal gender in response to labor induction in postdate pregnancy. J Matern Fetal Neonatal Med 26:1016–1019. doi:10.3109/14767058.2013.766712
Torricelli M, Voltolini C, Vellucci FL, et al (2013) Fetal gender effects on induction of labor in postdate pregnancies. Reprod Sci 20:670–674. doi:10.1177/1933719112462631
Schoen C, Navathe R (2015) Failed induction of labor. Semin Perinatol 39:483–487. doi:10.1053/j.semperi.2015.07.013
Ling N, Ying SY, Ueno N et al (1986) Pituitary FSH is released by a heterodimer of the beta-subunits from the two forms of inhibin. Nature 321:779–782. doi:10.1038/321779a0
Ueno N, Nishimatsu S, Murakami K (1990) Activin as a cell differentiation factor. Prog Growth Factor Res 2:113–124.
Wijayarathna R, de Kretser DM (2016) Activins in reproductive biology and beyond. Hum Reprod Update 22:342–357. doi:10.1093/humupd/dmv058
Schneider-Kolsky M, D’Antona D, Evans LW et al (2000) Maternal serum total activin A and follistatin in pregnancy and parturition. BJOG 107:995–1000
Muttukrishna S, Tannetta D, Groome N, Sargent I (2004) Activin and follistatin in female reproduction. Mol Cell Endocrinol 225:45–56
Petraglia F, Gallinelli A, de Vita D, et al (1994) Activin at parturition: Changes of maternal serum levels and evidence for binding sites in placenta and fetal membranes. Obstet Gynecol 84:278–282.
O’Connor AE, McFarlane JR, Hayward S et al (1999) Serum activin A and follistatin concentrations during human pregnancy: a cross-sectional and longitudinal study. Hum Reprod 14:827–832. doi:10.1093/humrep/14.3.827
Torricelli M, Voltolini C, Novembri R et al (2012) Activin A and its regulatory molecules in placenta and fetal membranes of women with preterm premature rupture of the membranes associated with acute chorioamnionitis. Am J Reprod Immunol 68:392–399. doi:10.1111/j.1600-0897.2012.01180.x
Norwitz ER, Robinson JN, Repke JT (2001) Labor and delivery. In: Gabbe SG, Niebyl GR, Simpson JL (eds) Obstetrics: normal and problem pregnancies, 3rd edn. Churchill Livingstone, New York 2003.
Gilstrop M, Sciscione A (2015) Induction of labor-Pharmacology methods. Semin Perinatol 39:463–465. doi:10.1053/j.semperi.2015.07.009
Thomas J, Fairclough A, Kavanagh J, Kelly AJ (2014) Vaginal prostaglandin (PGE2 and PGF2a) for induction of labour at term. Cochrane Database Syst Rev 6:CD003101. doi:10.1002/14651858.CD003101.pub3
Cindrova-Davies T, Yung H-W, Johns J et al (2007) Oxidative stress, gene expression, and protein changes induced in the human placenta during labor. Am J Pathol 171:1168–1179. doi:10.2353/ajpath.2007.070528
Cindrova-Davies T, Spasic-Boskovic O, Jauniaux E et al (2007) Nuclear factor-κB, p38, and stress-activated protein kinase mitogen-activated protein kinase signaling pathways regulate proinflammatory cytokines and apoptosis in human placental explants in response to oxidative stress. Am J Pathol 170:1511–1520. doi:10.2353/ajpath.2007.061035
Smith SC, Baker PN, Symonds EM (1997) Placental apoptosis in normal human pregnancy. Am J Obstet Gynecol 177:57–65. doi:10.1016/S0002-9378(97)70438-1
Halperin R, Peller S, Rotschild M et al (2000) Placental apoptosis in normal and abnormal pregnancies. Gynecol Obstet Investig 50:84–87
Jones KL, Brauman JN, Groome NP et al (2000) Activin A release into the circulation is an early event in systemic inflammation and precedes the release of follistatin. Endocrinology 141:1905–1908. doi:10.1210/en.141.5.1905
Jones KL, de Kretser DM, Patella S, Phillips DJ (2004) Activin A and follistatin in systemic inflammation. Mol Cell Endocrinol 225:119–125
Keelan JA, Zhou RL, Mitchell MD (2000) Activin a exerts both pro- and anti-inflammatory effects on human term gestational tissues. Placenta 21:38–43. doi:10.1053/plac.1999.0451
Keelan JA, Zhou RL, Evans LW, Groome NP, Mitchell MD (2000) Regulation of activin A, inhibin A, and follistatin production in human amnion and choriodecidual explants by inflammatory mediators. J Soc Gynecol Investig 7:291–296
Della Bella S, Giannelli S, Cozzi V et al (2011) Incomplete activation of peripheral blood dendritic cells during healthy human pregnancy. Clin Exp Immunol 164:180–192. doi:10.1111/j.1365-2249.2011.04330.x
Martin JA, Hamilton BE, Sutton PD et al (2003) Births: final data for 2002. Natl Vital Stat Rep 52:1–113
Rane SM, Guirgis RR, Higgins B, Nicolaides KH (2003) Pre-induction sonographic measurement of cervical length in prolonged pregnancy: the effect of parity in the prediction of the need for Cesarean section. Ultrasound Obstet Gynecol 22:45–48. doi:10.1002/uog.166
Bueno B, San-Frutos L, Pérez-Medina T et al (2007) The labor induction: integrated clinical and sonographic variables that predict the outcome. J Perinatol 27:4–8. doi:10.1038/sj.jp.7211619
Prado CA de C, Araujo Júnior E, Duarte G, et al (2016) Predicting success of labor induction in singleton term pregnancies by combining maternal and ultrasound variables. J Matern Fetal Neonatal Med 1–35. doi:10.3109/14767058.2015.1135124
Ben-Harush Y, Kessous R, Weintraub AY, Aricha-Tamir B, Steiner N, Spiegel E, Hershkovitz R (2015) The use of sonographic cervical length assessment for the prediction of time from induction to delivery. J Matern Fetal Neonatal Med 30:1–5
Acknowledgements
The authors thank the midwife team of Obstetric Unit of Siena, for contributing to collect data for this study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional research committee (Prot. 325/05 B) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Funghi, L., Torricelli, M., Novembri, R. et al. Placental and maternal serum activin A in spontaneous and induced labor in late-term pregnancy. J Endocrinol Invest 41, 171–177 (2018). https://doi.org/10.1007/s40618-017-0640-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-017-0640-z