Abstract
Purpose
Accumulating evidence indicates that intrauterine evolution disturbance can contribute to myocardial ischemia reperfusion (IR) injury; in addition, thyroid hormones (THs) have a crucial role in the development of different systems during fetal life. The aim of this study was to determine the effect of TH deficiency during fetal life on tolerance of isolated heart to ischemia during adulthood in both genders.
Methods
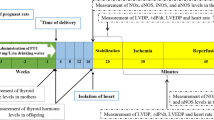
Hypothyroidism was induced in pregnant Wistar rats by administrating 0.025 % 6-propyl-2-thiouracil in drinking water throughout pregnancy. Offspring of rats with maternal hypothyroidism (MH) and control groups were tested in adulthood. Isolated hearts were perfused with Langendorff setup and exposed to 30 min of ischemia, followed by 45 min of reperfusion. Baseline values of the left ventricular end-diastolic pressure (LVEDP), left ventricular developed pressure (LVDP), heart rate (HR), and peak rates of positive and negative changes in left ventricular pressure (±dp/dt) were recorded.
Results
In the MH groups the baseline levels of LVDP (male: 23 %, female: 33 %), HR (male: 31 %, female: 26 %), and ±dp/dt were significantly (p < 0.01) lower, compared to controls. After ischemia, hearts from male rats with MH had less tolerance to IR injury as assessed in terms of reductions in recovery of hemodynamic parameters compared to controls, while in female rats there were no significant differences between MH and controls.
Conclusions
MH decreases hemodynamic parameters in the heart of both male and female offspring in adulthood; in addition, hearts of male rats with MH show less tolerance to ischemia, compared to those of females.





Similar content being viewed by others
References
Hausenloy DJ (2009) Signalling pathways in ischaemic postconditioning. Thromb Haemost 101:626–634
Hu S, Huang M, Li Z, Jia F, Ghosh Z, Lijkwan MA, Fasanaro P, Sun N, Wang X, Martelli F, Robbins RC, Wu JC (2010) MicroRNA-210 as a novel therapy for treatment of ischemic heart disease. Circulation 122:S124–S131
Thornburg KL (2011) Foetal programming reveals the dark side of AT(2)R. Cardiovasc Res 89:260–261
Sahay RK, Nagesh VS (2012) Hypothyroidism in pregnancy. Indian J Endocrinol Metab 16:364–370
Danzi S, Klein I (2004) Thyroid hormone and the cardiovascular system. Minerva Endocrinol 29:139–150
Klein I, Danzi S (2007) Thyroid disease and the heart. Circulation 116:1725–1735
LeBeau SO, Mandel SJ (2006) Thyroid disorders during pregnancy. Endocrinol Metab Clin North Am 35:117–136
Chizzonite RA, Zak R (1984) Regulation of myosin isoenzyme composition in fetal and neonatal rat ventricle by endogenous thyroid hormones. J Biol Chem 259:12628–12632
Goldenthal MJ, Weiss HR, Marin-Garcia J (2004) Bioenergetic remodeling of heart mitochondria by thyroid hormone. Mol Cell Biochem 265:97–106
Lee HL, Chen CL, Yeh ST, Zweier JL, Chen YR (2012) Biphasic modulation of the mitochondrial electron transport chain in myocardial ischemia and reperfusion. Am J Physiol Heart Circ Physiol 302:H1410–H1422
Farahani H, Ghasemi A, Roghani M, Zahediasl S (2013) Effect of neonatal hypothyroidism on carbohydrate metabolism, insulin secretion, and pancreatic islets morphology of adult male offspring in rats. J Endocrinol Invest 36:44–49
Zaman J, Jeddi S, Ghasemi A (2014) The effects of ischemic postconditioning on myocardial function and nitric oxide metabolites following ischemia-reperfusion in hyperthyroid rats. Korean J Physiol Pharmacol 18:481–487
Sugga GS, Khan MU, Khanam R (2012) Protective role of fibrates in cardiac ischemia/reperfusion. J Adv Pharm Technol Res 3:188–192
Fouron JC, Bourgin JH, Letarte J, Dussault JH, Ducharme G, Davignon A (1982) Cardiac dimensions and myocardial function of infants with congenital hypothyroidism. An echocardiographic study. Br Heart J 47:584–587
Crispi F, Hernandez-Andrade E, Pelsers MM, Plasencia W, Benavides-Serralde JA, Eixarch E, Le Noble F, Ahmed A, Glatz JF, Nicolaides KH, Gratacos E (2008) Cardiac dysfunction and cell damage across clinical stages of severity in growth-restricted fetuses. Am J Obstet Gynecol 199(254):e1–e8
Banovac K, Kekic M, Bzik L, Skreb F, Sekso M (1981) Reduced active thyroid hormone levels after delivery. J Endocrinol Invest 4:271–274
Parate V, Rode M, Pande S, Ansari T, Kamble P (2010) Thyroid function in mothers during the process of normal delivery. Int J Endocrinol Metab 8:39–45
Hapon MB, Motta AB, Ezquer M, Bonafede M, Jahn GA (2007) Hypothyroidism prolongs corpus luteum function in the pregnant rat. Reproduction 133:197–205
Santos SO, Loureiro SM, Alves IG, Jesus CS, Santos PR, Santos MR, Dias DP, Santana-Filho VJ, Badaue-Passos D Jr (2012) Experimental gestational hypothyroidism evokes hypertension in adult offspring rats. Auton Neurosci 170:36–41
Karbalaei N, Ghasemi A, Faraji F, Zahediasl S (2013) Comparison of the effect of maternal hypothyroidism on carbohydrate metabolism in young and aged male offspring in rats. Scand J Clin Lab Invest 73:87–94
Chen K, Carey LC, Valego NK, Liu J, Rose JC (2005) Thyroid hormone modulates renin and ANG II receptor expression in fetal sheep. Am J Physiol Regul Integr Comp Physiol 289:R1006–R1014
Wibo M, Feron O, Zheng L, Maleki M, Kolar F, Godfraind T (1998) Thyroid status and postnatal changes in subsarcolemmal distribution and isoform expression of rat cardiac dihydropyridine receptors. Cardiovasc Res 37:151–159
Thomas G, Chung M, Cohen CJ (1985) A dihydropyridine (Bay k 8644) that enhances calcium currents in guinea pig and calf myocardial cells. A new type of positive inotropic agent. Circ Res 56:87–96
Tie R, Ji L, Nan Y, Wang W, Liang X, Tian F, Xing W, Zhu M, Li R, Zhang H (2013) Achyranthes bidentata polypeptides reduces oxidative stress and exerts protective effects against myocardial ischemic/reperfusion injury in rats. Int J Mol Sci 14:19792–19804
Rohilla A, Khan MU, Khanam R (2012) Cardioprotective potential of simvastatin in the hyperhomocysteinemic rat heart. J Adv Pharm Technol Res 3:193–198
Pantos C, Malliopoulou V, Mourouzis I, Sfakianoudis K, Tzeis S, Doumba P, Xinaris C, Cokkinos AD, Carageorgiou H, Varonos DD, Cokkinos DV (2003) Propylthiouracil-induced hypothyroidism is associated with increased tolerance of the isolated rat heart to ischaemia-reperfusion. J Endocrinol 178:427–435
Mourouzis I, Dimopoulos A, Saranteas T, Tsinarakis N, Livadarou E, Spanou D, Kokkinos AD, Xinaris C, Pantos C, Cokkinos DV (2009) Ischemic preconditioning fails to confer additional protection against ischemia-reperfusion injury in the hypothyroid rat heart. Physiol Res 58:29–38
Bobadilla I, Franco M, Cruz D, Zamora J, Robles SG, Chavez E (2001) Hypothyroidism provides resistance to reperfusion injury following myocardium ischemia. Int J Biochem Cell Biol 33:499–506
Abe M, Obata H, Tanaka H (1992) Functional and metabolic responses to ischemia in the isolated perfused hypothyroid rat heart. Jpn Circ J 56:671–680
Perrelli MG, Pagliaro P, Penna C (2011) Ischemia/reperfusion injury and cardioprotective mechanisms: role of mitochondria and reactive oxygen species. World J Cardiol 3:186–200
Meehan J, Kennedy JM (1997) Influence of thyroid hormone on the tissue-specific expression of cytochrome c oxidase isoforms during cardiac development. Biochem J 327(Pt 1):155–160
Tanaka H, Sekine T, Terada M, Kobayashi Y, Shigenobu K (1997) Cardioprotective effect against ischemia-reperfusion injury of AHC-52, a dihydropyridine compound with inhibitory effect on Cl− but not Ca2+ current. Naunyn Schmiedebergs Arch Pharmacol 356:853–855
Huttemann M, Helling S, Sanderson TH, Sinkler C, Samavati L, Mahapatra G, Varughese A, Lu G, Liu J, Ramzan R, Vogt S, Grossman LI, Doan JW, Marcus K, Lee I (2012) Regulation of mitochondrial respiration and apoptosis through cell signaling: cytochrome c oxidase and cytochrome c in ischemia/reperfusion injury and inflammation. Biochim Biophys Acta 1817:598–609
Bae S, Zhang L (2005) Gender differences in cardioprotection against ischemia/reperfusion injury in adult rat hearts: focus on Akt and protein kinase C signaling. J Pharmacol Exp Ther 315:1125–1135
Fukumoto T, Yamashita N, Tawa M, Ohkita M, Matsumura Y (2012) Sex differences in postischemic cardiac dysfunction and norepinephrine overflow in rat heart: the role of estrogen against myocardial ischemia-reperfusion damage via an NO-mediated mechanism. J Cardiovasc Pharmacol 60:269–275
Zhai P, Eurell TE, Cotthaus R, Jeffery EH, Bahr JM, Gross DR (2000) Effect of estrogen on global myocardial ischemia-reperfusion injury in female rats. Am J Physiol Heart Circ Physiol 279:H2766–H2775
Xu Y, Arenas IA, Armstrong SJ, Plahta WC, Xu H, Davidge ST (2006) Estrogen improves cardiac recovery after ischemia/reperfusion by decreasing tumor necrosis factor-alpha. Cardiovasc Res 69:836–844
Dhote VV, Balaraman R (2007) Gender specific effect of progesterone on myocardial ischemia/reperfusion injury in rats. Life Sci 81:188–197
Deschamps AM, Murphy E, Sun J (2010) Estrogen receptor activation and cardioprotection in ischemia reperfusion injury. Trends Cardiovasc Med 20:73–78
Acknowledgments
This study was supported by a Grant No 542 from the Endocrine Physiology Research Center, Research Institute for Endocrine Sciences Shahid Beheshti University of Medical Sciences. We would like to thank Ms N. Shiva for critical editing of English grammar and syntax of the manuscript.
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
Animal handling was performed according to ethical standards approved by our local ethics committee.
Informed Consent
No Informed Consent.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghanbari, M., Jeddi, S., Bagheripuor, F. et al. The effect of maternal hypothyroidism on cardiac function and tolerance to ischemia–reperfusion injury in offspring male and female rats. J Endocrinol Invest 38, 915–922 (2015). https://doi.org/10.1007/s40618-015-0267-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-015-0267-x