Abstract
Background and Aims
Normal ranges of serum alanine aminotransferase (ALT) may vary by race. However, results from research studies are contradictory, and many of these studies have included only small numbers of African Americans. We investigated ALT values in patients without evidence of liver disease to determine whether normal ranges differ across race groups. We also evaluated whether a race- and sex-dependent upper limit of normal (ULN) would improve the ability of ALT to predict liver disease compared to the sex-dependent ULN currently in use.
Methods
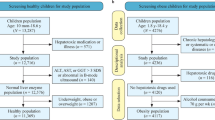
We identified ICD9 codes for liver conditions and diabetes in medical records from a sample of 6719 patients. Analysis of variance (ANOVA) was used to assess differences in ALT log-transformed distributions by race. Logistic regression was used to evaluate whether the addition of race to the current sex-dependent ULN improves the ability of ALT to predict liver disease (assessed by area under the receiver operating characteristic curve (AUROC)).
Results
Among 1200 patients with BMI 18.5 < 25 and no evidence of liver disease or type 2 diabetes in their medical record, African Americans demonstrated significantly lower ALT (23.47 IU/L; 95% CL 22.87–24.10) than a combined group of Asian American/White/Other patients (25.71 IU/L; 95% CL 24.69–26.77). This difference remained across BMI categories. The race- and sex-dependent model demonstrated significantly better predictive ability than the sex-dependent model (AUROC = 66.6% versus 59.6%, respectively; p < 0.0001).
Conclusions
In a large, racially diverse sample, African Americans demonstrated significantly lower ALT compared to non-African Americans; this difference remained as BMI increased. The establishment of race-specific normal ranges for ALT could contribute to better screening and care for African American patients.



Similar content being viewed by others
References
Kim WR, Flamm SL, di Bisceglie AM, Bodenheimer HC, Public Policy Committee of the American Association for the Study of Liver Disease. Serum activity of alanine aminotransferase (ALT) as an indicator of health and disease. Hepatology. 2008;47(4):1363–70.
Fraser A, Harris R, Sattar N, Ebrahim S, Davey Smith G, Lawlor DA. Alanine aminotransferase, gamma-glutamyltransferase, and incident diabetes: the British Women’s Heart and Health Study and meta-analysis. Diabetes Care. 2009;32(4):741–50.
Liu Z, Que S, Ning H, Wang L, Peng T. Elevated alanine aminotransferase is strongly associated with incident metabolic syndrome: a meta-analysis of prospective studies. PLoS One. 2013;8(12):e80596.
Lee TH, Kim WR, Benson JT, Therneau TM, Melton LJ III. Serum aminotransferase activity and mortality risk in a United States community. Hepatology. 2008;47(3):880–7.
Marshall W. Alanine Aminotransferase (serum, plasma). Association for Clinical Biochemistry and Laboratory Medicine: London, UK; 2011. https://www.acb.org.uk/resource/alanine-aminotransferase.html.
Gerbi GB, Rupp LB, Ko SC, Moorman AC, Holmberg SD, Xu F, et al. Reported reasons for testing among hepatitis B virus-infected patients - Chronic Hepatitis Cohort Study (CHeCS), United States, 2006–2010. Liver Int. 2014;34(6):e162-3.
Pacifico L, Ferraro F, Bonci E, Anania C, Romaggioli S, Chiesa C. Upper limit of normal for alanine aminotransferase: quo vadis? Clin Chim Acta. 2013;422:29–39.
Park SH, Heo NY, Kim CH, Suk KT, Kim DJ, Lee HY. Upper reference limits for aminotransferase activities and the prevalence of elevated aminotransferase activities in a Korean population. J Clin Gastroenterol. 2013;47(1):76–82.
Wu WC, Wu CY, Wang YJ, Hung HH, Yang HI, Kao WY, et al. Updated thresholds for serum alanine aminotransferase level in a large-scale population study composed of 34 346 subjects. Aliment Pharmacol Ther. 2012;36(6):560–8.
Ruhl CE, Everhart JE. Upper limits of normal for alanine aminotransferase activity in the United States population. Hepatology. 2012;55(2):447–54.
Kahn RA, et al. The distribution of serum alanine aminotransferase levels in a blood donor population. Am J Epidemiol. 1982;115(6):929–40.
Shulman IA, Saxena S, Nelson JM. The impact of alanine aminotransferase testing on Hispanic blood donations. Arch Pathol Lab Med. 1987;111(10):988–9.
Goldie DJ, McConnell AA. Serum alanine transaminase (ALT) reference ranges estimated from blood donors. J Clin Pathol. 1990;43(11):929–31.
Piton A, Poynard T, Imbert-Bismut F, Khalil L, Delattre J, Pelissier E, et al. Factors associated with serum alanine transaminase activity in healthy subjects: consequences for the definition of normal values, for selection of blood donors, and for patients with chronic hepatitis C. MULTIVIRC Group. Hepatology. 1998;27(5):1213–9.
Beutler E, West C. Hematologic differences between African-Americans and whites: the roles of iron deficiency and alpha-thalassemia on hemoglobin levels and mean corpuscular volume. Blood. 2005;106(2):740–5.
Lim E, Miyamura J, Chen JJ. Racial/ethnic-specific reference intervals for common laboratory tests: a comparison among Asians, Blacks, Hispanics, and White. Hawaii J Med Public Health. 2015;74(9):302–10.
Flores YN, Yee HF Jr, Leng M, Escarce JJ, Bastani R, Salmerón J, et al. Risk factors for chronic liver disease in Blacks, Mexican Americans, and Whites in the United States: results from NHANES IV, 1999-2004. Am J Gastroenterol. 2008;103(9):2231–8.
Carter-Pokras OD, et al. Alanine aminotransferase levels in Hispanics. Ethn Dis. 1993;3(2):176–80.
Stewart SH, Connors GJ. Ethnicity, alcohol drinking and changes in transaminase activity among heavy drinkers. J Natl Med Assoc. 2007;99(5):564–9.
WHO Consultation on Obesity. Obesity : preventing and managing the global epidemic : report of a WHO consultation. Geneva: World Health Organization; 2000. p. 9.
Dong MH, Bettencourt R, Barrett-Connor E, Loomba R. Alanine aminotransferase decreases with age: the Rancho Bernardo Study. PLoS One. 2010;5(12):e14254.
Saab S, et al. Hepatitis C in African Americans. Am J Gastroenterol. 2014;109(10):1576–84 quiz 1575, 1585.
Sattar N, Forrest E, Preiss D. Non-alcoholic fatty liver disease. BMJ. 2014;349:g4596.
Chalasani N, et al. The diagnosis and management of non-alcoholic fatty liver disease: practice guideline by the American Gastroenterological Association, American Association for the Study of Liver Diseases, and American College of Gastroenterology. Gastroenterology. 2012;142(7):1592–609.
Browning JD, Szczepaniak LS, Dobbins R, Nuremberg P, Horton JD, Cohen JC, et al. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology. 2004;40(6):1387–95.
Rich NE, Oji S, Mufti AR, Browning JD, Parikh ND, Odewole M, et al. Racial and ethnic disparities in nonalcoholic fatty liver disease prevalence, severity, and outcomes in the United States: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2018;16(2):198–210 e2.
Schneider AL, et al. Racial differences in nonalcoholic fatty liver disease in the U.S. population. Obesity (Silver Spring). 2014;22(1):292–9.
Funding
This analysis was funded by Henry Ford Health System (Detroit, MI).
Author information
Authors and Affiliations
Contributions
Study concept and design: AV, SJ, KB, VL, ML, SG.
Acquisition of data: AV, SJ, RK, KB, VL, SG.
Analysis and interpretation of data: ST, YZ, LL, ML, SG.
Drafting of the manuscript: ST, SG.
Critical revision of the manuscript for important intellectual content: AV, ST, YZ, SJ, RK, LL, KB, VL, ML, SG.
Statistical analysis: YZ, LL, ML.
Obtained funding: ML, SG.
Study supervision: SG.
Corresponding author
Ethics declarations
All protocols were reviewed and approved by the HFHS Institutional Review Board prior to study initiation. The requirement for written informed consent was waived due to the observational nature of the study and the use of de-identified data.
Conflict of Interest
Stuart C. Gordon receives grant/research support from AbbVie Inc., Conatus Pharmaceuticals, CymaBay Therapeutics, Gilead Pharmaceuticals, Intercept Pharmaceuticals, and Merck & Co. He serves as an ad hoc consultant/advisor for Abbvie Inc., CVS Caremark, Gilead Pharmaceuticals, Intercept Pharmaceuticals, and Merck & Co.
Kimberly Brown receives grant/research support from Novartis, Gilead Pharmaceuticals, Conatus Pharmaceuticals, and Allergan. She has served as an advisor for Merck & Co, Gilead Pharmaceuticals, and AbbVie Inc.
Sheri Trudeau, Yueren Zhou, and Mei Lu receive grant/research support from Gilead Pharmaceuticals.
Lois Lamerato receives research support from AstraZeneca, Pfizer inc, Merck & Co, Policy Analysis Inc., Analytica International, Xcenda, eMaxHealth, and Evidera.
Adarsh Varma, Syed-Mohammed Jafri, Richard Krajenta, and Veronica Luzzi have no potential conflicts of interest to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Varma, A., Trudeau, S., Zhou, Y. et al. African Americans Demonstrate Significantly Lower Serum Alanine Aminotransferase Compared to Non-African Americans. J. Racial and Ethnic Health Disparities 8, 1533–1538 (2021). https://doi.org/10.1007/s40615-020-00916-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40615-020-00916-2