Abstract
Background
Osteoarthritis is the most prevalent type of arthritis. Many approaches exist for characterising radiographic knee OA, including machine learning (ML).
Aims
To examine Kellgren and Lawrence (K&L) scores from ML and expert observation, minimum joint space and osteophyte in relation to pain and function.
Methods
Participants from the Hertfordshire Cohort Study, comprising individuals born in Hertfordshire from 1931 to 1939, were analysed. Radiographs were assessed by clinicians and ML (convolutional neural networks) for K&L scoring. Medial minimum joint space and osteophyte area were ascertained using the knee OA computer-aided diagnosis (KOACAD) program. The Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) was administered. Receiver operating characteristic analysis was implemented for minimum joint space, osteophyte, and observer- and ML-derived K&L scores in relation to pain (WOMAC pain score > 0) and impaired function (WOMAC function score > 0).
Results
359 participants (aged 71–80) were analysed. Among both sexes, discriminative capacity regarding pain and function was fairly high for observer-derived K&L scores [area under curve (AUC): 0.65 (95% CI 0.57, 0.72) to 0.70 (0.63, 0.77)]; results were similar among women for ML-derived K&L scores. Discriminative capacity was moderate among men for minimum joint space in relation to pain [0.60 (0.51, 0.67)] and function [0.62 (0.54, 0.69)]. AUC < 0.60 for other sex-specific associations.
Discussion
Observer-derived K&L scores had higher discriminative capacity regarding pain and function compared to minimum joint space and osteophyte. Among women, discriminative capacity was similar for observer- and ML-derived K&L scores.
Conclusion
ML as an adjunct to expert observation for K&L scoring may be beneficial due to the efficiency and objectivity of ML.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis (OA) is the most prevalent type of arthritis and is characterised by joint stiffness and pain, leading to functional decline [1]. The Global Burden of Disease 2017 Study found that OA accounted for 14.9 million incident cases, 303.1 million prevalent cases, and 9.6 million years lived with disability in 2017 [2]. The knee is the most common site of OA, with the prevalence of knee OA estimated at around 50% among those aged 75 years and older [3].
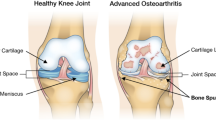
Knee OA can be characterised through use of radiography and clinical information relating to patient-reported symptoms and function [4]. However, previous studies have established discordance between the presence of radiographic and clinical knee OA [5,6,7] and much interest has focussed on how to characterise radiographic OA. One approach for deciding between different methods is to examine their predictive capacity regarding pain and degree of impaired function, two of the key clinical symptoms of OA [4]. It has been suggested that Kellgren and Lawrence (K&L), with its composite joint space, osteophytes, sclerosis and altered joint congruity, provides a better index than individual radiographic features alone for the prediction of knee pain [8, 9].
Supervised machine learning (ML), the process by which algorithms 'are taught' to recognise labelled data such that they can accurately predict future outcomes from new, unlabelled data, has been widely applied in medical research [10] and in the field of osteoarthritis [11]. There can be wide variation in the subjective assessment of knee radiographs with regard to the K&L grading of osteoarthritis severity [12] which could be avoided by applying ML techniques which also have the potential to improve efficiency by assisting radiologists and radiographers in their assessment of knee radiographs. This is important in the context of a widespread shortage of radiologists; in 2021, the consultant radiologist workforce shortfall stood at 29% (1669 whole-time equivalents) in the UK alone [13].
To our knowledge, no studies have compared how strongly individual radiographic features (minimum joint space and osteophyte), observer-derived K&L scores and ML-derived K&L scores are related to pain and function. Therefore, we explored this in a population-based cohort of community-dwelling older men and women from the United Kingdom.
Methods
The Hertfordshire Cohort Study
The Hertfordshire Cohort Study (HCS) comprises men and women born in Hertfordshire from 1931–1939 and who still lived there in 1998–2004 when they completed a clinic visit and home interview for a detailed characterisation of their health. The HCS and further details of the associated follow-up studies have been described in detail previously [14, 15].
Ascertainment of participant characteristics in 2011
Smoking status, alcohol consumption and average daily outdoor physical activity in minutes (Longitudinal Aging Study Amsterdam Physical Activity Questionnaire (LAPAQ) [16]) were ascertained at the home interview through nurse-administered questionnaires. The Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), a 24-item knee questionnaire with subscales measuring pain, stiffness and physical function [17], was also administered. Height was measured (wall-mounted SECA stadiometer) along with weight (calibrated SECA 770 digital floor scales, SECA Ltd, Hamburg) and used to derive BMI (kg/m2).
Anterior–posterior and lateral patellofemoral knee X-rays were taken of both knees at a local hospital after the 2011 home visit and joints were graded based on the (K&L) criteria [18]. This criteria is described as follows: Grade 1—possible osteophytes on the radiograph and unlikely narrowing of the joint space; Grade 2—small osteophytes and possible narrowing of the joint space; Grade 3—multiple, moderately sized osteophytes, definite joint space narrowing, some sclerotic areas and possible deformation of bone ends; Grade 4—multiple large osteophytes, severe joint space narrowing, marked sclerosis and definite bony end deformity [18].
Derivation of minimum joint space and osteophyte from radiographs
The automatic knee OA computer-aided diagnosis (KOACAD) program to quantify key OA parameters from digital knee radiographs has been described in detail previously [19]. In brief, filtering of the radiograph was performed to reduce image noise and to extract outlines of the tibia and femur for estimation of medial and lateral sides. Measurements of joint space area and minimum joint space were ascertained after determination of the region of interest. The medial and lateral tibial and femoral margins were then constructed using a horizontal neighbourhood difference filter and Canny’s filter in order to calculate inflection points for these margins. The medial tibial outline from the joint level to the inflection point was then drawn; osteophyte area was regarded as the area that was medially prominent over the extended outline.
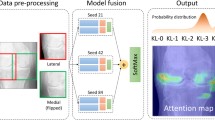
Derivation of K&L grades from machine learning
To develop the machine learning algorithm, data from the Osteoarthritis Initiative (OAI), a prospective observational study of 4796 individuals with, or at risk of, developing knee OA [20], were separated into a training dataset and a validation dataset. The final algorithm was then applied in the Hertfordshire Cohort Study.
To perform the ML, data from HCS participants, comprising knee radiographs and the corresponding K&L grade, were combined with similar data obtained from Mendeley Data [21]. The latter contained 2889 training and 828 testing radiographs from OAI participants.
To detect joints in radiographs, Faster R-CNN was used [22]. This consists of the following process: network filters are convolved with the radiograph to yield two-dimensional feature maps; regions of feature maps are then generated by sliding a window through them; finally, feature vectors corresponding to each region are extracted, from which the probability that the region contains the joint, along with the coordinates of the boundary of the region, are estimated. For the training process, the Mendeley training dataset was used. Radiographs were resized to 320 × 256 and those containing joint replacements were removed. The backbone network used was a ResNet-50 [23]. All network parameters were randomly initialised. The model was then fine-tuned for 10 epochs, using the Adam optimiser [24] with a starting learning rate of 5 × 10–5, which was decreased by a factor of 0.1 every 3 epochs. At the end of each epoch, the test data were assessed. Batch sizes of 5 and 1 were used for training and testing, respectively.
For predicting the K&L grade corresponding to each radiograph, ResNet-152, a type of Convolutional Neural Network [25], was used. All detected joints were cropped. The Mendeley dataset was already split into training (5778 knee joints) and testing (1656 knee joints) arms. Each model was run three times for three different seeds (0, 1, 2). Data augmentation was used to enlarge the training data where every image was horizontally flipped and rotated 30°. All models were trained for ten epochs and at the end of each epoch, the test data were assessed. The epoch with the highest level of accuracy was used. A stochastic gradient descent (SGD) optimiser [26] with momentum of 0.9 and with weight decay of 5 × 103 was implemented. Two different learning rate values were used, 1 × 10–3 for all networks except the classifier, which used a learning rate of 5 × 10–2. The learning rate was decreased by a factor of 0.1 every 2 epochs. The same batch sizes were used as in the detection process.
Ethical approval and informed consent
The baseline Hertfordshire Cohort Study had ethical approval from the Hertfordshire and Bedfordshire Local Research Ethics Committee and the follow-up had ethical approval from the East and North Hertfordshire Ethical Committees. Investigations were conducted in accordance with the principles expressed in the Declaration of Helsinki.
Statistical analysis
Analyses were performed at the person-level as WOMAC scores were only available for individual participants and not for each knee. As a result, the worse value from both knees (highest K&L score and osteophyte, and lowest minimum joint space) was used in analyses. Predictors in analyses were: low minimum joint space, defined as having values in the sex-specific lower third of the distribution (< 3.2 mm for men, < 2.8 mm for women); observer-derived and ML-derived K&L scores, categorised as 0/1, 2 and 3/4; and osteophyte, dichotomised as 0 mm2 and > 0 mm2. Outcomes in analyses were pain (WOMAC pain score > 0) and impaired function (WOMAC function score > 0).
Participant characteristics were described using summary statistics. Predictors in relation to outcomes were examined using chi-squared and Fisher’s exact tests. Logistic regression was used to perform receiver operating characteristic (ROC) analyses to calculate the area under curve (AUC) for each uncategorised predictor in relation to each outcome.
Men and women were analysed separately and P < 0.05 was regarded as statistically significant. Analyses were conducted using Stata, release 17.1.
Results
Determination of analysis sample from the Hertfordshire Cohort Study
The Hertfordshire Cohort Study (HCS) comprised 2997 participants at baseline (1998–2004). In 2004, of the 966 participants from East Hertfordshire who had a dual-energy X-ray absorptiometry (DXA) scan at the start of the study, 642 were recruited for a musculoskeletal follow-up study. In 2011, 591 were invited to participate in a further follow-up study; 443 agreed to participate. The analysis sample comprised 359/433 participants with data on at least one key predictor (minimum joint space, osteophyte and ML-derived K&L scores) and at least one outcome (WOMAC scores for pain and impaired function).
Participant characteristics of the analysis sample
The characteristics of the study population are presented in Table 1. Mean (SD) age at the 2011 follow-up was 75.5 (2.5) years. Mean (SD) minimum joint space was 3.6 (1.0) mm and 3.2 (1.0) mm among men and women, respectively; values for median (lower quartile, upper quartile) osteophyte were 0.8 (0.0, 6.9) mm2 among men and 2.1 (0.0, 8.3) mm2 among women. Overall, 53 (30.1%) men and 67 (36.6%) women had pain (WOMAC pain score > 0); 57 (34.1%) men and 67 (39.6%) women had impaired function (WOMAC function score > 0).
Minimum joint space, osteophyte and K&L scores in relation to pain and impaired function
The proportion with pain and impaired function according to each predictor (minimum joint space, osteophyte, observer-derived K&L score and ML-derived K&L score) is presented in Table 2. Among men, the proportion with impaired function was greater among those with low minimum joint space compared to those without (46.6% vs 26.3%, p = 0.009). Among both men and women, observer- and ML-derived K&L scores were associated with both pain and impaired function (p < 0.05 for all associations); higher proportions with pain and impaired function were observed among participants with higher K&L scores. Osteophyte was not related to pain or impaired function among men or women.
Receiver operating characteristic analysis for each predictor with pain and impaired function as outcomes
The AUCs for each predictor (minimum joint space, osteophyte, observer-derived K&L score and ML-derived K&L score) in relation to pain and impaired function as outcomes are presented in Table 3 and Fig. 1. Among men and women, discriminative capacity regarding pain and impaired function was fairly high for observer-derived K&L scores with AUCs ranging from 0.65 (95% CI 0.57, 0.72) to 0.70 (0.63, 0.77), depending on the outcome and whether the subsample comprised men or women; this was only the case among women for ML-derived K&L scores with AUCs of 0.63 (0.56, 0.70) and 0.68 (0.61, 0.75) for pain and impaired function, respectively. Discriminative capacity was moderate among men for minimum joint space in relation to pain [0.60 (0.51, 0.67)] and impaired function [0.62 (0.54, 0.69)]. All other sex-specific associations, including those for osteophyte, had AUCs of less than 0.60.
Receiver operating characteristic curves with pain (WOMAC pain score > 0) and impaired function (WOMAC function score > 0) as outcomes. K&L Kellgren & Lawrence, WOMAC Western Ontario and McMaster Universities Osteoarthritis Index. The worse value from both knees (highest K&L score and osteophyte area, and lowest minimum joint space width) was used in analyses
Discussion
In this study, K&L assessment by expert observer had higher discriminative capacity regarding WOMAC pain and function compared to minimum joint space and osteophyte, derived from the automatic KOACAD program. For example, AUCs (95% CI) for pain and impaired function ranged from 0.65 (95% CI 0.57, 0.72) to 0.70 (0.63, 0.77) for observer-derived K&L scores among both men and women. In contrast, AUCs for minimum joint space among men were 0.60 (0.51, 0.67) and 0.62 (0.54, 0.69) for pain and impaired function, respectively, with other associations for minimum joint space and osteophyte having AUCs of less than 0.60. To our knowledge, no studies have compared minimum joint space and osteophyte, assessed using the KOACAD system, against observer-defined K&L scores regarding their strength of association with WOMAC pain and function. However, a previous study examined clinical OA (knee pain plus crepitus) in relation to the severity of osteophytes, joint space narrowing and K&L scores (all assessed qualitatively from radiographs) among participants of the Framingham Osteoarthritis Study [9]. Similar to our findings, this study reported that efficiency ([sensitivity + specificity]/2) was highest for K&L scores, suggesting that these should be preferentially deployed in clinical practice.
Our study illustrates that automatic K&L scoring from radiographs can be performed using ML. This may offer advantages such as a reduction in the time required for K&L assessment, reducing the burden on the radiology workforce, and the avoidance of observer-dependent subjectivity. However, K&L assessment by ML did not perform as well as K&L assessment by expert observer in the prediction of the clinical variables of pain and function. Whilst AUCs for ML-derived K&L scores were similar to observer-derived scores among women for pain and impaired function, they were lower among men for pain [0.57 (0.50, 0.65) vs 0.68 (0.60, 0.74)] and impaired function [0.56 (0.48, 0.64) vs 0.70 (0.63, 0.77)]. However, these inconsistencies could be due to the fairly small sample of HCS participants used in the analysis.
Previous studies have used ML to assess knee OA severity by automatically estimating K&L scores from radiographs [27,28,29,30,31]. These studies applied convolutional neural networks to images from the OAI or Multicenter Osteoarthritis Study (MOST) and achieved a classification accuracy of 63% to 78% for uncategorised ML-derived K&L scores in relation to uncategorised observer-derived K&L scores. In our sample, the classification accuracy for this was lower at 50% with a Matthew’s correlation coefficient [32] of 0.27, perhaps due to the fairly small sample size. However, these other studies treated the observer-derived K&L scores as the gold standard even though assigning K&L scores is subjective, reflected in the high level of disagreement between radiographers [33,34,35]. In light of this, our study compared the ML- and observer-derived K&L scores by examining them each in relation to WOMAC pain and function.
We found that discriminative capacity was moderate among men for minimum joint space in relation to pain and impaired function (AUCs: 0.60–0.62) but weaker for osteophyte. In agreement with these findings, knee pain was more strongly associated with minimum joint space than osteophyte in an analysis of 1001 Japanese participants from the Research on Osteoarthritis Against Disability (ROAD) study which also used the KOACAD system: minimum joint space was associated with knee pain after adjustment for potential confounders but associations regarding osteophyte were not statistically significant [19]. However, an analysis of 2039 participants from the same cohort reported that minimum joint space was significantly associated with WOMAC pain, and osteophyte was significantly associated with impaired WOMAC function after adjustment for age and BMI [36]. Moreover, a longitudinal study comprising 1525 ROAD participants found that among men, osteophyte area was an independent predictor of WOMAC pain and impaired function at the 3-year follow-up but minimum joint space was not; among women, minimum joint space was an independent predictor of these outcomes but osteophyte area was not [37]. These differences in findings could be due to differences in adjustments used, whether studies were longitudinal or cross-sectional, or the fact that some studies analysed knees as individual units whereas others regarded the knee with the lowest minimum joint space as the designated knee for each participant.
Our study has some limitations. Firstly, a healthy participant effect is, unsurprisingly, evident in HCS [14] and sample attrition across the various follow-up waves could have resulted in further selection effects. However, the cohort has been shown to be broadly comparable with participants in the nationally representative Health Survey for England [14]. Furthermore, substantial bias would only have been introduced if associations of interest differed markedly between those who participated in comparison with those who were invited to participate but chose not to; this seems unlikely. Secondly, the sample size for this study was fairly small (n = 359). However, our main findings are biologically plausible and similar to those of previous studies. Thirdly, WOMAC scores were only available for individual participants and not for each knee; the worse value from both knees (highest K&L score and osteophyte, and lowest minimum joint space) was used in analyses. This may have led to an underestimation in the magnitude of the reported associations. Finally, machine learning was only performed with the ResNet family of architectures. However, this is a fairly stable architecture that has the advantage of skip connections between layers, which enables efficient gradient propagation during training. While there is a wide range of architectures in computer vision literature, some of which outperform ResNet in tasks considered in that field, we note that those architectures use millions of images for training and are not appropriate for our study. Furthermore, in a recent study by Matsoukas et al., comparing four different architectures on five different medical inference problems, ResNet achieved competitive performance [38]. Strengths of this study are that the HCS has been phenotyped according to strict protocols by highly-trained fieldworkers and managed by an experienced multi-disciplinary team.
In conclusion, observer-derived K&L scores had higher discriminative capacity for pain and function compared to minimum joint space and osteophyte, derived from the automatic KOACAD program. Among women, discriminative capacity was similar for observer- and ML-derived K&L scores. ML as an adjunct to expert observation in the classification of K&L scores may be beneficial due to the efficiency and objectivity of this method, though further work is required.
Availability of data and materials
Hertfordshire Cohort Study data are accessible via collaboration. Initial enquires should be made to EMD. Potential collaborators will be sent a collaborators’ pack and asked to submit a detailed study proposal to the HCS Steering Group.
References
Bruyère O, Honvo G, Veronese N et al (2019) An updated algorithm recommendation for the management of knee osteoarthritis from the European Society for Clinical and Economic Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases (ESCEO). Semin Arthritis Rheum 49:337–350
Safiri S, Kolahi A-A, Smith E et al (2020) Global, regional and national burden of osteoarthritis 1990–2017: a systematic analysis of the Global Burden of Disease Study 2017. Ann Rheum Dis 79:819–828
Litwic A, Edwards MH, Dennison EM et al (2013) Epidemiology and burden of osteoarthritis. Br Med Bull 105:185–199
Bernetti A, Agostini F, Alviti F et al (2021) New Viscoelastic Hydrogel Hymovis MO.RE. Single intra-articular injection for the treatment of knee osteoarthritis in sportsmen: safety and efficacy study results. Front Pharmacol 12:673988
Bedson J, Croft PR (2008) The discordance between clinical and radiographic knee osteoarthritis: a systematic search and summary of the literature. BMC Musculoskelet Disord 9:116
Skou ST, Thomsen H, Simonsen OH (2014) The value of routine radiography in patients with knee osteoarthritis consulting primary health care: a study of agreement. Eur J Gen Pract 20:10–16
Parsons C, Clynes M, Syddall H et al (2015) How well do radiographic, clinical and self-reported diagnoses of knee osteoarthritis agree? Findings from the Hertfordshire cohort study. Springerplus 4:177
Felson DT, Niu J, Guermazi A et al (2011) Defining radiographic incidence and progression of knee osteoarthritis: suggested modifications of the Kellgren and Lawrence scale. Ann Rheum Dis 70:1884–1886
Felson DT, McAlindon TE, Anderson JJ et al (1997) Defining radiographic osteoarthritis for the whole knee. Osteoarthr Cartil 5:241–250
Kokkotis C, Moustakidis S, Papageorgiou E et al (2020) Machine learning in knee osteoarthritis: a review. Osteoarthr Cartil Open 2:100069
Binvignat M, Pedoia V, Butte AJ et al (2022) Use of machine learning in osteoarthritis research: a systematic literature review. RMD Open 8:e001998
Schwartz AJ, Clarke HD, Spangehl MJ et al (2020) Can a convolutional neural network classify knee osteoarthritis on plain radiographs as accurately as fellowship-trained knee arthroplasty surgeons? J Arthroplast 35:2423–2428
The Royal College of Radiologists (2022) Clinical radiology census report 2021. London. https://www.rcr.ac.uk/sites/default/files/clinical_radiology_census_report_2021.pdf. Accessed: 2nd Dec 2022
Syddall H, Sayer AA, Dennison E et al (2005) Cohort profile: the Hertfordshire cohort study. Int J Epidemiol 34:1234–1242
Syddall HE, Simmonds SJ, Carter SA et al (2019) The Hertfordshire Cohort Study: an overview. F1000Research 8:82
Stel VS, Smit JH, Pluijm SM et al (2004) Comparison of the LASA Physical Activity Questionnaire with a 7-day diary and pedometer. J Clin Epidemiol 57:252–258
Bellamy N, Buchanan WW, Goldsmith CH et al (1988) Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J Rheumatol 15:1833–1840
Kellgren JH, Lawrence J (1957) Radiological assessment of osteo-arthrosis. Ann Rheum Dis 16:494–502
Oka H, Muraki S, Akune T et al (2008) Fully automatic quantification of knee osteoarthritis severity on plain radiographs. Osteoarthr Cartil 16:1300–1306
Peterfy CG, Schneider E, Nevitt M (2008) The osteoarthritis initiative: report on the design rationale for the magnetic resonance imaging protocol for the knee. Osteoarthr Cartil 16:1433–1441
Chen P (2018) Knee osteoarthritis severity grading dataset. Mendeley Data V1. https://doi.org/10.17632/56rmx5bjcr.1
Ren S, He K, Girshick R et al (2015) Faster R-CNN: towards real-time object detection with region proposal networks. Adv Neural Inf Process Syst 28:91–99
Ikechukwu AV, Murali S, Deepu R et al (2021) ResNet-50 vs VGG-19 vs training from scratch: a comparative analysis of the segmentation and classification of pneumonia from chest X-ray images. Global Trans Proc 2:375–381
Kingma DP, Ba JL (2015) Adam: a method for stochastic optimization. arXiv:1412.6980. https://arxiv.org/pdf/1412.6980.pdf. Accessed 15 May 2023
Ajit A, Acharya K, Samanta A (2020) A review of convolutional neural networks. In: International Conference on Emerging Trends in Information Technology and Engineering. https://www.mriquestions.com/uploads/3/4/5/7/34572113/icetite049pid6395729.pdf. Accessed 15 May 2023
Ruder S (2016) An overview of gradient descent optimization algorithms. arXiv:1609.04747. https://arxiv.org/pdf/1609.04747.pdf. Accessed 15 May 2023
Chen P, Gao L, Shi X et al (2019) Fully automatic knee osteoarthritis severity grading using deep neural networks with a novel ordinal loss. Comput Med Imaging Graph 75:84–92
Norman B, Pedoia V, Noworolski A et al (2019) Applying densely connected convolutional neural networks for staging osteoarthritis severity from plain radiographs. J Digit Imaging 32:471–477
Tiulpin A, Thevenot J, Rahtu E et al (2018) Automatic knee osteoarthritis diagnosis from plain radiographs: a deep learning-based approach. Sci Rep 8:1727
Thomas KA, Kidziński Ł, Halilaj E et al (2020) Automated classification of radiographic knee osteoarthritis severity using deep neural networks. Radiol Artif Intell 18:e190065
Antony J, McGuinness K, Moran K et al (2017) Automatic detection of knee joints and quantification of knee osteoarthritis severity using convolutional neural networks. arXiv:1703.09856. https://arxiv.org/pdf/1703.09856.pdf. Accessed 15 May 2023
Chicco D, Jurman G (2020) The advantages of the Matthews correlation coefficient (MCC) over F1 score and accuracy in binary classification evaluation. BMC Genom 21:6
Culvenor AG, Engen CN, Øiestad BE et al (2015) Defining the presence of radiographic knee osteoarthritis: a comparison between the Kellgren and Lawrence system and OARSI atlas criteria. Knee Surg Sports Traumatol Arthrosc 23:3532–3539
Gossec L, Jordan J, Mazzuca S et al (2008) Comparative evaluation of three semi-quantitative radiographic grading techniques for knee osteoarthritis in terms of validity and reproducibility in 1759 X-rays: report of the OARSI–OMERACT task force. Osteoarthr Cartil 16:742–748
Felson DT, Nevitt MC, Yang M et al (2008) A new approach yields high rates of radiographic progression in knee osteoarthritis. J Rheumatol 35:2047–2054
Muraki S, Oka H, Akune T et al (2011) Independent association of joint space narrowing and osteophyte formation at the knee with health-related quality of life in Japan: a cross-sectional study. Arthritis Rheum 63:3859–3864
Muraki S, Akune T, Nagata K et al (2015) Does osteophytosis at the knee predict health-related quality of life decline? A 3-year follow-up of the ROAD study. Clin Rheumatol 34:1589–1597
Matsoukas C, Haslum JF, Sorkhei M et al (2022) What makes transfer learning work for medical images: feature reuse & other factors. arXiv:2203.01825. https://arxiv.org/pdf/2203.01825.pdf. Accessed 15 May 2023
Funding
The Hertfordshire Cohort Study was supported by the Medical Research Council University Unit Partnership grant number MRC_MC_UP_A620_1014. CC, EMD and LDW are supported by the UK Medical Research Council [MC_PC_21003; MC_PC_21001]. The funders had no role in the study design, collection, analysis and interpretation of data; in the writing of the manuscript; or in the decision to submit the manuscript for publication. For the purpose of open access, the author has applied a Creative Commons attribution license (CC BY) to any Author Accepted Manuscript version arising from this submission.
Author information
Authors and Affiliations
Contributions
LDW conducted the statistical analysis and produced the first draft of the manuscript; NRF designed the study, contributed to the interpretation of data and the analysis strategy; DP conducted the machine learning; HO, NY, and NO analysed the radiographs using the KOACAD program and provided these data; SM and MN devised the strategy for the machine learning analysis; EMD contributed to the interpretation of data; and CC designed the study. All authors made substantial contributions to the drafting of the manuscript and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
CC reports personal fees (outside the submitted work) from Amgen, Danone, Eli Lilly, GSK, Kyowa Kirin, Medtronic, Merck, Nestle, Novartis, Pfizer, Roche, Servier, Shire, Takeda and UCB. EMD reports personal fees and honoraria (outside the submitted work) from UCB, Pfizer, Lilly and Viatris. NRF has received travel bursaries from Pfizer and Eli Lilly. The remaining authors declare that they have no conflicts of interest.
Ethics approval
The procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964, as revised in 2000. The baseline Hertfordshire Cohort Study had ethical approval from the Hertfordshire and Bedfordshire Local Research Ethics Committee and the follow-up had ethical approval from the East and North Hertfordshire Ethical Committees.
Informed consent
All participants provided written informed consent to participate in the study and for their health records to be accessed in the future.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Westbury, L.D., Fuggle, N.R., Pereira, D. et al. Machine learning as an adjunct to expert observation in classification of radiographic knee osteoarthritis: findings from the Hertfordshire Cohort Study. Aging Clin Exp Res 35, 1449–1457 (2023). https://doi.org/10.1007/s40520-023-02428-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-023-02428-5