Abstract
Background
The cardiovascular benefits of resveratrol (RSV) have been well established by previous experimental and clinical studies. The aim of this study was to investigate the effectiveness of RSV administration on the impaired endothelial function induced by methylglyoxal (MGO), and to elucidate the role of endothelial nitric oxide synthase (eNOS) on its protective effect.
Methods
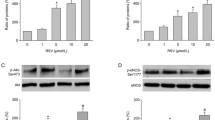
Aged Wistar rats (80 weeks old, n = 15) were used in this study. The thoracic aorta was isolated and cut into rings for organ culture. Aortic segments of rats were incubated with MGO (420 µM) in the presence or absence of RSV (30 µM) for 4 h (short-term) or 24 h (long-term). Isometric tension studies were performed by an isolated organ bath in response to acetylcholine (ACh, an endothelium-dependent vasodilator) and sodium nitroprusside (SNP, an endothelium-independent vasodilator). Beside, expressions of eNOS and phospho-eNOS (p-eNOS) (Ser 1177) in thoracic aorta rings were evaluated by immunohistochemistry.
Results
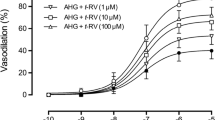
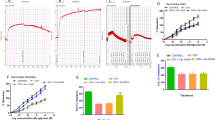
Both short-term and long-term MGO incubation significantly inhibited the relaxation response induced by ACh, while the relaxation to SNP was not significantly altered. In addition, eNOS and p-eNOS expressions decreased significantly in arteries incubated with MGO. The impaired endothelial reactivity as well as decreased expressions of eNOS and p-eNOS in MGO-incubated vessels were significantly improved by RSV treatment.
Conclusions
Endothelium-dependent vasodilatation of the thoracic aorta was significantly inhibited by MGO administration, and RSV may improve vascular endothelial function. The protective effect of RSV against MGO-induced endothelial dysfunction seems to be via increased eNOS expression and activity.






Similar content being viewed by others
References
Boyle JP, Thompson TJ, Gregg EW, et al (2010) Projection of the year 2050 burden of diabetes in the US adult population: dynamic modeling of incidence, mortality, and prediabetes prevalence. Popul Health Metr 8:29
Narayan KM, Boyle JP, Geiss LS, et al (2006) Impact of recent increase in incidence on future diabetes burden: U.S., 2005–2050. Diabetes Care 29:2114–2116
Gu K, Cowie CC, Harris MI (1999) Diabetes and decline in heart disease mortality in US adults. JAMA 281:1291–1297
American Diabetes A (2017) cardiovascular disease and risk management. Diabetes Care 40:S75-S87
Halter JB, Musi N, Horne FMF, et al (2014) Diabetes and cardiovascular disease in older adults: current status and future directions. Diabetes 63:2578–2589
Cigolle CT, Blaum CS, Halter JB (2009) Diabetes and cardiovascular disease prevention in older adults. Clin Geriatr Med 25:607–641
Kirkman MS, Briscoe VJ, Clark N, et al (2012) Diabetes in older adults. Diabetes Care 35:2650–2664
Gregg EW, Li Y, Wang J (2014) Changes in diabetes-related complications in the United States, 1990–2010. N Engl J Med 370:1514–1523
Potenza MA, Gagliardi S, Nacci C, et al (2009) Endothelial dysfunction in diabetes: from mechanisms to therapeutic targets. Curr Med Chem 16:94–112
Barthelmes J, Nagele MP, Ludovici V, et al (2017) Endothelial dysfunction in cardiovascular disease and Flammer syndrome-similarities and differences. EPMA J 8:99–109
Versari D, Daghini E, Virdis A, et al (2009) The ageing endothelium, cardiovascular risk and disease in man. Exp Physiol 94:317–321
Yim HS, Kang SO, Hah YC, et al (1995) Free radicals generated during the glycation reaction of amino acids by methylglyoxal. A model study of protein-cross-linked free radicals. J Biol Chem 270:28228–28233
Lo CY, Li S, Tan D, et al (2006) Trapping reactions of reactive carbonyl species with tea polyphenols in simulated physiological conditions. Mol Nutr Food Res 50:1118–1128
Wu L (2005) The pro-oxidant role of methylglyoxal in mesenteric artery smooth muscle cells. Can J Physiol Pharmacol 83:63–68
Lapolla A, Flamini R, Dalla Vedova A, et al (2003) Glyoxal and methylglyoxal levels in diabetic patients: quantitative determination by a new GC/MS method. Clin Chem Lab Med 41:1166–1173
Hanssen NMJ, Scheijen J, Jorsal A (2017) Higher plasma methylglyoxal levels are associated with incident cardiovascular disease in individuals with type 1 diabetes: a 12-year follow-up study. Diabetes 66:2278–2283
Kong X, Ma MZ, Huang K, et al (2014) Increased plasma levels of the methylglyoxal in patients with newly diagnosed type 2 diabetes 2. J Diabetes 6:535–540
Srikanth V, Westcott B, Forbes J, et al (2013) Methylglyoxal, cognitive function and cerebral atrophy in older people. J Gerontol A Biol Sci Med Sci 68:68–73
Hallam KM, Li Q, Ananthakrishnan R (2010) Aldose reductase and AGE-RAGE pathways: central roles in the pathogenesis of vascular dysfunction in aging rats. Aging Cell 9:776–784
Brouwers O, Niessen PM, Haenen G, et al (2010) Hyperglycaemia-induced impairment of endothelium-dependent vasorelaxation in rat mesenteric arteries is mediated by intracellular methylglyoxal levels in a pathway dependent on oxidative stress. Diabetologia 53:989–1000
Dhar A, Dhar I, Desai KM, et al (2010) Methylglyoxal scavengers attenuate endothelial dysfunction induced by methylglyoxal and high concentrations of glucose. Br J Pharmacol 161:1843–1856
Do MH, Kim SY (2017) Hypericin, a naphthodianthrone derivative, prevents methylglyoxal-induced human endothelial cell dysfunction. Biomol Ther (Seoul) 25:158–164
Mukohda M, Morita T, Okada M, et al (2013) Long-term methylglyoxal treatment causes endothelial dysfunction of rat isolated mesenteric artery. J Vet Med Sci 75:151–157
Turkseven S, Ertuna E, Yetik-Anacak G, et al (2014) Methylglyoxal causes endothelial dysfunction: the role of endothelial nitric oxide synthase and AMP-activated protein kinase alpha. J Basic Clin Physiol Pharmacol 25:109–115
Sena CM, Matafome P, Crisostomo J, et al (2012) Methylglyoxal promotes oxidative stress and endothelial dysfunction. Pharmacol Res 65:497–506
Wu JM, Wang ZR, Hsieh TC, et al (2001) Mechanism of cardioprotection by resveratrol, a phenolic antioxidant present in red wine. Int J Mol Med 8:3–17
Das DK, Maulik N (2006) Resveratrol in cardioprotection: a therapeutic promise of alternative medicine. Mol Interv 6:36–47
Di Pascoli M, Divi M, Rodriguez-Vilarrupla A, et al (2013) Resveratrol improves intrahepatic endothelial dysfunction and reduces hepatic fibrosis and portal pressure in cirrhotic rats. J Hepatol 58:904–910
Takahashi S, Nakashima Y (2012) Repeated and long-term treatment with physiological concentrations of resveratrol promotes NO production in vascular endothelial cells. Br J Nutr 107:774–780
Yu HP, Hwang TL, Hwang TL, et al (2010) Resveratrol prevents endothelial dysfunction and aortic superoxide production after trauma hemorrhage through estrogen receptor-dependent hemeoxygenase-1 pathway. Crit Care Med 38:1147–1154
Xia N, Forstermann U, Li H (2017) Effects of resveratrol on eNOS in the endothelium and the perivascular adipose tissue. Ann NY Acad Sci 1403:132–141
Ugurel SS, Kuscu N, Ozenci CC, et al (2016) Resveratrol prevented lipopolysaccharide-induced endothelial dysfunction in rat thoracic aorta through increased eNOS expression. Balkan Med J 33:138–143
Li JY, Huang WQ, Tu RH, et al (2017) Resveratrol rescues hyperglycemia-induced endothelial dysfunction via activation of Akt. Acta Pharmacol Sin 38:182–191
Murat N, Korhan P, Kizer O, et al (2016) Resveratrol protects and restores endothelium-dependent relaxation in hypercholesterolemic rabbit corpus cavernosum. J Sex Med 13:12–21
Randell EW, Vasdev S, Gill V (2005) Measurement of methylglyoxal in rat tissues by electrospray ionization mass spectrometry and liquid chromatography. J Pharmacol Toxicol Methods 51:153–157
Figarola JL, Singhal J, Rahbar S, et al (2014) LR-90 Prevents methylglyoxal-induced oxidative stress and apoptosis in human endothelial cells. Apoptosis 19:776–788
Fleming I (2010) Molecular mechanisms underlying the activation of eNOS. Pflugers Arch 459:793–806
Fulton D, Gratton JP, McCabe TJ, et al (1999) Regulation of endothelium-derived nitric oxide production by the protein kinase Akt. Nature 399:597–601
Wang S, Wang J, Zhao A, et al (2017) SIRT1 Activation inhibits hyperglycemia-induced apoptosis by reducing oxidative stress and mitochondrial dysfunction in human endothelial cells. Mol Med Rep 16:3331–3338
Chen F, Qian LH, Deng B, et al (2013) Resveratrol protects vascular endothelial cells from high glucose-induced apoptosis through inhibition of NADPH oxidase activation-driven oxidative stress. CNS Neurosci Ther 19:675–681
Dimmeler S, Zeiher AM (1999) Nitric oxide-an endothelial cell survival factor. Cell Death Differ 6:964–968
Hoffmann J, Haendeler J, Aicher A, et al (2001) Aging enhances the sensitivity of endothelial cells toward apoptotic stimuli: important role of nitric oxide. Circ Res 89:709–715
Seo K, Seo S, Han JY, et al (2014) Resveratrol attenuates methylglyoxal-induced mitochondrial dysfunction and apoptosis by Sestrin2 induction. Toxicol Appl Pharmacol 280:314–322
Klinge CM, Wickramasinghe NS, Ivanova MM, et al (2008) Resveratrol stimulates nitric oxide production by increasing estrogen receptor alpha-Src-caveolin-1 interaction and phosphorylation in human umbilical vein endothelial cells. FASEB J 22:2185–2197
Zhang H, Zhang J, Ungvari Z, et al (2009) Resveratrol improves endothelial function: role of TNF{alpha} and vascular oxidative stress. Arterioscler Thromb Vasc Biol 29:1164–1171
Acknowledgements
This study was partially supported by Akdeniz University Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Statement of human and animal rights
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. The study protocol was approved by the Institutional Animal Ethics Committee of the University.
Informed consent
For this type of study informed consent is not required.
Rights and permissions
About this article
Cite this article
Tasatargil, A., Tanriover, G., Barutcigil, A. et al. Protective effect of resveratrol on methylglyoxal-induced endothelial dysfunction in aged rats. Aging Clin Exp Res 31, 331–338 (2019). https://doi.org/10.1007/s40520-018-0986-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-018-0986-x