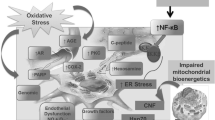
Abstract
A long history of diabetes mellitus and increasing age are associated with the onset of diabetic neuropathy, a painful and highly disabling complication with a prevalence peaking at 50% among elderly diabetic patients. Acetyl-l-carnitine (ALC) is a molecule derived from the acetylation of carnitine in the mitochondria that has an essential role in energy production. It has recently been proposed as a therapy to improve the symptoms of diabetic neuropathy. ALC is widely distributed in mammalian tissues, including the brain, blood–brain barrier, brain neurons, and astrocytes. Aside from its metabolic activity, ALC has demonstrated cytoprotective, antioxidant, and antiapoptotic effects in the nervous system. It exerts an analgesic action by reducing the concentration of glutamate in the synapses. It facilitates nerve regeneration and damage repair after primary trauma: its positive effects on metabolism promote the synthesis, fluidity, and functionality of neuronal membranes, increase protein synthesis, and improve the axonal transport of neurofilament proteins and tubulin. It also amplifies nerve growth factor responsiveness, an effect that is believed to enhance overall neurite growth. ALC has been proposed for the treatment of various neurological and psychiatric diseases, such as mood disorders and depression, dementias, Alzheimer’s disease, and Parkinson’s disease, because synaptic energy states and mitochondrial dysfunction are core factors in their pathogenesis.

Similar content being viewed by others
References
Bansal D, Gudala K, Muthyala H, Prasad Esam H, Nayakallu R, Bhansali A (2014) Prevalence and risk factors of development of peripheral diabetic neuropathy in type 2 diabetes mellitus in a tertiary care setting. J Diabetes Invest 5:714–721. doi:10.1111/jdi.12223
Evans J, Jacobs T, Evans E (2008) Role of acetyl-l-carnitine in the treatment of diabetic peripheral neuropathy. Ann Pharmacother 42:1686–1691. doi:10.1345/aph1L201
Callaghan B, Cheng H, Stables C, Smith A, Feldman E (2012) Diabetic neuropathy: clinical manifestations and current treatments. Lancet Neurol 11:521–534
Abbott CA, Malik RA, van Ross E, Kulkarni J, Boulton A (2011) Prevalence and characteristics of painful diabetic neuropathy in a large community-based diabetic population in the UK. Diabetes Care 34:2220–2224. doi:10.2337/dc11-1108
Vincent AM, Hayes JM, McLean LL, Vivekanandan-Giri A, Pennathur S, Feldman EL (2009) Dyslipidemia-induced neuropathy in mice. Diabetes 58:2376–2385. doi:10.2337/db09-0047
Padilla A, Descorbeth M, Almeyda AL, Payne K, De Leon M (2011) Hyperglycemia magnifies Schwann cell dysfunction and cell death triggered by PA-induced lipotoxicity. Brain Res 25:64–79. doi:10.1016/j.brainres.2010.11.013
McCall KD, Holliday D, Dickerson E, Wallace B, Schwartz AL, Schwartz C, Lewis CJ, Kohn LD, Schwartz FL (2010) Phenylmethimazole blocks palmitate-mediated induction of inflammatory cytokine pathways in 3T3L1 adipocytes and RAW 264.7 macrophages. J Endocrinol 207:343–353. doi:10.1677/JOE-09-0370
Chiechio S, Caricasole A, Barletta E, Storto M, Catania M, Copani A, Vertechy M, Nicolai R, Calvani M, Melchiorri D, Nicoletti F (2002) l-acetylcarnitine induces analgesia by selectively up-regulating mglu2 metabotropic glutamate receptors. Mol Pharmacol 61:989–996
Chiechio S, Copani A, Nicoletti F, Gereau RW 4th (2006) l-acetylcarnitine: a proposed therapeutic agent for painful peripheral neuropathies. Curr Neuropharmacol 4:233–237
Januszewicz E, Pajak B, Gajkowska B, Samluk L, Djavadian R, Hinton B, Nalecz K (2009) Organic cation/carnitine transporter OCTN3 is present in astrocytes and is up-regulated by peroxisome proliferators-activator receptor agonist. Int J Biochem Cell Biol 41:2599–2609. doi:10.1016/j.biocel.2009.08.020
De Grandis D (2007) Acetyl-l-carnitine for the treatment of chemotherapy-induced peripheral neuropathy. CNS Drugs 21(Suppl. 1):39–43
Chiechio S, Copani A, Zammataro M, Battaglia G, Gereau RW 4th, Nicoletti F (2010) Transcriptional regulation of type-2 metabotropic glutamate receptors: an epigenetic path to novel treatments for chronic pain. Trends Pharmacol Sci 31:153–160. doi:10.1016/j.tips.2009.12.003
De Grandis D, Minardi C (2002) Acetyl-l-carnitine (levacecarnine) in the treatment of diabetic neuropathy. A long-term, randomised, double-blind, placebo-controlled study. Drugs R D 3:223–231
De Grandis D, Santoro L, Di Benedetto P (1995) l-acetylcarnitine in the treatment of patients with peripheral neuropathies: a short-term, double-blind clinical study of 426 patients. Clin Drug Investig 10:317–322. doi:10.2165/00044011-199510060-00001
Sima AA, Calvani M, Mehra M, Amato A (2005) Acetyl-l-carnitine improves pain, nerve regeneration, and vibratory perception in patients with chronic diabetic neuropathy: an analysis of two randomized placebo-controlled trials. Diabetes Care 28:89–94
Li S, Li Q, Li Y, Li L, Tian H, Sun X (2015) Acetyl-l-carnitine in the treatment of peripheral neuropathic pain: a systematic review and meta-analysis of randomized controlled trials. PLoS One 10:e0119479. doi:10.1371/journal.pone.0119479
Veronese N, Sergi G, Stubbs B, The EUGMS special interest group on diabetes, Bourdel-Marchasson I, Tessier D, Sieber C, Strandberg T, Gillain S, Barbagallo M, Crepaldi G, Maggi S, Manzato E (2017) Effect of acetyl-l-carnitine in the treatment of diabetic peripheral neuropathy: a systematic review and meta-analysis. Eur Geriatr Med. doi:10.1016/j.eurger.2017.01.002
Wilson AD, Hart A, Wiberg M, Terenghi G (2010) Acetyl-l-carnitine increases nerve regeneration and target organ reinnervation—a morphological study. J Plast Reconstr Aesthet Surg 63:1186–1195. doi:10.1016/j.bjps.2009.05.039
Pisano C, Pratesi G, Laccabue D, Zunino F, Lo Giudice P, Bellucci A, Pacifici L, Camerini B, Vesci L, Castorina M, Cicuzza S, Tredici G, Marmiroli P, Nicolini G, Galbiati S, Calvani M, Carminati P, Cavaletti G (2003) Paclitaxel and cisplatin-induced neurotoxicity: a protective role of acetyl-l-carnitine. Clin Cancer Res 9:5756–5767
Madiraju P, Pande SV, Prentki M, Madiraju SR (2009) Mitochondrial acetylcarnitine provides acetyl groups for nuclear histone acetylation. Epigenetics 4:399–403
Ferrari F, Gorini A, Villa RF (2015) Functional proteomics of synaptic plasma membrane ATP-ases of rat hippocampus: effect of l-acetylcarnitine and relationships with dementia and depression pathophysiology. Eur J Pharmacol 756:67–74. doi:10.1016/j.ejphar.2015.03.011
Rossini M, Di Munno O, Valentini G, Bianchi G, Biasi G, Cacace E, Malesci D, La Montagna G, Viapiana O, Adami S (2007) Double-blind, multicenter trial comparing acetyl l-carnitine with placebo in the treatment of fibromyalgia patients. Clin Exp Rheumatol 25:182–188
Malaguarnera M (2013) Acetyl-l-carnitine in hepatic encephalopathy. Metab Brain Dis 28:193–199. doi:10.1007/s11011-013-9376-4
Montgomery SA, Thal LJ, Amrein R (2003) Meta-analysis of double blind randomized controlled clinical trials of acetyl-l-carnitine versus placebo in the treatment of mild cognitive impairment and mild Alzheimer’s disease. Int Clin Psychopharmacol 18:61–71
Watson JC, Dyck PJ (2015) Peripheral neuropathy: a practical approach to diagnosis and symptom management. Mayo Clin Proc 90:940–951
Traina G, Federighi G, Macchi M, Bernardi R, Durante M, Brunelli M (2011) Modulation of myelin basic protein gene expression by acetyl-l-carnitine. Mol Neurobiol 44:1–6
Feldman EL, Nave KA, Jensen TS, Bennett DLH (2017) New horizons in diabetic neuropathy: mechanisms, bioenergetics, and pain. Neuron 93:1296–1313
Sun Y, Shu Y, Liu B, Liu P, Wu C, Zheng R, Zhang X, Zhuang Z, Deng Y, Zheng L, Xu Q, Jiang B, Ouyang X, Gao J, Xu N, Li X, Jiang S, Liang C, Yao Y (2016) A prospective study to evaluate the efficacy and safety of oral acetyl-l-carnitine for the treatment of chemotherapy-induced peripheral neuropathy. Experimental and therapeutic. Medicine (Baltimore) 12:4017–4024
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Sergi, G., Pizzato, S., Piovesan, F. et al. Effects of acetyl-l-carnitine in diabetic neuropathy and other geriatric disorders. Aging Clin Exp Res 30, 133–138 (2018). https://doi.org/10.1007/s40520-017-0770-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40520-017-0770-3