Abstract
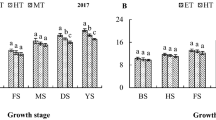
Water availability in plant organs is important for survival and yield production but has been underestimated during biomass allocation to organs. Assessment of fresh biomass and moisture content in root, culm and leaf during water deficit stress (WDS) can improve grain yield prediction in upland rice. Four upland rice cultivars, NERICAs 1, 4, 6 and Moroberekan, were subjected to 10-day WDS at reproductive stage. Fresh and dry biomass to organs were taken at the end of stress. Leaf relative water content (LRWC), drought-tolerant degree, soil moisture content, were significantly (p < 0.01) reduced under WDS. Actual fresh, dry and moisture and their fraction to organs followed a similar trend with culm having the largest weight (14.5 g) and fraction (0.49 g g−1). Fresh and moisture fractions to root were higher than that of leaf and vice-versa for dry fractions under a stressed condition. Moroberekan and NERICA 4 maintained a higher fraction of biomasses and moisture to root (0.40 and 0.30, respectively) than leaf and higher LRWC and percentage fertile spikelets at the end of 10-day stress. Actual biomass captured higher variation in yield than fraction of biomass to organs, except for dry biomass. The variation in yield of rice captured are in decreasing order of moisture content (54.82/52.61) > fresh biomass (41.60/34.84) > dry biomass (0.00/22.61) for actual and fraction of biomass to organs, respectively with the leaf accounting for the largest share of this variation. Fresh biomass rather than dry biomass of plant organs can predict grain yield better in upland rice during WDS.





Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Atayese, M. O., Olagunju, S. O., Sakariyawo, O. S., Oyekanmi, A. A., Babalola, O. A., Aderibigbe, S. G., Okonji, C. J., Olayiwola, M. O., Soremi, P. A., & Okeleye, K. A. (2012). Root response of some selected rice varieties to soil moisture stress at different phenological stages. Journal of Agricultural Science and Environment, 12(2), 96–113.
Búrquez, A. (1987). Leaf thickness and water deficit in plants: A tool for field studies. Journal of Experimental Botany, 38(1), 109–114. https://doi.org/10.1093/jxb/38.1.109
Dhanda, S. S., Sethi, G. S., & Behl, R. K. (2004). Indices of drought tolerance in wheat genotypes at early stages of plant growth. Journal of Agronomy and Crop Science, 190, 6–12. https://doi.org/10.1111/j.1439-037X.2004.00592.x
Dolezal, J., Jandova, V., Macek, M., & Liancourt, P. (2021). Contrasting biomass allocation responses across ontogeny and stress gradients reveal plant adaptations to drought and cold. Functional Ecology, 35, 32–42. https://doi.org/10.1111/1365-2435.13687
Fan, Z.-X., Sterck, F., Zhang, S.-B., Fu, P.-L., & Hao, G.-Y. (2017). Tradeoff between stem hydraulic efficiency and mechanical strength affects leaf–stem allometry in 28 Ficus tree species. Frontiers in Plant Science, 8, 1–10. https://doi.org/10.3389/fpls.2017.01619
Freeman, K. W., Raun, W. R., Johnson, G. V., Mullen, R. W., Stone, M. L., & Solie, J. B. (2003). Late-season prediction of wheat grain yield and grain protein late-season prediction of wheat grain yield. Communications in Soil Science and Plant Analysis, 34(13–14), 1837–1852. https://doi.org/10.1081/CSS-120023219
Gowda, V. R. P., Henry, A., Yamauchi, A., Shashidhar, H. E., & Serraj, R. (2011). Root biology and genetic improvement for drought avoidance in rice. Field Crops Research, 122(1), 1–13. https://doi.org/10.1016/j.fcr.2011.03.001
Guo, Y., Ma, Y., Zhan, Z., Li, B., Dingkuhn, M., Luquet, D., & De Reffye, P. (2006). Parameter optimization and field validation of the functional—Structural model GREENLAB for maize. Annals of Botany, 97, 217–230. https://doi.org/10.1093/aob/mcj033
Haffani, S., Mezni, M., Slama, I., Ksontini, M., & Chaïbi, W. (2014). Plant growth, water relations and proline content of three vetch species under water-limited conditions. Grass and Forage Science, 69(2), 323–333. https://doi.org/10.1111/gfs.12034
Herrera, J. (2009). Visibility vs. biomass in flowers: Exploring corolla allocation in Mediterranean entomophilous plants. Annals of Botany, 103(7), 1119–1127. https://doi.org/10.1093/aob/mcp046
Huang, W., Ratkowsky, D. A., Hui, C., Wang, P., Su, J., & Shi, P. (2019). Leaf fresh weight versus dry weight: Which is better for describing the scaling relationship between leaf biomass and leaf area for broad-leaved plants ? Forests, 10, 256. https://doi.org/10.3390/f10030256
Ilyas, M., Nisar, M., Khan, N., Hazrat, A., Khan, A. H., Hayat, K., Fahad, S., Khan, A., & Ullah, A. (2021). Drought tolerance strategies in plants: A mechanistic approach. Journal of Plant Growth Regulation, 40, 926–944. https://doi.org/10.1007/s00344-020-10174-5
Kage, H., Kochler, M., & Stützel, H. (2004). Root growth and dry matter partitioning of cauliflower under drought stress conditions: Measurement and simulation. European Journal of Agronomy, 20, 379–394. https://doi.org/10.1016/S1161-0301(03)00061-3
Kameoka, E., Suralta, R. R., Mitsuya, S., & Yamauchi, A. (2015). Matching the expression of root plasticity with soil moisture availability maximizes production of rice plants grown in an experimental sloping bed having soil moisture gradients. Plant Production Science, 18(3), 267–276. https://doi.org/10.1626/pps.18.267
Kovalchuk, N., Laga, H., Cai, J., Kumar, P., Parent, B., Lu, Z., Miklavcic, S. J., & Haefele, S. M. (2016). Phenotyping of plants in competitive but controlled environments: A study of drought response in transgenic wheat. Functional Plant Biology, 44(3), 290–301. https://doi.org/10.1071/FP16202
Krishna, S. S., & Reddy, Y. A. N. (2021). Root growth pattern in finger millet under well-watered and drought stress condition: Its relation to shoot mass production. Plant Physiology Reports. https://doi.org/10.1007/s40502-021-00592-2
Lafitte, R. (2002). Relationship between leaf relative water content during reproductive stage water decit and grain formation in rice. Field Crops Research, 76, 165–174. https://doi.org/10.1016/S0378-4290(02)00037-0
Larson, J. E., & Funk, J. L. (2016). Seedling root responses to soil moisture and the identification of a belowground trait spectrum across three growth forms. New Phytologist, 210(3), 827–838. https://doi.org/10.1111/nph.13829
Li, L., Zhang, T., Zhao, C., Li, Y., Li, Y., & Mu, C. (2019). Leaf and stem traits variation of Stellera chamaejasme Linn. with slope aspect in alpine steppe. Ecological Research, 34, 119–126. https://doi.org/10.1111/1440-1703.1007
Liu, H., Searle, I. R., Mather, D. E., Able, A. J., & Able, J. A. (2015). Morphological, physiological and yield responses of durum wheat to pre-anthesis water-deficit stress are genotype-dependent. Crop and Pasture Science, 66(10), 1024–1038. https://doi.org/10.1071/CP15013
Liu, C., Li, C., Liang, D., Ma, F., Wang, S., Wang, P., & Wang, R. (2013). Aquaporin expression in response to water-deficit stress in two Malus species: Relationship with physiological status and drought tolerance. Plant Growth Regulation, 70, 187–197. https://doi.org/10.1007/s10725-013-9791-x
Magney, T. S., Eitel, J. U. H., Huggins, D. R., & Vierling, L. A. (2016). Proximal NDVI derived phenology improves in-season predictions of wheat quantity and quality. Agricultural and Forest Meteorology, 217, 46–60. https://doi.org/10.1016/j.agrformet.2015.11.009
Mahey, R. K., Singh, R., Sidhu, S. S., & Narang, R. S. (1991). The use of remote sensing to assess the effects of water stress on wheat. Experimental Agriculture, 27, 423–429. https://doi.org/10.1017/S0014479700019402
Nada, R. M., & Abogadallah, G. M. (2018). Root-favoured biomass allocation improves growth and yield of field-grown rice (Oryza sativa L.) plants only when the shoot sink is expandable. Acta Physiologiae Plantarum, 40(123), 1–17. https://doi.org/10.1007/s11738-018-2697-5
Nassir, A. L., Olagunju, S. O., Adewusi, K. M., & Mustapha, A. (2019). Differential expression of upland rice traits as influenced by soil moisture conditions. Ife Journal of Agriculture, 31(3), 77–91.
Nehemy, M. F., Benettin, P., Asadollahi, M., Pratt, D., Rinaldo, A., & Mcdonnell, J. J. (2021). Tree water deficit and source water partitioning. Hydrological Processes, 35(1), e14004. https://doi.org/10.1002/hyp.14004
Olagunju, S. O., Atayese, M. O., Sakariyawo, O. S., & Dare, E. O. (2022). Effects of multi-growth stage water deficit and orthosilicic acid fertiliser on lodging resistance of rice cultivars. Crop and Pasture Science, 73(4), 370–389. https://doi.org/10.1071/CP21563
Olagunju, S. O., Nassir, A. L., Adewusi, K. M., Oguntade, O. A., Odusanya, O. A., & Azeez, A. A. (2018). Patterns of biomass allocation in upland rice cultivars grown on soils along a toposequence. Pertanika Journal of Tropical Agricultural Science, 41(1), 287–303.
Olagunju, S. O., Sakariyawo, O. S., Atayese, M. O., Dare, E. O., Nassir, A. L., & Adesina, E. H. (2020). Analyses of grain yield components of selected upland rice grown in valley bottom soil under rates of foliar ortho-silicate acid fertiliser. African Crop Science Journal, 28(1), 79–92. https://doi.org/10.4314/acsj.v28i1.7
Payne, R. W., Harding, S. A., Murray, D. A., Soutar, D. M., Baird, D. B., Glaser, A. I., Channing, I. C., Welham, S. J., Gilmour, A., Thompson, R., & Webster, R. (2009). GenStat for windows: Introduction (12th ed.). VSN International.
Petrov, V., Hille, J., Mueller-roeber, B., & Gechev, T. S. (2015). ROS-mediated abiotic stress-induced programmed cell death in plants. Frontiers in Plant Science, 6(69), 1–16. https://doi.org/10.3389/fpls.2015.00069
Raju, B. R., Narayanaswamy, B. R., Mohankumar, M. V., Sumanth, K. K., Rajanna, M. P., Mohanraju, B., Udayakumar, M., & Sheshshayee, M. S. (2014). Root traits and cellular level tolerance hold the key in maintaining higher spikelet fertility of rice under water limited conditions. Functional Plant Biology, 41(9), 939–939. https://doi.org/10.13140/RG.2.1.4994.4089
Sharma, K. D., Kumar, A., Yadav, R., & Singh, K. (2014). Leaf water status evaluation and its associations in green gram (Vigna radiata L.) under rainfed environment. Indian Juornal of Plant Physiology, 19(2), 144–148. https://doi.org/10.1007/s40502-014-0088-5
Sicher, R. C., Timlin, D., & Bailey, B. (2012). Responses of growth and primary metabolism of water-stressed barley roots to rehydration. Journal of Plant Physiology, 162(7), 686–695. https://doi.org/10.1016/j.jplph.2012.01.002
Smith, M. R., Rao, I. M., & Merchant, A. (2018). Source-sink relationships in crop plants and their influence on yield development and nutritional quality. Frontiers in Plant Science, 9, 1889. https://doi.org/10.3389/fpls.2018.01889
Sofi, P. A., Rehman, K., Gull, M., Kumari, J., Djanaguiraman, M., & Prasad, P. V. V. (2021). Integrating root architecture and physiological approaches for improving drought tolerance in common bean (Phaseolus vulgaris L.). Plant Physiology Reports, 26(1), 4–22. https://doi.org/10.1007/s40502-021-00570-8
Song, Y., Birch, C. J., & Hanan, J. (2010). Maize canopy production under contrasted water regimes. Annals of Applied Biology, 157, 111–123. https://doi.org/10.1111/j.1744-7348.2010.00412.x
Song, Y., Birch, C., Rui, Y., & Hanan, J. (2015). Allometric relationships of maize organ development under different water regimes and plant densities. Plant Production Science, 18(1), 1–10. https://doi.org/10.1626/pps.18.1
Sun, S., Jin, D., & Shi, P. (2006). The leaf size—Twig size spectrum of temperate woody species along an altitudinal gradient: An invariant allometric scaling relationship. Annals of Botany, 97, 97–107. https://doi.org/10.1093/aob/mcj004
Tuna, A. L., Kaya, C., Ashraf, M., Altunlu, H., Yokas, I., & Yagmur, B. (2007). The effect of calcium sulphate on growth, membrane stability and nutrient uptake of tomato plants grain under salt stress. Environmental and Experimental Botany, 59, 173–178. https://doi.org/10.1016/j.envexpbot.2005.12.007
Wang, E., Robertson, M. J., Hammer, G. L., Carberry, P. S., Holzworth, D., Meinke, H., Chapman, S. C., Hargreaves, J. N., Huth, N. I., & McLean, G. (2002). Development of a generic crop model template in the cropping system model APSIM. European Journal of Agronomy, 18(1–2), 121–140. https://doi.org/10.1016/S1161-0301(02)00100-4
Williams, L. E., & Araujo, F. (2002). Correlations among predawn leaf, midday leaf, and midday stem water potential and their correlations with other measures of soil and plant water status in Vitis vinifera. Journal of American Society for Horticultural Science, 127(3), 448–454.
Wu, C. A., Lowry, D. B., Nutter, L. I., & Willis, J. H. (2010). Natural variation for drought-response traits in the Mimulus guttatus species complex. Oecologia, 162, 23–33. https://doi.org/10.1007/s00442-009-1448-0
Yamasaki, S., & Dillenburg, L. R. (1999). Measurement of leaf relative water content in Araucaria angustifolia. Revista Brasileira De Fisiologia Vegetal, 11(2), 69–75.
Yan, H.-P., Kang, M., Reffye, P. D., & Dingkuhn, M. (2004). A dynamic, architectural plant model simulating resource-dependent growth. Annals of Botany, 93, 591–602. https://doi.org/10.1093/aob/mch078
Zhang, Y., Li, Y., & Xie, J. B. (2016). Fixed allocation patterns, rather than plasticity, benefit recruitment and recovery from drought in seedlings of a desert shrub. AoB Plants, 8, 1–11. https://doi.org/10.1093/aobpla/plw020
Zhou, X. B., Yang, L., Wang, G. Y., Zhao, Y. X., & Wu, Y. H. (2020). Effect of deficit irrigation scheduling and planting pattern on leaf water status and radiation use efficiency of winter wheat. Journal of Agronomy and Crop Science, 207(3), 437–449. https://doi.org/10.1111/jac.12466
Zu, X., Lu, Y., Wang, Q., Chu, P., Miao, W., Wang, H., & La, H. (2017). A new method for evaluating the drought tolerance of upland rice cultivars. Crop Journal, 5(6), 488–498. https://doi.org/10.1016/j.cj.2017.05.002
Acknowledgements
Appreciation goes to Tertiary Education Trust Fund (TETFund) for sponsoring projects OOU/IBR/012 and OOU/IBR/014 in the College. The equipment and materials purchased from the projects made it possible to successfully complete this research.
Author information
Authors and Affiliations
Contributions
Study conception, design, data analysis and drafting of manuscript: SOO; Conduction of experiment: OCF and EOA; Revision of manuscript: ALN, OAO, ROO, OSS.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Olagunju, S.O., Folarin, O.C., Adenaike, E.O. et al. Fresh weight of vegetative organs improves grain yield prediction in upland rice under pre-anthesis water deficit. Plant Physiol. Rep. 27, 358–373 (2022). https://doi.org/10.1007/s40502-022-00685-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-022-00685-6