Abstract
Background
Aging is a complex systems level problem that needs a systems level solution. However, system models of aging and longevity, although urgently needed, are still lacking, largely due to the paucity of conceptual frameworks for modeling such a complex process.
Results
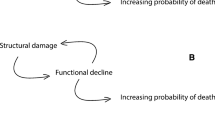
We propose that aging can be viewed as a decline in system capacity, defined as the maximum level of output that a system produces to fulfill demands. Classical aging hallmarks and anti-aging strategies can be well-aligned to system capacity. Genetic variants responsible for lifespan variation across individuals or species can also be explained by their roles in system capacity.We further propose promising directions to develop systems approaches to modulate system capacity and thus extend both healthspan and lifespan.
Conclusions
The system capacity model of aging provides an opportunity to examine aging at the systems level. This model predicts that the extent to which aging can be modulated is normally limited by the upper bound of the system capacity of a species.Within such a boundary, aging can be delayed by moderately increasing an individual’s system capacity. Beyond such a boundary, increasing the upper bound is required, which is not unrealistic given the unlimited potential of regenerative medicine in the future, but it requires increasing the capacity of the whole system instead of only part of it.

Article PDF
Similar content being viewed by others
References
Liao, C. Y., Rikke, B. A., Johnson, T. E., Diaz, V. and Nelson, J. F. (2010) Genetic variation in the murine lifespan response to dietary restriction: from life extension to life shortening. Aging Cell, 9, 92–95
Schleit, J., Johnson, S. C., Bennett, C. F., Simko, M., Trongtham, N., Castanza, A., Hsieh, E. J., Moller, R. M., Wasko, B. M., Delaney, J. R., et al. (2013) Molecular mechanisms underlying genotype-dependent responses to dietary restriction. Aging Cell, 12, 1050–1061
Beekman, M., Blanché, H., Perola, M., Hervonen, A., Bezrukov, V., Sikora, E., Flachsbart, F., Christiansen, L., De Craen, A. J., Kirkwood, T. B., et al. (2013) Genome-wide linkage analysis for human longevity: genetics of healthy aging study. Aging Cell, 12, 184–193
Deelen, J., Beekman, M., Uh, H. W., Helmer, Q., Kuningas, M., Christiansen, L., Kremer, D., van der Breggen, R., Suchiman, H. E., Lakenberg, N., et al. (2011) Genome-wide association study identifies a single major locus contributing to survival into old age; the APOE locus revisited. Aging Cell, 10, 686–698
Flachsbart, F., Caliebe, A., Kleindorp, R., Blanché, H., von Eller-Eberstein, H., Nikolaus, S., Schreiber, S. and Nebel, A. (2009) Association of FOXO3A variation with human longevity confirmed in German centenarians. Proc. Natl. Acad. Sci. USA, 106, 2700–2705
Li, Y., Wang, W. J., Cao, H., Lu, J., Wu, C., Hu, F. Y., Guo, J., Zhao, L., Yang, F., Zhang, Y. X., et al. (2009) Genetic association of FOXO1A and FOXO3A with longevity trait in Han Chinese populations. Hum. Mol. Genet., 18, 4897–4904
Nebel, A., Kleindorp, R., Caliebe, A., Nothnagel, M., Blanché, H., Junge, O., Wittig, M., Ellinghaus, D., Flachsbart, F., Wichmann, H. E., et al. (2011) A genome-wide association study confirms APOE as the major gene influencing survival in long-lived individuals. Mech. Ageing Dev., 132, 324–330
Newman, A. B., Walter, S., Lunetta, K. L., Garcia, M. E., Slagboom, P. E., Christensen, K., Arnold, A. M., Aspelund, T., Aulchenko, Y. S., Benjamin, E. J., et al. (2010) A meta–analysis of four genome–wide association studies of survival to age 90 years or older: the cohorts for heart and aging research in genomic epidemiology consortium. J. Gerontol. A Biol. Sci. Med. Sci., 65A, 478–487
Hou, L., Wang, D., Chen, D., Liu, Y., Zhang, Y., Cheng, H., Xu, C., Sun, N., McDermott, J., Mair, W. B., et al. (2016) A systems approach to reverse engineer lifespan extension by dietary restriction. Cell Metab., 23, 529–540
Špicar, R. (2014) System dynamics archetypes in capacity planning. Procedia Eng., 69, 1350–1355
Hahm, J. H., Kim, S., DiLoreto, R., Shi, C., Lee, S. J., Murphy, C. T. and Nam, H. G. (2015) C. elegans maximum velocity correlates with healthspan and is maintained in worms with an insulin receptor mutation. Nat. Commun., 6, 8919
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. and Kroemer, G. (2013) The hallmarks of aging. Cell, 153, 1194–1217
Rea, S. L., Ventura, N. and Johnson, T. E. (2007) Relationship between mitochondrial electron transport chain dysfunction, development, and life extension in Caenorhabditis elegans. PLoS Biol., 5, e259
Cohen, E., Du, D., Joyce, D., Kapernick, E. A., Volovik, Y., Kelly, J.W. and Dillin, A. (2010) Temporal requirements of insulin/IGF-1 signaling for proteotoxicity protection. Aging Cell, 9, 126–134
Kenyon, C. J. (2010) The genetics of ageing. Nature, 464, 504–512
Hashizume, O., Ohnishi, S., Mito, T., Shimizu, A., Ishikawa, K., Nakada, K., Soda, M., Mano, H., Togayachi, S., Miyoshi, H., et al. (2015) Epigenetic regulation of the nuclear-coded GCAT and SHMT2 genes confers human age-associated mitochondrial respiration defects. Sci. Rep., 5, 10434
Edgar, D., Shabalina, I., Camara, Y., Wredenberg, A., Calvaruso, M. A., Nijtmans, L., Nedergaard, J., Cannon, B., Larsson, N. G. and Trifunovic, A. (2009) Random point mutations with major effects on protein-coding genes are the driving force behind premature aging in mtDNA mutator mice. Cell Metab., 10, 131–138
Hiona, A., Sanz, A., Kujoth, G. C., Pamplona, R., Seo, A. Y., Hofer, T., Someya, S., Miyakawa, T., Nakayama, C., Samhan-Arias, A. K., et al. (2010) Mitochondrial DNA mutations induce mitochondrial dysfunction, apoptosis and sarcopenia in skeletal muscle of mitochondrial DNA mutator mice. PLoS One, 5, e11468
Sena, L. A. and Chandel, N. S. (2012) Physiological roles of mitochondrial reactive oxygen species. Mol. Cell, 48, 158–167
Dillin, A., Hsu, A. L., Arantes-Oliveira, N., Lehrer-Graiwer, J., Hsin, H., Fraser, A. G., Kamath, R. S., Ahringer, J. and Kenyon, C. (2002) Rates of behavior and aging specified by mitochondrial function during development. Science, 298, 2398–2401
Vilchez, D., Morantte, I., Liu, Z., Douglas, P. M., Merkwirth, C., Rodrigues, A. P., Manning, G. and Dillin, A. (2012) RPN-6 determines C. elegans longevity under proteotoxic stress conditions. Nature, 489, 263–268
Bernardes de Jesus, B., Vera, E., Schneeberger, K., Tejera, A. M., Ayuso, E., Bosch, F. and Blasco, M. A. (2012) Telomerase gene therapy in adult and old mice delays aging and increases longevity without increasing cancer. EMBO Mol. Med., 4, 691–704
Greer, E. L., Maures, T. J., Hauswirth, A. G., Green, E. M., Leeman, D. S., Maro, G. S., Han, S., Banko, M. R., Gozani, O. and Brunet, A. (2010) Members of the H3K4 trimethylation complex regulate lifespan in a germline-dependent manner in C. elegans. Nature, 466, 383–387
Jin, C., Li, J., Green, C. D., Yu, X., Tang, X., Han, D., Xian, B., Wang, D., Huang, X., Cao, X., et al. (2011) Histone demethylase UTX-1 regulates C. elegans life span by targeting the insulin/IGF-1 signaling pathway. Cell Metab., 14, 161–172
Satoh, A., Brace, C. S., Rensing, N., Cliften, P., Wozniak, D. F., Herzog, E. D., Yamada, K. A. and Imai, S. (2013) Sirt1 extends life span and delays aging in mice through the regulation of Nk2 homeobox 1 in the DMH and LH. Cell Metab., 18, 416–430
Viswanathan, M. and Guarente, L. (2011) Regulation of Caenorhabditis elegans lifespan by sir-2.1 transgenes. Nature, 477, E1–E2
Zhou, B., Yang, L., Li, S., Huang, J., Chen, H., Hou, L., Wang, J., Green, C. D., Yan, Z., Huang, X., et al. (2012) Midlife gene expressions identify modulators of aging through dietary interventions. Proc. Natl. Acad. Sci. USA, 109, E1201–E1209
Burkewitz, K., Zhang, Y. and Mair, W. B. (2014) AMPK at the nexus of energetics and aging. Cell Metab., 20, 10–25
Greer, E. L. and Brunet, A. (2009) Different dietary restriction regimens extend lifespan by both independent and overlapping genetic pathways in C. elegans. Aging Cell, 8, 113–127
Johnson, S. C., Rabinovitch, P. S. and Kaeberlein, M. (2013) mTOR is a key modulator of ageing and age-related disease. Nature, 493, 338–345
Longo, V. D. and Kennedy, B. K. (2006) Sirtuins in aging and agerelated disease. Cell, 126, 257–268
Loffredo, F. S., Steinhauser, M. L., Jay, S. M., Gannon, J., Pancoast, J. R., Yalamanchi, P., Sinha, M., Dall’ Osso, C., Khong, D., Shadrach, J. L., et al. (2013) Growth differentiation factor 11 is a circulating factor that reverses age-related cardiac hypertrophy. Cell, 153, 828–839
Miller, J. D., Ganat, Y. M., Kishinevsky, S., Bowman, R. L., Liu, B., Tu, E. Y., Mandal, P. K., Vera, E., Shim, J. W., Kriks, S., et al. (2013) Human iPSC-based modeling of late-onset disease via progerin-induced aging. Cell Stem Cell, 13, 691–705
Studer, L., Vera, E. and Cornacchia, D. (2015) Programming and reprogramming cellular age in the era of induced pluripotency. Cell Stem Cell, 16, 591–600
Dong, X., Milholland, B. and Vijg, J. (2016) Evidence for a limit to human lifespan. Nature, 538, 257–259
Ashur-Fabian, O., Avivi, A., Trakhtenbrot, L., Adamsky, K., Cohen, M., Kajakaro, G., Joel, A., Amariglio, N., Nevo, E. and Rechavi, G. (2004) Evolution of p53 in hypoxia-stressed Spalax mimics human tumor mutation. Proc. Natl. Acad. Sci. USA, 101, 12236–12241
Avivi, A., Ashur-Fabian, O., Joel, A., Trakhtenbrot, L., Adamsky, K., Goldstein, I., Amariglio, N., Rechavi, G. and Nevo, E. (2007) P53 in blind subterranean mole rats — loss-of-function versus gain-of-function activities on newly cloned Spalax target genes. Oncogene, 26, 2507–2512
Seluanov, A., Hine, C., Azpurua, J., Feigenson, M., Bozzella, M., Mao, Z., Catania, K. C. and Gorbunova, V. (2009) Hypersensitivity to contact inhibition provides a clue to cancer resistance of naked mole-rat. Proc. Natl. Acad. Sci. USA, 106, 19352–19357
Andziak, B., O’ Connor, T. P., Qi, W., DeWaal, E. M., Pierce, A., Chaudhuri, A. R., Van Remmen, H. and Buffenstein, R. (2006) High oxidative damage levels in the longest-living rodent, the naked mole-rat. Aging Cell, 5, 463–471
Pérez, V. I., Buffenstein, R., Masamsetti, V., Leonard, S., Salmon, A. B., Mele, J., Andziak, B., Yang, T., Edrey, Y., Friguet, B., et al. (2009) Protein stability and resistance to oxidative stress are determinants of longevity in the longest-living rodent, the naked mole-rat. Proc. Natl. Acad. Sci. USA, 106, 3059–3064
Azpurua, J., Ke, Z., Chen, I. X., Zhang, Q., Ermolenko, D. N., Zhang, Z. D., Gorbunova, V. and Seluanov, A. (2013) Naked molerat has increased translational fidelity compared with the mouse, as well as a unique 28S ribosomal RNA cleavage. Proc. Natl. Acad. Sci. USA, 110, 17350–17355
Rodriguez, K., Wywial, E. I., Perez, V. J., Lambert, A. H., Edrey, Y. N., Lewis, K., Grimes, K. L., Lindsey, M. D., Brand, M. and Buffenstein, R. (2011) Walking the oxidative stress tightrope: a perspective from the naked mole-rat, the longest-living rodent. Curr. Pharm. Des., 17, 2290–2307
Zhao, S., Lin, L., Kan, G., Xu, C., Tang, Q., Yu, C., Sun, W., Cai, L., Xu, C. and Cui, S. (2014) High autophagy in the naked mole rat may play a significant role in maintaining good health. Cell. Physiol. Biochem., 33, 321–332
Buffenstein, R. and Yahav, S. (1991) The effect of diet on microfaunal population and function in the caecum of a subterranean naked mole-rat, Heterocephalus glaber. Br. J. Nutr., 65, 249–258
Kim, E. B., Fang, X., Fushan, A. A., Huang, Z., Lobanov, A. V., Han, L., Marino, S. M., Sun, X., Turanov, A. A., Yang, P., et al. (2011) Genome sequencing reveals insights into physiology and longevity of the naked mole rat. Nature, 479, 223–227
Fang, X., Nevo, E., Han, L., Levanon, E. Y., Zhao, J., Avivi, A., Larkin, D., Jiang, X., Feranchuk, S., Zhu, Y., et al. (2014) Genome-wide adaptive complexes to underground stresses in blind mole rats Spalax. Nat. Commun., 5, 3966
Heintz, C., Doktor, T. K., Lanjuin, A., Escoubas, C. C., Zhang, Y., Weir, H. J., Dutta, S., Silva-García, C. G., Bruun, G. H., Morantte, I., et al. (2017) Splicing factor 1 modulates dietary restriction and TORC1 pathway longevity in C. elegans. Nature, 541, 102–106
Wang, E. (2007) MicroRNA, the putative molecular control for mid-life decline. Ageing Res. Rev., 6, 1–11
Bell, R., Hubbard, A., Chettier, R., Chen, D., Miller, J. P., Kapahi, P., Tarnopolsky, M., Sahasrabuhde, S., Melov, S. and Hughes, R. E. (2009) A human protein interaction network shows conservation of aging processes between human and invertebrate species. PLoS Genet., 5, e1000414
Fernandes, M., Wan, C., Tacutu, R., Barardo, D., Rajput, A., Wang, J., Thoppil, H., Thornton, D., Yang, C., Freitas, A., et al. (2016) Systematic analysis of the gerontome reveals links between aging and age-related diseases. Hum. Mol. Genet., 25, 4804–4818
Xue, H., Xian, B., Dong, D., Xia, K., Zhu, S., Zhang, Z., Hou, L., Zhang, Q., Zhang, Y. and Han, J. D. (2007) A modular network model of aging. Mol. Syst. Biol., 3, 147
Acknowledgments
We thank Yizhen Yan for illustration suggestions and acknowledge supports from China Ministry of Science and Technology 2015CB964803 and 2016YFE0108700 and the National Natural Science Foundation of China 91329302, 31210103916 and 91519330, and Chinese Academy of Sciences XDB19020301 and XDA01010303 to J.D.J.H.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Han, JD.J., Hou, L., Sun, N. et al. The system capacity view of aging and longevity. Quant Biol 5, 251–259 (2017). https://doi.org/10.1007/s40484-017-0115-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40484-017-0115-4