Abstract
Purpose
This study aimed to explore the reliability and stability of ultrasound-guided attenuation parameter (UGAP) values obtained by two measuring methods and different measuring times.
Methods
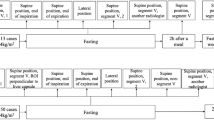
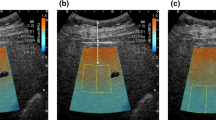
Patients who underwent liver UGAP examinations in our hospital from September 2022 to December 2022 were retrospectively analyzed. The clinical data and UGAP measurements results were collected. Two different measuring methods: static single-frame multi-point measuring and dynamic multi-frame single-point measuring, were performed for each patient, and 10 UGAP values of each measuring method were recorded. The medians of the UGAP values of the 1st–3rd, 1st–5th, 1st–7th and 1st–10th by each measuring method were taken as the final UGAP values of measuring 3, 5, 7 and 10 times. The UGAP values obtained by the two different measuring methods and different measuring times (3, 5, 7 or 10 times) were compared.
Results
206 patients were included in this study. There was no statistical difference between UGAP values measured by static single-frame multi-point measuring and dynamic multi-frame single-point measuring (P = 0.689, P = 0.270, P = 0.298, P = 0.091), regardless of measuring times (3, 5, 7, 10 times). No significant difference between the UGAP values obtained by 3, 5, 7 and 10 measurements was found (P = 0.554, P = 0.916).
Conclusion
The UGAP values obtained by the two different measuring methods and different measuring times (3, 5, 7 and 10 times) are stable and reliable. Additionally, 3 times of UGAP measurements might be enough for each patient in clinical practice.


Similar content being viewed by others
Data availability
No data have been fabricated or manipulated (including images) to support the conclusions. No data, text, or theories by others are presented as if they are the author's own. All co-authors have contributed sufficiently to the scientific work.
References
Cusi K, Isaacs S, Barb D et al (2022) American Association of Clinical Endocrinology Clinical Practice guideline for the diagnosis and management of nonalcoholic fatty liver disease in primary care and endocrinology clinical settings: co-sponsored by the American Association for the study of liver diseases (AASLD). Endocr Pract 28(5):528–562. https://doi.org/10.1016/j.eprac.2022.03.010
Zhou J, Zhou F, Wang W et al (2020) Epidemiological features of NAFLD from 1999 to 2018 in China. Hepatology 71(5):1851–1864. https://doi.org/10.1002/hep.31150
Park J, Lee JM, Lee G et al (2022) Quantitative evaluation of hepatic steatosis using advanced imaging techniques: focusing on new quantitative ultrasound techniques. Korean J Radiol 23(1):13–29. https://doi.org/10.3348/kjr.2021.0112
Castera L, Friedrich-Rust M, Loomba R (2019) Noninvasive assessment of liver disease in patients with nonalcoholic fatty liver disease. Gastroenterology 156(5):1264-1281.e4. https://doi.org/10.1053/j.gastro.2018.12.036
European Association for the Study of the Liver (2021) EASL Clinical Practice Guidelines on non-invasive tests for evaluation of liver disease severity and prognosis—2021 update. J Hepatol 75(3):659–689. https://doi.org/10.1016/j.jhep.2021.05.025
Miele L, Zocco MA, Pizzolante F et al (2020) Use of imaging techniques for non-invasive assessment in the diagnosis and staging of non-alcoholic fatty liver disease. Metabolism 112:154355. https://doi.org/10.1016/j.metabol.2020.154355
Ferraioli G, Berzigotti A, Barr RG et al (2021) Quantification of liver fat content with ultrasound: a WFUMB position paper. Ultrasound Med Biol 47(10):2803–2820. https://doi.org/10.1016/j.ultrasmedbio.2021.06.002
Tamaki N, Ajmera V, Loomba R (2022) Non-invasive methods for imaging hepatic steatosis and their clinical importance in NAFLD. Nat Rev Endocrinol 18(1):55–66. https://doi.org/10.1038/s41574-021-00584-0
Starekova J, Hernando D, Pickhardt PJ et al (2021) Quantification of liver fat content with CT and MRI: state of the art. Radiology 301(2):250–262. https://doi.org/10.1148/radiol.2021204288
Pirmoazen AM, Khurana A, El Kaffas A et al (2020) Quantitative ultrasound approaches for diagnosis and monitoring hepatic steatosis in nonalcoholic fatty liver disease. Theranostics 10(9):4277–4289. https://doi.org/10.7150/thno.40249
Ballestri S, Nascimbeni F, Lugari S et al (2019) A critical appraisal of the use of ultrasound in hepatic steatosis. Expert Rev Gastroenterol Hepatol 13(7):667–681. https://doi.org/10.1080/17474124.2019.1621164
Kumada T, Ogawa S, Goto T et al (2022) Intra-individual comparisons of the ultrasound-guided attenuation parameter and the magnetic resonance imaging-based proton density fat fraction using bias and precision statistics. Ultrasound Med Biol 48(8):1537–1546. https://doi.org/10.1016/j.ultrasmedbio.2022.03.019
Imajo K, Toyoda H, Yasuda S et al (2022) Utility of ultrasound-guided attenuation parameter for grading steatosis with reference to MRI-PDFF in a large cohort. Clin Gastroenterol Hepatol 20(11):2533-2541.e7. https://doi.org/10.1016/j.cgh.2021.11.003
Jeon SK, Lee JM, Joo I et al (2022) Assessment of the inter-platform reproducibility of ultrasound attenuation examination in nonalcoholic fatty liver disease. Ultrasonography 41(2):355–364. https://doi.org/10.14366/usg.21167
Fujiwara Y, Kuroda H, Abe T et al (2018) The B-mode image-guided ultrasound attenuation parameter accurately detects hepatic steatosis in chronic liver disease. Ultrasound Med Biol 44(11):2223–2232. https://doi.org/10.1016/j.ultrasmedbio.2018.06.017
Kuroda H, Abe T, Fujiwara Y et al (2021) Diagnostic accuracy of ultrasound-guided attenuation parameter as a noninvasive test for steatosis in non-alcoholic fatty liver disease. J Med Ultrason 48(4):471–480. https://doi.org/10.1007/s10396-021-01123-0
Tada T, Kumada T, Toyoda H et al (2020) Liver stiffness does not affect ultrasound-guided attenuation coefficient measurement in the evaluation of hepatic steatosis. Hepatol Res 50(2):190–198. https://doi.org/10.1111/hepr.13442
Zhao Y, Jia M, Zhang C et al (2022) Reproducibility of ultrasound-guided attenuation parameter (UGAP) to the noninvasive evaluation of hepatic steatosis. Sci Rep 12(1):2876. https://doi.org/10.1038/s41598-022-06879-0
Iwashita H, Shakado S, Yoshimaru N et al (2022) Clinical utility of ultrasound-guided attenuation parameter for the detection and quantification of hepatic steatosis in patients with fatty liver diagnosed by computed tomography. Ultrasound Med Biol 48(7):1282–1289. https://doi.org/10.1016/j.ultrasmedbio.2022.02.023
Yoon H, Kim J, Lim HJ et al (2022) Attenuation coefficient measurement using a high-frequency (2–9 MHz) convex transducer for children including fatty liver. Ultrasound Med Biol 48(6):1070–1077. https://doi.org/10.1016/j.ultrasmedbio.2022.02.005
Kuroda H, Fujiwara Y, Abe T et al (2021) Two-dimensional shear wave elastography and ultrasound-guided attenuation parameter for progressive non-alcoholic steatohepatitis. PLoS ONE 16(4):e0249493. https://doi.org/10.1371/journal.pone.0249493
Jeon SK, Joo I, Kim SY et al (2021) Quantitative ultrasound radiofrequency data analysis for the assessment of hepatic steatosis using the controlled attenuation parameter as a reference standard. Ultrasonography 40(1):136–146. https://doi.org/10.14366/usg.20042
Kim H, Varghese T (2007) Attenuation estimation using spectral cross-correlation. IEEE Trans Ultrason Ferroelectr Freq Control 54(3):510–519. https://doi.org/10.1109/tuffc.2007.274
Funding
This work was supported by the National Natural Science Foundation of China (82272011), the Natural Science Foundation of Guangdong Province, China (2022A1515012155), the Shenzhen Science and Technology Program (JCYJ20220530160208018), and the Futian Healthcare Research Project (FTWS2020022, FTWS2021071).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection were performed by XZ and LL. Data analysis was performed by HL and SL. The first draft of the manuscript was written by XZ and LL. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the Eighth Affiliated Hospital of Sun Yat-sen University (Date: 21 June 2023/No. 2023–042-01).
Informed consent
Informed consent was waived by the Ethics Committee of the Eighth Affiliated Hospital of Sun Yat-sen University in view of the retrospective nature of the study and all the procedures being performed were part of the routine care.
Consent for publication
The manuscript has not been submitted elsewhere. The manuscript has not been published previously (partly or in full). It has not been split up into several parts and submitted to various journals or to one journal over time.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Luo, L., Liu, H. et al. Reliability and stability of ultrasound-guided attenuation parameter in evaluating hepatic steatosis. J Ultrasound 27, 145–152 (2024). https://doi.org/10.1007/s40477-023-00856-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40477-023-00856-7