Abstract
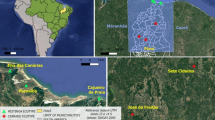
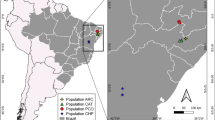
Libidibia is a small genus of caesalpinioid legumes with seven species spanning from Mexico and the Caribbean to southern South America. Within this genus, Libidibia ferrea stands out as an iconic Brazilian tree currently classified into the varieties ferrea, glabrescens, leiostachya, and parvifolia. They comprise a species complex together with three other varieties currently accepted as synonyms (var. cearensis, var. megaphylla and var. petiolulata). Together they exhibit complex morphological variation, along with confusion regarding their common names and geographic distribution. Five distinct morphotypes were recognized which were compared using a morphometric study of 26 quantitative leaf characters. We also performed ecological niche modeling for those morphotypes spanning from Quaternary to the present. Principal Component Analysis (PCA) and Discriminant Analysis (DA) revealed four main clusters which also present distinct niche preferences throughout the Quaternary and current distinct geographical distributions. Based on our findings, we propose recognizing four morphotypes as separate species: L. ferrea, L. juca, L. leiostachya, and L. parvifolia. Libidibia ferrea and L. juca are small trees and shrubs, distributed respectively in the southern portion of the ‘Caatinga’ and from the Amazon to the northern ‘Caatinga’ region. On the other hand, L. leiostachya and L. parvifolia are both tall trees, predominantly inhabiting the wetter regions of the ‘Caatinga’ (L. parvifolia) and extending into the coastal rainforests of southeastern Brazil (L. leiostachya). Three new combinations are proposed and an identification key, diagnostic descriptions, and taxonomic notes are presented.




Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available in the Zenodo repository, [https://zenodo.org/records/10580269?token=eyJhbGciOiJIUzUxMiJ9.eyJpZCI6IjEyM2JhMTMwLTk4OWYtNDA0OS04NDVlLTc3OTlmMzUzNzcxYSIsImRhdGEiOnt9LCJyYW5kb20iOiI3ODBhODcxNjk1NmZkYTUxMGJhM2NkMGJiMmZiZTNiNyJ9.7rE7BaPrlSdfpcEqIZV4ghuQZOoKdjQxHBdrprMvvN5YvslkSza6O-KkbONg5UlYjZNCruU3fCofq4poLjtahg].
References
Abdi H, Williams LJ (2010) Principal component analysis. WIREs. Comput Stat 2:433–459
Aguirre-Gutiérrez J, Serna-Chavez HM, Villalobos-Arambula AR, Rosa JAP, Raes N (2014) Similar but not equivalent: ecological niche comparison across closely–related Mexican white pines. Divers Distrib 21:245–257
Albuquerque IS (2019) Morfologia, distribuição, origem e filogeografia de citótipos diploides e tetraploides de Libidibia ferrea (Leguminosae). PhD Thesis, Univ. Federal de Pernambuco
Allemão FF (1859) Estudo da árvore chamada Jucá. In: Flora Cearense, p 158
Almeida G (1943) Contribuição à dendrometria das essências florestais. Ministério da Agricultura do Brasil, Rio de Janeiro
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46
Andrade-Lima D (1982) Present day forest refuges in Northeastern Brazil. In: Prance GT (ed) Biological diversification in the tropics. Columbia University Press, New York, pp 245–254
Bentham G (1870) Leguminosae, Swartzieae, caesalpinieae, mimoseae. In: Martius KFP von (ed), Flora Brasiliensis, vol. 15, pp 63–74
BIOCLIM (2024) WorldClim. https://www.worldclim.org/data/bioclim.html. Accessed 29 January 2024
Borges LA, Souza LGR, Guerra M, Machado IC, Lewis GP, Lopes AV (2012) Reproductive isolation between diploid and tetraploid cytotypes of Libidibia ferrea (= Caesalpinia ferrea) (Leguminosae): ecological and taxonomic implications. Plant Syst Evol 298:1371–1381
Brito AFS, Pontes AN (2021) Metabólitos secundários de plantas medicinais usadas em garrafadas populares comercializadas em feiras livres de Belém, Pará, Brasil. Enci Bio 18:207–222
Bruneau A, Mercure M, Lewis GP, Herendeen PS (2008) Phylogenetic patterns and diversification in the Caesalpinioid legumes. Bot 86:697–718
Carvalho JCT, Teixeira JRM, Souza PJC, Bastos JK, Filho DS, Sarti SJ (1996) Preliminary studies of analgesic and anti-inflammatory properties of Caesalpinia ferrea crude extract. J Ethnopharmacol 53:175–178
Carvalho PER (2003) Espécies arbóreas Brasileiras. Brasília: embrapa informação Tecnológica. Colombo, Paraná
Castro-Bonilla MA, Bueno MLB, Romano PSR, Dutra VF, Fregonezi JN, Garcia FCP (2022) Species delimitation of the Atlantic forest endemic Inga subnuda (Leguminosae, Caesalpinioideae, Mimosoid Clade) Subspecies based on morphological, ecological, and palaeoecological data. Syst Bot 47:417–430
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Austral Ecol 18:117–143
Conceição AS, Queiroz LP, Lambert SM, Pereira ACS, Borba EL (2007) Biosystematics of Chamaecrista sect. Absus subsect. Baseophyllum (Leguminosae-Caesalpinioideae) based on allozyme and morphometric analyses. Plant Syst Evol 270:183–207
Costa AF, Rodrigues PJFP, Wanderley MGL (2009) Morphometric analysis and taxonomic revision of the Vriesea paraibica complex (Bromeliaceae). Bot J Linn Soc 159:163–181
Dantas AS (2023) Germinação de sementes e estabelecimento in vitro de segmentos nodais de Libidibia ferrea (Mart. ex Tul.) L.P. Queiroz. Undergraduate thesis, Universidade do Estado do Amazonas
Ducke A (1953) As Leguminosas de Pernambuco e Paraíba. Mem Inst Oswaldo Cruz 51:417–461
Egan AN (2015) Species delimitation and recognition in the Pediomelum megalanthum complex (Fabaceae) via multivariate morphometrics. PhytoKeys 44:65–87
Escobar NAG (2018) Estudos taxonômicos no gênero Diptychandra (Leguminosae-Caesalpinioideae). Dissertation, Universidade Estadual de Campinas
Estrella MDL, Aedo C, Velayos M (2009) A morphometric analysis of Daniellia (Fabaceae–Caesalpinioideae). Bot J Linn Soc 159:268–279
Figueiredo N, Silva AV (1967) Alguns elementos novos para o estudo dos batuques de Belém. Atas Do Simpósio Sobre a Biota Amazônica 2:101–122
Fisher RA (1936) The Use of multiple measurements in taxonomic problems. Ann Hum Genet 7:179–188
Ford C (1995) A preliminary revision of the Libidibia group of the genus Caesalpinia (Caesalpinioideae: Leguminosae). Unpublished undergraduate diss., Univ. Bradford
Gagnon E, Lewis GP, Sotuyo JS, Hughes CE, Bruneau A (2013) A molecular phylogeny of Caesalpinia sensu lato: increased sampling reveals new insights and more genera than expected. S Afr J Bot 89:111–127
Gagnon E, Bruneau A, Hughes CE, De Queiroz LP, Lewis GP (2016) A new generic system for the pantropical Caesalpinia group (Leguminosae). PhytoKeys 71:1–160
Gagnon E, Ringelberg JJ, Bruneau A, Lewis GP, Hughes CE (2019) Global succulent biome phylogenetic conservatism across the pantropical Caesalpinia group (Leguminosae). New Phytol 222:1994–2008
Gama R, Aguirre-Gutiérrez J, Stech M (2017) Ecological niche comparison and molecular phylogeny segregate the invasive moss species Campylopus introflexus (Leucobryaceae, Bryophyta) from its closest relatives. Ecol Evol 7:8017–8031
Gasson P, Warner K, Lewis G (2009) Wood anatomy of Caesalpinia s.s., Coulteria, Erythrostemon, Guilandina, Libidibia, Mezoneuron, Poincianella, Pomaria and Tara (Leguminosae, Caesalpinioideae, Caesalpinieae). IAWA J 30:247–276
Glaziou AFM (1906) Plantes du Brésil central. Bull Soc Bot Fr 53:158
Guenther K (1931) A naturalist in Brazil: the flora and fauna and the people of Brazil. G. Allen & Unwin, London
Gunn CR (1991) Fruits and seeds of genera in the subfamily Caesalpinioideae (Fabaceae). Technical Bulletin. US Department of Agriculture, United States
Hammer O, Harper DAT, Ryan PD (2023) PAST: paleontological statistics software package for education and data analysis. http://palaeo-electronica.org/2001_1/past/issue1_01.htm. Accessed 05 Sept 2023
Hawlitschek O, Porch N, Hendrich L, Balke M (2011) Ecological niche modelling and nDNA sequencing support a new, morphologically cryptic beetle species unveiled by DNA barcoding. PLoS ONE 6(2):e16662
Henderson A (2006) Traditional morphometrics in plant systematics and its role in palm systematics. Bot J Linn Soc 151:103–111
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978
Huber JE (1901) Plantae Cearenses. Bulletin De L’herbier Boissier Sér 2(1):304
Jensen BB (2020) Avaliação da fração diclorometano de Libidibia ferrea (Fabales: Fabaceae) em estudos Pré-clínicos da Leishmaniose cutânea. PhD thesis, Universidade Federal do Amazonas
Lewis GP (1987) Legumes of Bahia. Royal Botanic Gardens, Kew
Lewis GP (1998) A revision of the Poincianella-Erythrostemon Group. Royal Botanic Gardens, Kew
Lewis GP (2005) Tribe Caesalpineae. In: Lewis G, Schrire B, Mackinder B, Lock M (eds) Legumes of the world. Royal Botanic Gardens, Kew, pp 126–161
Lewis GP, Schrire BD (1995) A reappraisal of the Caesalpinia group (Caesalpinioideae: Caesalpineae) using phylogenetic analyses. In: Crisp M, Doyle JJ (eds) Advances in Legume Systematics 7: Phylogeny, Royal Botanic Gardens, Kew, UK, pp 41–52
Lorenzi H (1992) Árvores brasileiras: manual de identificação e cultivo de plantas arbóreas nativas do Brasil. Nova Odessa: Plantarum, São Paulo
Martius CFP von, Spix JB von (1828) Reise in Brasilien. Gedruckt bei M. Lindauer, München
Morales M, Arenas L, Remis MI, Wulff AF, Poggio L, Fortunato RH (2014) Morphometric and cytogenetic studies in Mimosa diversipila (Mimosoideae, Leguminosae) and Their taxonomic and evolutionary inferences. Syst Bot 39:875–883
Nores JM, Simpson BB, Hick P, Anton AM, Fortunato RH (2012) The phylogenetic relationships of four monospecific caesalpinioids (Leguminosae) endemic to southern South America. Taxon 61:790–802
Oliveira FG, Fernando EMP (2023) Libidibia in flora e funga do Brasil. Jardim Botânico do Rio de Janeiro. http://floradobrasil.jbrj.gov.br/reflora/floradobrasil/FB109827. Accessed 05 Sept 2023
Pearson K (1901) On Lines and Planes of Closest Fit to Systems of Points in Space. Philo Mag 2:559–572
Pedersen HAE (2010) Species delimitation and recognition in the Brachycorythis helferi complex (Orchidaceae) resolved by multivariate morphometric analysis. Bot J Linn Soc 162:64–76
Pereira LP, Silva RO, Bringel PHSF, Silva KES, Assreuy AMS, Pereira MG (2011) Polysaccharide fractions of Caesalpinia ferrea pods: potential anti-inflammatory usage. J Ethnopharmacol 139:642–648
Phillips SJ, Dudík M (2008) Modeling of species distributions with Maxent: new extensions and a comprehensive evaluation. Ecography 31:161–175
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190:231–259
Pinzon JP, Ramírez-Morillo IM, Fernández-Concha GC (2011) Morphometric analyses within the Tillandsia utriculata L. complex (Bromeliaceae) allow for the recognition of a new species, with notes on its phylogenetic position. J Torrey Bot Soc 138:353–365
Polhill R, Vidal J (1981) Caesalpinieae. In: Polhill RM, Raven PH (eds) Advances in legume systematics, part 1. Royal Botanic Gardens, Kew, pp 81–95
Porter-Utley K (2014) A revision of Passiflora L. subgenus Decaloba (DC.) Rchb. supersection Cieca (Medik.) J. M. MacDougal & Feuillet (Passifloraceae). PhytoKeys 43:1–224
POWO (2023) Libidibia ferrea (Mart. ex Tul.) L.P. Queiroz. https://powo.science.kew.org. Accessed 5 Sept 2023
Prance GT (1971) An index of plant collectors in Brazilian Amazonia. Acta Amaz 1:1
Queiroz K (2007) Species concepts and species delimitation. Syst Biol 56:879–886
Queiroz LP (2009) Leguminosas da Caatinga. Universidade Estadual de Feira de Santana, Feira de Santana
Queiroz LP (2010) New combinations in Libidibia (DC.) Schltdl. and Poincianella Britton & Rose (Leguminosae, Caesalpinioideae). Neodiversity 5:11–12
R Core Team (2023) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 03 Nov 2023
Raxworthy CJ, Ingram CM, Rabibisoa N, Pearson RG (2007) Applications of ecological niche modeling for species delimitation: a review and empirical evaluation using day geckos (Phelsuma) from madagascar. Syst Biol 56:907–923
Rizzini CT, Mattos-Filho AM (1968) Espécies Novas da Flora Brasileira. An Acad Brasil Ciênc 40:231–235
Rocha FD (1919) Botânica Médica Cearense. Typographia Moderna—Carneiro e C.ª, Fortaleza
Rosenegger PF, Falcão LS, Bernardes LG, Oliveira MD, Machado AM, Machado ARSR, Parente AFA, Pando SC (2017) Investigation of antimicrobial and anticoagulant effects of trypsin inhibitor from Caesalpinia ferrea var. cearensis. J Med Biol Sci 4:1–6
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Scrivanti LR, Norrmann GA, Anton AM (2013) Delimiting species boundaries within the Bothriochloa saccharoides complex (Poaceae) through morphometric analysis. Phytotaxa 89:24–42
Shorthouse DP (2010) SimpleMappr, an online tool to produce publication-quality pointmaps. https://www.simplemappr.net. Acessed 10 Jan 2024
Shrestha N, Zhang X (2014) Recircumscription of Huperzia serrata complex in China using morphological and climatic data. J Syst Evol 9999:1–18
Soladoye MO, Sonibare MA, Chykwuma EC (2010) Morphometric study of the genus Indigofera Linn. (Leguminosae-Papilionoideae) in South-Western Nigeria. Int J Botany 6:343–350
Souza IM, Funch LS, de Queiroz LP (2014) Morphological analyses suggest a new taxonomic circumscription for Hymenaea courbaril L. (Leguminosae, Caesalpinioideae). PhytoKeys 38:101–118
Stafleu FA, Cowan RS (1976) Taxonomic literature: A selective guide to botanical publications and collections with dates, commentaries and types (Second edition, vol. 1). Bohn, Scheltema, and Holkema, The Netherlands
Stafleu FA, Cowan RS (1985) Taxonomic literature: A selective guide to botanical publications and collections with dates, commentaries and types (Second edition, vol. 5). Bohn, Scheltema, and Holkema, The Netherlands
Stearn WT (1966) Botanical latin: History, grammar, sybtax, Terminology and Vocabulary. Hafner Publishing Company, New York
Sullivan LM (2006) Adventure guide to Aruba, Bonaire & Curaçao. Hunter Publishing Inc, New Jersey
Thiers B (2023) Index herbariorum: a global directory of public herbaria and associated staff. New York Botanical Garden's Virtual Herbarium. http://sweetgum.nybg.org/science/ih/. Accessed 05 Sept 2023
Tulasne LR (1844) Légumineuses arborescentes de L’Amérique du Sud. Arch Mus Hist Nat 4:137
Turland NJ, Wiersema JH, Barrie FR, Greuter W, Hawksworth DL, Herendeen PS, Knapp S, Kusber WH, Li DZ, Marhold K, May TW, McNeill J, Monro AM, Prado J, Price MJ, Smith GF (eds.) (2018) International Code of nomenclature for algae, fungi, and plants (Shenzhen Code) adopted by the Nineteenth International Botanical Congress Shenzhen, China, July 2017. Regnum Vegetabile 159. Glashütten: Koeltz Botanical Books, Saxony
Ulibarri E (1996) Sinopsis de Caesalpinia y Hoffmannseggia (Leguminosae-Caesalpinioideae) de Sudamérica. Darwiniana 34:299–348
Urban I (1906) Tabulae physiognomicae explicata. Vitae intineraque collectorum botanicorum, notae collaboratorum biographicae, florae brasiliensis ratio edendi chronologica, systema, infex familiarum. In: Martius CPF, Eichler AG, Urban I. Flora Brasiliensis Monachii et Lipsiae, pp 1–268
Vasquez SI, Welker CAD, Neffa VGS, Peichoto MC (2022) Morphometric analysis of the Saccharum complex (Poaceae, Andropogoneae). Plant Syst Evol 308:10
Wiens JJ, Graham CH (2005) Niche conservatism: integrating evolution, ecology, and conservation biology. Annu Rev Ecol Syst 36:519–539
Wurdack JJ (1970) Erroneous data in Glaziou collections of Melastomataceae. Taxon 19:911–913
Acknowledgements
This study was financed by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001 and UEFS-FINAPESQ 045/2023. We thank the Postgraduate Program in Botany at the State University of Feira de Santana (PPGBOT/UEFS) and CNPq/PPBIO Semiárido network (Process 441271/2023-5) for financial support for field trips and laboratory equipment. LPQ and RPO are supported by CNPq grants (Processes 305230/2021-2 and 310515/2022-0, respectively). We thank Haroldo C. de Lima, Luiz Gustavo Rodrigues Pereira and two anonymous reviewers for their comments and suggestions and Rosana Simões Medeiros for her assistance in searching for information on Caesalpinia juca and the works of Freire-Allemão. We also express gratitude to Carolina Lima Ribeiro and Deivid Andrés Fonseca Cortés for their support in the field and laboratory work. Special thanks to the photographers of the images reproduced from the iNaturalist website. The specimens used in this work are registered in the Sistema Nacional de Gestão do Patrimônio Genético e do Conhecimento Tradicional Associado (SisGen) under registration number AE4D7C9.
Author information
Authors and Affiliations
Contributions
FGO and LPQ designed the study and carried out the morphometric analyses. FSS performed niche modeling analyses. FGO, FSS and LPQ carried out fieldworks. All authors contributed to the writing of the article. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Luciano Paganucci de Queiroz is a Guest Editor for the Brazilian Journal of Botany and the peer-review process for this article was independently handled by another member of the journal editorial board.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oliveira, F.G., Santos, F.d.S., Lewis, G.P. et al. Reassessing the taxonomy of the Libidibia ferrea complex, the iconic Brazilian tree “pau-ferro” using morphometrics and ecological niche modeling. Braz. J. Bot (2024). https://doi.org/10.1007/s40415-024-01011-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40415-024-01011-0