Abstract
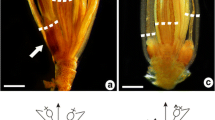
Most species of Sapindaceae are described as monoecious, sharing the presence of staminate flowers and flowers that are morphologically perfect but functionally pistillate. Duodichogamy has also been recorded in the family, but with few evaluations of the structures of different flower types or the functionality of reproductive structures in the different genera analyzed. Cupania L. is a monophyletic genus that occurs in tropical and subtropical America, consisting of monoecious duodichogamous trees with functionally unisexual, actinomorphic, and nectariferous flowers. Cupania emarginata Cambess. is endemic to Brazil, occurring in the northeast and southeast regions, mainly on sandy coastal plains (restingas). This study described and analyzed the flowers of this species based on morphometry and anatomy, and evaluated the functionality of the reproductive whorls and nectaries in the different floral types. Staminate flowers from the first and third flowering phases differ morpho-structurally; the gynoecium (pistillode) in first-flowering staminate flowers is markedly more collapsed than the gynoecium of third-flowering staminate flowers, in which the ovules may differentiate and contain embryo sacs with hypertrophied cells; mature anthers of pistillate flowers contain pollen grains at the stage of two-celled microgametophytes, similar to those of mature anthers of staminate flowers; and anthers of pistillate flowers are indehiscent, probably due to ineffective dehydration, as suggested by the persistence of the septum, which remains intact. The floral nectary is functional in all three flowering phases.
















Similar content being viewed by others
References
Acevedo-Rodríguez P, van Welsen PC, Adema F, van Der Ham RWJM (2011) Sapindaceae. In: Kubitzki K (ed) The families and genera of vascular plants. X. Flowering Plants. Eudicots: Sapindales, Cucurbitales, Myrtaceae. Springer, Berlin, pp 357–407
Aluri JSR, Reddi CS, Das KR (1998) Temporal dioecism and pollination by wasps and bees in Allophylus serratus (Roxb.) Radlk. (Sapindaceae). Plant Species Biol 13:1–5. https://doi.org/10.1111/j.1442-1984.1998.tb00242.x
Appanah S (1982) Pollination of Androdioecious Xerospermum intermedium Radlk. (Sapindaceae) in a rain forest. Biol J Linn Soc 18:11–34. https://doi.org/10.1111/j.1095-8312.1982.tb02031.x
Avalos AA, Lattar EC, Ferrucci MS, Torretta JP (2019a) Reproductive biology of duodichogamous Koelreuteria elegans (Sapindaceae): the rare case of a female-male-female flowering sequence. Aust J Bot 67:149–158. https://doi.org/10.1071/BT18159
Avalos AA, Zini LM, Ferrucci MS, Lattar EC (2019b) Anther and gynoecium structure and development of male and female gametophytes of Koelreuteria elegans subsp. formosana (Sapindaceae): phylogenetic implications. Flora 255:98–109. https://doi.org/10.1016/j.flora.2019.04.003
Bawa KS (1977) The reproductive biology of Cupania guatemalensis Radlk. (Sapindaceae). Evolution 31:52–63. https://doi.org/10.1111/j.1558-5646.1977.tb00981.x
Bawa KS, Opler PA (1975) Dioecism in tropical forest trees. Evolution 29:167–179. https://doi.org/10.2307/2407150
Budar F, Pelletier G (2001) Male sterility in plants: occurrence, determinism, significance and use. Comptes Rendus De L’académie Des Sci Ser III Sci De La Vie 324:543–550. https://doi.org/10.1016/S0764-4469(01)01324-5
Buerki S, Forest F, Acevedo-Rodrígues P, Callmander MW, Nylander JAA, Harrington M, Sanmartim I, Kupfer P, Alvarez N (2009) Plastid and nuclear DNA markers reveal intricate relationships at subfamilial and tribal levels in the soapberry family (Sapindaceae). Mol Phylogenet Evol 51:238–258. https://doi.org/10.1016/j.ympev.2009.01.012
Cao LM, Xia NH, Deng YF (2008) Embryology of Handeliodendron bodinieri (Sapindaceae) and its systematic value: development of male and female gametophytes. Plant Syst Evol 274:17–23. https://doi.org/10.1007/s00606-008-0024-0
Cao LM, Cao M, Liu JH, Wang ZX, Lin Q, Xia NH (2014) Sporogenesis and gametogenesis of Delavaya toxocarpa (Sapindaceae) and their systematic implications. J Syst Evol 52:533–539. https://doi.org/10.1111/jse.12083
Cupania in Flora do Brasil 2020. Jardim Botânico do Rio de Janeiro. http://floradobrasil.jbrj.gov.br/reflora/floradobrasil/FB20891. Accessed from 4 Jun 2021
Dafni A, Kevan PG, Husband BC (2005) Practical pollination biology. Ontario, Cambridge
Dellaporta SL, Calderon-Urrea A (1993) Sex determination in flowering plants. Plant Cell 5:1241–1251. https://doi.org/10.1105/tpc.5.10.1241
Diggle PK, Di Stilio VS, Gschwend AR, Golenberg EM, Moore RC, Russell JRW, Sinclair JP (2011) Multiple developmental processes underlie sex differentiation in angiosperms. Trends Genet 27:368–376. https://doi.org/10.1016/j.tig.2011.05.003
Endress PK, Matthews ML (2005) Elaborate petals and staminodes in eudicots: diversity, function, and evolution. Org Divers Evol 6:257–293. https://doi.org/10.1016/j.ode.2005.09.005
Faegri K, van der Pijl L (1979) The principles of pollination ecology. Pergamon Press, London
Feder N, O’Brien TP (1968) Plant microtechnique: some principles and new methods. Am J Bot 55:123–142. https://doi.org/10.1002/j.1537-2197.1968.tb06952.x
Ferreira DL (2010) Interações entre Cupania vernalis Camb. (Sapindaceae) e insetos antófilos em fragmentos florestais no sul do Brasil. Dissertation, Pontifícia Universidade Católica do Rio Grande do Sul, Porto Alegre
Gabe M (1968) Techiniques histologiques. Masson, Paris
Gahan PB (1984) Plant histochemistry and cytochemistry: an introduction. Academic Press Inc., London
González VV, Solís SM, Ferrucci MS (2014) Anatomía reproductiva en flores estaminadas y pistiladas de Allophylus edulis (Sapindaceae). Bol Soc Argent Bot 49:207–216
González VV, Solís SM, Ferrucci MS (2017) Embryological studies of Magonia pubescens (Dodonaeaeae, Sapindaceae): development of male and female gametophytes in both floral morphs and its phylogenetic implications. Aust Syst Bot 30:279–289. https://doi.org/10.1071/SB17021
González-Melendi P, Uyttewaal M, Morcillo CN, Mora JRH, Fajardo S, Budar F, Lucas MM (2008) A light and electron microscopy analysis of the events leading to male sterility in Ogu-INRA CMS of rapeseed (Brassica napus). J Exp Bot 59:827–838. https://doi.org/10.1093/jxb/erm365
Haddad IVN, Santiago-Fernandes LDR, Machado SR (2018) Autophagy is associated with male sterility in pistillate flowers of Maytenus obtusifolia (Celastraceae). Aust J Bot 66:108–115. https://doi.org/10.1071/BT17174
Haddad IVN, Sá-Haiad B, Santiago-Fernandes LDR, Machado SR (2019) Megagametophyte development and female sterility in Maytenus obtusifolia Mart. (Celastraceae). Protoplasma 256:1667–1680. https://doi.org/10.1007/s00709-019-01413-y
Kawanabe T, Ariizumi T, Kawai-Yamada M, Uchimiya H, Toriyama K (2006) Abolition of the tapetum suicide program ruins microsporogenesis. Plant Cell Physiol 47:784–787. https://doi.org/10.1093/pcp/pcj039
Langeron M (1949) Précis de microscopie. Masson et Cie, Paris
Lima HA, Somner GV, Giulietti AM (2016) Duodichogamy and sex lability in Sapindaceae: the case of Paullinia weinmanniifolia. Plant Syst Evol 302:109–120. https://doi.org/10.1007/s00606-015-1247-5
Martin FW (1959) Staining and observing pollen tubes in the style by means of fluorescence. Stain Technol 34:125–128. https://doi.org/10.3109/10520295909114663
Pannell JR (2017) Plant sex determination. Curr Biol 27:R191–R197. https://doi.org/10.1016/j.cub.2017.01.052
Rathcke BJ, Kass LB (2003) Temporal dioecy in Three Fingers, Thouinia discolor (Sapindaceae), a medicinal shrub endemic to the Bahamas. In: Smith DL, Smith S (eds) Proceedings of the Ninth Symposium on the Natural History of the Bahamas. San Salvador, Bahamas, pp 28–33
Renner SS, Beenken L, Grimm GW, Kocyan A, Ricklefs RE (2007) The evolution of dioecy, heterodichogamy, and labile sex expression in Acer. Evolution 61:2701–2719. https://doi.org/10.1111/j.1558-5646.2007.00221.x
Solís SM, Galati B, Ferrucci MS (2010) Microsporogenesis and microgametogenesis of Cardiospermum grandiflorum and Urvillea chacoensis (Sapindaceae, Paullinieae). Aust J Bot 58:597–604. https://doi.org/10.1071/BT10162
Somner GV, Carvalho ALG, Siqueira CT (2009a) Sapindaceae da restinga da Marambaia, Rio de Janeiro, Brasil. Rodriguesia 60:485–507. https://doi.org/10.1590/2175-7860200960303
Somner GV, Ferrucci MS, Rosa MMT (2009b) Cupania. In: Somner GV (coord) Sapindaceae. In: Wanderley MGL, Shepherd GJ, Melhem TS, Giulietti AM, Martins SE (coords). Flora Fanerogâmica do Estado de São Paulo 6 In: Martins SE, Wanderley MGL, Shepherd GJ, Giulietti AM, Melhem TS (eds). Instituto de Botânica/ Fapesp/Imprensa Oficial, São Paulo, pp 202–207
Statistica for Windows: Version 8.0 (2008) Statsoft Institute Corporation, Tulsa
Taboga SR, Vilamaior PSL (2013) Citoquímica. In: Carvalho HF, Recco-Pimentel SM (eds) A célula. Manole, Barueri, pp 42–50
Weckerle CS, Rutishauser R (2005) Gynoecium, fruit and seed structure of Paullinieae (Sapindaceae). Bot J Linn Soc 147:159–189. https://doi.org/10.1111/j.1095-8339.2005.00365.x
Welzen PC (1989) Guioa Cav. (Sapindaceae): taxonomy, phylogeny, and historical biogeography. Leiden Bot Ser 12:1–314
Wu H, Cheung AY (2000) Programmed cell death in plant reproduction. In: Lam E, Fukuda H, Greenberg J (eds) Programmed cell death in higher plants. Springer, Dordrecht
Yadav N, Pandey AK, Bhatnagar AK (2018) Comparative anther and pistil anatomy of three flowering morphs of andromonoecious Acer oblongum (Sapindaceae s.l.) and its adaptive significance. Nord J Bot 36:1–9. https://doi.org/10.1111/njb.01572
Zhou QY, Liu GS (2012) The embryology of Xanthoceras and its phylogenetic implications. Plant Syst Evol 298:457–468. https://doi.org/10.1007/s00606-011-0558-4
Zini LM, Galati GB, Solís SM, Ferrucci MS (2012) Anther structure and pollen development in Melicoccus lepidopetalus (Sapindaceae): an evolutionary approach to dioecy in the family. Flora 207:712–720. https://doi.org/10.1016/j.flora.2012.07.003
Acknowledgements
We are grateful to Dr. Simon Mayo for valuable comments on the manuscript and linguistic editing. This study forms part of the master’s thesis of VCS, which was carried out in the Programa de Pós-graduação em Ciências Biológicas (Botânica), Museu Nacional, Universidade Federal do Rio de Janeiro (UFRJ).
Funding
RASS was supported by PIBIC (Programa Institucional de Bolsas de Iniciação Científica) granted by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). BSH was supported by research grant from the Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ) (Proc. E-26/200.088/2019).
Author information
Authors and Affiliations
Contributions
BSH, GVS and VCS contributed to the study conception and design. Material preparation, data collection and analysis were performed by VCS and RASS. The first draft of the manuscript was written by VCS and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
dos Santos, V.C., Silva, R.d., Somner, G.V. et al. Floral anatomy of Cupania emarginata, a duodichogamous tree. Braz. J. Bot 45, 463–483 (2022). https://doi.org/10.1007/s40415-021-00758-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40415-021-00758-0