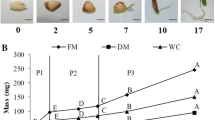
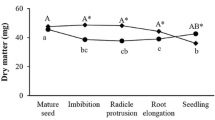
Abstract
Due to the economic importance of wood production, species including Cedrela fissilis Vell. and Cariniana legalis (Mart.) Kuntze have been heavily exploited and are currently threatened with extinction. In addition, these species produce seeds with low viability when stored for long periods. In this work, the effect of storage time on seedling emergence and alterations in the levels of biomolecules such as carbohydrates, polyamines (PAs), and amino acids were analyzed in C. fissilis and C. legalis seeds. Seedling emergence and the emergence speed index were significantly reduced during seed storage in both species, with a greater reduction for C. legalis seeds. Sucrose was detected in both species, with the highest levels in C. fissilis seeds, whereas the monosaccharides glucose and fructose were detected only in C. legalis seeds. Higher levels of putrescine (Put) were observed in C. legalis seeds than in C. fissilis seeds. In contrast, C. fissilis seeds showed a significant increase in most of the amino acids analyzed. These results suggest that the presence of glucose and fructose, the higher level of Put, and the lower levels of amino acids observed in C. legalis seeds may be associated with greater reductions in vigor and seedling emergence compared with C. fissilis seeds. This is the first report showing these relationships among carbohydrates, PAs, and amino acids in these species and may be useful for further studies on seed conservation of timber species.





Similar content being viewed by others
References
Abbade LC, Takaki M (2014) Biochemical and physiological changes of Tabebuia roseoalba (Ridl.) Sandwith (Bignoniaceae) seeds under storage. J Seed Sci 36:100–107
Aguiar FFA, Tavares AR, Kanashiro S, Luz PB, Santos Júnior NA (2010) Germination of Dalbergia nigra (Vell.) Allemao ex Benth. (Fabaceae-Papilionoideae) during storage. Sci Agrotec 34:1624–1629
Akashi K, Miyake C, Yokota A (2001) Citrulline, a novel compatible solute in drought-tolerant wild watermelon leaves, is an efficient hydroxyl radical scavenger. FEBS Lett 508:438–442
Alcázar R, Altabella T, Marco F, Bortolotti C, Reymond M, Koncz C, Carrasco P, Tiburcio AF (2010) Polyamines: molecules with regulatory functions in plant abiotic stress tolerance. Planta 231:1237–1249
Alves EU, Silva KB, Bruno RLA, Alves AU, Cardoso EA, Gonçalves EP, Braz MSS (2008) Comportamento fisiológico de sementes de pitombeira [Talisia esculenta (A. ST. Hil) Radlk] submetidas à desidratação. Rev Bras Frutic 30:509–516
Antunes CGC, Pelacani CR, Ribeiro RC, Gomes HLR, Castra RD (2010) Influência do armazenamento na qualidade fisiológica de sementes de Caesalpinia pyramidalis Tul. Rev Árvore 34:1001–1008
Aragão VPM, Navarro BV, Passamani LZ, Macedo AF, Floh EIS, Silveira V, Santa-Catarina C (2015) Free amino acids, polyamines, soluble sugars and proteins during seed germination and early seedling growth of Cedrela fissilis Vellozo (Meliaceae), an endangered hardwood species from the Atlantic Forest in Brazil. Theor Exp Plant Physiol 27:157–169
Astarita LV, Floh EI, Handro W (2003a) Changes in IAA, tryptophan and activity of soluble peroxidase associated with zygotic embryogenesis in Araucaria angustifolia (Brazilian pine). Plant Growth Regul 39:113–118
Astarita LV, Handro W, Floh EI (2003b) Changes in polyamines content associated with zygotic embryogenesis in the Brazilian pine, Araucaria angustifolia (Bert.) O. Ktze. Braz J Bot 26:163–168
Azevedo RA (2002) Analysis of the aspartic acid metabolic pathway using mutant genes. Amino Acids 22:217–230
Azevedo RA, Lancien M, Lea PJ (2006) The aspartic acid metabolic pathway, an exciting and essential pathway in plants. Amino Acids 30:143–162
Baron K, Stasolla C (2008) The role of polyamines during in vivo and in vitro development. In Vitro Cell Dev Biol Plant 44:384–395
Benedito CP, Ribeiro MCC, Torres SB, Camacho RGV, Soares ANR, Guimarães LMS (2011) Storage of Catanduva seed (Piptadenia moniliformis Benth.) in different environments and packaging. Rev Bras Sementes 33:28–37
Bernal-Lugo I, Leopold AC (1992) Changes in soluble carbohydrates during seed storage. Plant Physiol 98:1207–1210
Bewley JD, Black M (1994) Seeds: physiology of development and germination. Plenum Press, New York
Bezold TN, Loy JB, Minocha SC (2003) Changes in the cellular content of polyamines in different tissues of seed and fruit of a normal and a hull-less seed variety of pumpkin during development. Plant Sci 164:743–752
Borges E, Castro J, Borges R (1990) Avaliação fisiológica de sementes de cedro submetidas ao envelhecimento precoce. Rev Bras Sementes 12:56–62
Cabral EL, Barbosa DCA, Simabukuro EA (2003) Armazenamento e germinação de sementes de Tabebuia aurea (Manso) Benth. & Hook. f. ex. S. Moore. Acta Bot Bras 17:609–617
Carvalho PER (2003) Espécies arbóreas brasileiras. EMBRAPA: Informação Tecnológica, Colombo
Carvalho LR, Silva EAA, Davide AC (2006) Classificação de sementes florestais quanto ao comportamento no armazenamento. Rev Bras Sementes 28:15–25
Carvalho LR, Davide AC, Silva EAA, Carvalho MLM (2008) Classificação de sementes de espécies florestais dos gêneros Nectandra e Ocotea (Lauraceae) quanto ao comportamento no armazenamento. Rev Bras Sementes 30:1–9
Chandrashekar KR (2012) Effect of desiccation on viability and biochemical changes in Knema attenuata seeds. J For Res 23:703–706
Chen Y, Burris JS (1990) Role of carbohydrates in desiccation tolerance and membrane behavior in maturing maize seed. Crop Sci 30:971–975
Chen M, Chory J, Fankhauser C (2004) Light signal transduction in higher plants. Annu Rev Genet 38:87–117
Corte VB, Lima EE, Borges HGL, de Almeida Leite IT (2010) Qualidade fisiológica de sementes de Melanoxylon brauna envelhecidas natural e artificialmente. Sci Agric 38:181–189
Curien G, Biou V, Mas-Droux C, Robert-Genthon M, Ferrer J-L, Dumas R (2008) Amino acid biosynthesis: new architectures in allosteric enzymes. Plant Physiol Biochem 46:325–339
Dias L, Santa-Catarina C, Silveira V, Pieruzzi F, Floh E (2009) Polyamines, amino acids, IAA and ABA contents during Ocotea catharinensis seed germination. Seed Sci Technol 37:42–51
DiTomaso JM, Shaff JE, Kochian LV (1989) Putrescine-induced wounding and its effects on membrane integrity and ion transport processes in roots of intact corn seedlings. Plant Physiol 90:988–995
Dutra NT, Silveira V, de Azevedo IG, Gomes Neto LR, Facanha AR, Steiner N, Guerra MP, Floh EIS, Santa-Catarina C (2013) Polyamines affect the cellular growth and structure of pro-embryogenic masses in Araucaria angustifolia embryogenic cultures through the modulation of proton pump activities and endogenous levels of polyamines. Physiol Plant 148:121–132
Edreva A, Velikova V, Tsonev T (2007) Phenylamides in plants. Russ J Plant Physiol 54:287–301
Engqvist M, Drincovich MF, Flugge UI, Maurino VG (2009) Two D-2-hydroxy-acid dehydrogenases in Arabidopsis thaliana with catalytic capacities to participate in the last reactions of the methylglyoxal and beta-oxidation pathways. J Agric Food Chem 284:25026–25037
Fariduddin Q, Varshney P, Yusuf M, Ahmad A (2013) Polyamines: potent modulators of plant responses to stress. J Plant Interact 8:1–16
Ferreira CS, Piedade MTF, Tiné MAS, Rossatto DR, Parolin P, Buckeridge MS (2009) The role of carbohydrates in seed germination and seedling establishment of Himatanthus sucuuba, an Amazonian tree with populations adapted to flooded and non-flooded conditions. Ann Bot 104:1111–1119
Filson PB, Dawson-Andoh BE (2009) Characterization of sugars from model and enzyme-mediated pulp hydrolyzates using high-performance liquid chromatography coupled to evaporative light scattering detection. Bioresource Technol 100:6661–6664
Fundação SOS Mata Atlântica (2014) Relatório anual. https://www.sosma.org.br/link/relatorio_anual_sosma_2014/index.html. Accessed 25 April 2015
Galili G, Tang G, Zhu X, Gakiere B (2001) Lysine catabolism: a stress and development super-regulated metabolic pathway. Curr Opin Plant Biol 4:261–266
Garcia I, Souza A, Barbedo C, Dietrich S, Figueiredo-Ribeiro R (2006) Changes in soluble carbohydrates during storage of Caesalpinia echinata Lam. (Brazilwood) seeds, an endangered leguminous tree from the Brazilian Atlantic Forest. Braz J Biol 66:739–745
Guedes R, Alves E, Bruno R, Gonçalves E, Costa E, Medeiros M (2012) Armazenamento de sementes de Myracrodruon urundeuva Fr. All. em diferentes embalagens e ambientes. Rev Bras Plantas Med 14:68–75
Hellmann ME, Mello J, Barbedo CJ, Figueiredo-Ribeiro R (2008) Variações dos carboidratos de reserva de sementes de Caesalpinia echinata (pau-brasil) armazenadas sob diferentes temperaturas. Hoehnea 35:255–264
Hoekstra FA, Golovina EA, Buitink J (2001) Mechanisms of plant desiccation tolerance. Trends Plant Sci 6:431–438
Horbowicz M, Brenac P, Obendorf RL (1998) Fagopyritol B1, O-α-d-galactopyranosyl-(1 → 2)-d-chiro-inositol, a galactosyl cyclitol in maturing buckwheat seeds associated with desiccation tolerance. Planta 205:1–11
IUCN (2015) International Union for Conservation of Nature. The Red List of Threatened species http://www.iucnredlist.org/about. Accessed 25 April 2015
Johnson TR, Kane ME, Pérez HE (2011) Examining the interaction of light, nutrients and carbohydrates on seed germination and early seedling development of Bletia purpurea (Orchidaceae). Plant Growth Regul 63:89–99
José SCBR, Von Pinho EVR, Dias MAGS (2006) Açúcares e tolerância à alta temperatura de secagem sementes de milho. Rev Bras Sementes 28:60–68
Kapoor N, Arya A, Siddiqui MA, Kumar H, Amir A (2011) Physiological and biochemical changes during seed deterioration in aged seeds of rice (Oryza sativa L.). Am J Plant Physiol 6:28–35
Kirma M, Araújo WL, Fernie AR, Galili G (2012) The multifaceted role of aspartate-family amino acids in plant metabolism. J Exp Bot 63:4995–5001
Koster KL, Leopold AC (1988) Sugars and desiccation tolerance in seeds. Plant Physiol 88:829–832
Krohn NG, Malavasi MM (2004) Qualidade fisiológica de sementes de soja tratadas com fungicidas durante e após o armazenamento. Rev Bras Sementes 26:91–97
Kuo TM, VanMiddlesworth JF, Wolf WJ (1988) Content of raffinose oligosaccharides and sucrose in various plant seeds. J Agric Food Chem 36:32–36
Kusano T, Berberich T, Tateda C, Takahashi Y (2008) Polyamines: essential factors for growth and survival. Planta 228:367–381
Lea PJ, Miflin BJ (2003) Glutamate synthase and the synthesis of glutamate in plants. Plant Physiol Biochem 41:555–564
Leprince O, Werf A, Deltour R, Lambers H (1992) Respiratory pathways in germinating maize radicles correlated with desiccation tolerance and soluble sugars. Physiol Plant 85:581–588
Limami AM, Rouillon C, Glevarec G, Gallais A, Hirel B (2002) Genetic and physiological analysis of germination efficiency in maize in relation to nitrogen metabolism reveals the importance of cytosolic glutamine synthetase. Plant Physiol 130:1860–1870
Lin TP, Huang NH (1994) The relationship between carbohydrate composition of some tree seeds and their longevity. J Exp Bot 45:1289–1294
Maguire JD (1962) Speed of germination-aid in selection and evaluation for seedling emergence and vigor. Crop Sci 2:176–177
Mapelli S, Brambilla I, Bertani A (2001) Free amino acids in walnut kernels and young seedlings. Tree Physiol 21:1299–1302
Marcos Filho J (2005) Fisiologia de sementes de plantas cultivadas. Fealq, Piracicaba
Martinelli G, Moraes MA (2013) Livro vermelho da flora do Brasil. Instituto de Pesquisa Jardim Botânico, Rio de Janeiro
Medeiros A, Eira M (2006) Comportamento fisiológico, secagem e armazenamento de sementes florestais nativas. Comun Téc EMBRAPA 127:1–13
Minocha R, Smith DR, Reeves C, Steele KD, Minocha SC (1999) Polyamine levels during the development of zygotic and somatic embryos of Pinus radiata. Physiol Plant 105:155–164
Myers N, Mittermeier R, Mittermeier C, Da Fonseca G, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Obendorf RL (1997) Oligosaccharides and galactosyl cyclitols in seed desiccation tolerance. Seed Sci Res 7:63–74
Oliveira I, Brenner E, Chiu J, Hsieh M, Kouranov A, Lam H, Shin M, Coruzzi G (2001) Metabolite and light regulation of metabolism in plants: lessons from the study of a single biochemical pathway. Braz J Med Biol Res 34:567–575
Phartyal SS, Thapliyal R, Koedam N, Godefroid S (2002) Ex situ conservation of rare and valuable forest tree species through seed-gene bank. Curr Sci 83:1351–1357
Pieruzzi FP, Dias LLC, Balbuena TS, Santa-Catarina C, Santos ALW, Floh EIS (2011) Polyamines, IAA and ABA during germination in two recalcitrante seeds: Araucaria angustifolia (Gymnosperm) and Ocotea odorifera (Angiosperm). Ann Bot 108:337–345
Santa-Catarina C, Silveira V, Balbuena TS, Viana AM, Estelita MEM, Handro W, Floh EI (2006) IAA, ABA, polyamines and free amino acids associated with zygotic embryo development of Ocotea catharinensis. Plant Growth Regul 49:237–247
Sasaki K, Takahashi H, Kojima S, Ushimiya H, Sato T (2013) Metabolic alteration in free amino acid contentes of rice (Oryza sativa L.) seeds during high humidity storage. Res J Seed Sci 6:16–23
Shevyakova N, Shorina M, Rakitin VY, Kuznetsov VV (2006) Stress-dependent accumulation of spermidine and spermine in the halophyte Mesembryanthemum crystallinum under salinity conditions. Russ J Plant Physiol 53:739–745
Shibata M, Coelho CMM, Oliveira LM, Garcia C (2012) Accelerated aging of ipê seeds under controlled conditions of storage. J Seed Sci 34:247–254
Shoeb F, Yadav J, Bajaj S, Rajam M (2001) Polyamines as biomarkers for plant regeneration capacity: improvement of regeneration by modulation of polyamine metabolism in different genotypes of indica rice. Plant Sci 160:1229–1235
Silveira V, Balbuena TS, Santa-Catarina C, Floh EI, Guerra MP, Handro W (2004) Biochemical changes during seed development in Pinus taeda L. Plant Growth Regul 44:147–156
Silveira V, Santa-Catarina C, Tun NN, Scherer GF, Handro W, Guerra MP, Floh EI (2006) Polyamine effects on the endogenous polyamine contents, nitric oxide release, growth and differentiation of embryogenic suspension cultures of Araucaria angustifolia (Bert.) O. Ktze. Plant Sci 171:91–98
Silveira V, Vita AM, Macedo AF, Dias MFR, Floh EIS, Santa-Catarina C (2013) Morphological and polyamine content changes in embryogenic and non-embryogenic callus of sugarcane. Plant Cell Tiss Org Cult 114:351–364
Smeekens S, Ma J, Hanson J, Rolland F (2010) Sugar signals and molecular networks controlling plant growth. Curr Opin Plant Biol 13:273–278
Souza VC, Bruno RLA, Andrade LA (2005) Vigor de sementes armazenadas de ipê-amarelo Tabebuia serratifolia (Vahl.) Nich. Rev Árvore 29:833–841
Stepansky A, Less H, Angelovici R, Aharon R, Zhu X, Galili G (2006) Lysine catabolism, an effective versatile regulator of lysine level in plants. Amino Acids 30:121–125
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant Sci 151:59–66
Veselova TV, Veselovsky VA, Obroucheva NV (2015) Deterioration mechanisms in air-dry pea seeds during early aging. Plant Physiol Biochem 87:133–139
Vieira FA, Gusmão E (2008) Biometria, armazenamento de sementes e emergência de plântulas de Talisia esculenta Radlk. (Sapindaceae). Ciênc Agrotec 32:1073–1079
Yamaguchi K, Takahashi Y, Berberich T, Imai A, Takahashi T, Michael AJ, Kusano T (2007) A protective role for the polyamine spermine against drought stress in Arabidopsis. Biochem Biophys Res Commun 352:486–490
Zhu J-K (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53:247–273
Acknowledgments
This research was supported by the Carlos Chagas Filho Foundation for Research Support in the State of Rio de Janeiro—FAPERJ (E26/111.389-2012, E26/102.989/2012, and E26/010.001507/2014), and the National Council for Scientific and Technological Development—CNPq (305645/2013-7 and 444453/2014-8). Scholarships were granted by the Coordination for the Improvement of Higher Education Personnel—CAPES to KRS, RSR, and VPMA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sousa, K.R., Aragão, V.P.M., Reis, R.S. et al. Polyamine, amino acid, and carbohydrate profiles during seed storage of threatened woody species of the Brazilian Atlantic Forest may be associated with seed viability maintenance. Braz. J. Bot 39, 985–995 (2016). https://doi.org/10.1007/s40415-016-0293-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40415-016-0293-8