Abstract
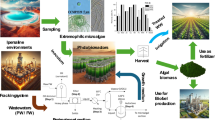
Three marine microalgae Chaetoceros muelleri Lemmerman, Dunaliella salina (Dunal) Teod., and Arthrospira platensis (Gomont) Geitler were grown in their respective medium under laboratory conditions. Harvested biomass of all the three algae was grown in salt refinery effluent, to reduce or eliminate the hazardous chemicals for a period of 21, 18, and 20 days, respectively. The initial and final levels of hazardous chemicals (Cu, Zn, P, K, Na, Cl, Fe, F, Mg, SO4 and BOD, COD) in the effluents were analyzed according to APHA and their results were recorded. Heavy metals such as barium, aluminum, silver, strontium, and cadmium were identified by Inductively coupled plasma atomic emission spectroscopy. Among three microalgae, A. platensis was unable to survive after 3 days due to the presence of harmful chemicals in the effluent. The effect of Seaweed liquid fertilizers (SLF) extracted from Kappaphycus alvarezii (Doty) Doty ex P. C. Silva var. tambalang (Doty) and Turbinaria conoides (J. Agardh) Kützing were examined on the two microalgal species except A. platensis. Extracts were added in four different percentages such as 0.4, 0.9, 1.3, and 1.7 % to C. muelleri and D. salina. The changes in the microalgal cells and SLFs were analyzed under confocal microscopy.

Similar content being viewed by others
References
APHA (American Public Health Administration) (2005) 21st Edition, New York, USA
Arnon DI (1949) Copper enzymes in isolated chloroplast polyphenoloxidase in beta vulgaris. Plant Physiol 24:1–15
Booth E (1969) The manufacture and properties of liquid seaweed extracts. Proceedings of 6th International Seaweed Symposium. pp. 655-662
Cho IY, Jin HJ, Lim HJ, Whyte JNC, Hong YK (1998) Growth activation of the microalga, Isochrysis galbana by the aqueous extract of the seaweed Monostroma nitidum. J Appl Phycol 10:561–567
Clarens AF, Resurreccion EP, White MA, Colosi LM (2010) Environmental life cycle comparison of algae to other bioenergy feedstocks. Environ Sci Technol 44:1813–1819
Fereshteh G, Yassaman B, Reza AMM, Zavar A, Hossein M (2007) Phytoremediation of arsenic by macroalga: implication in natural contaminated water. North East Iran J Appl Sci 7:1614–1619
Fisher RA (1990) Statistical Methods. Experimental Design and Scientific Interence. Oxford University Press, Oxford
Fornes F, Sa`nchez-perales M, Guadiola JL (2002) Effect of a seaweed extract on the productivity of ‘de Nules’ clementine mandarin and navelina orange. Bot Mar 45:486–489
Forsberg A, Soderlund S, Frank A, Peterson LR, Pedersen M (1988) Studies on metal content in the brown seaweed, Fucus vesiculosus from the Archipelago of Stockholm. Environ Poll 49:245–263
Franqueira D, Orosa M, Torres E, Herrero C, Cid A (2000) Potential use of flow cytometry in toxicity studies with microalgae. Science Total Environ 247:119–126
Gerasimenko LM, Mikhodyuk OS (2009) Halophilic algal-bacterial and cyanobacterial communities and their role in carbonate precipitation. Paleontol J 43:940–957
Gorden SA, Paleg LG (1957) Quantitative measurement of indole acetic acid. Plant Physiol 10:37–48
Guillard RRL (1975) Culture of phytoplankton for feeding marine invertebrates. In: Smith WL, Chanley MH (eds) culture of marine invertebrate animals. Plenum press, New York, pp 26–60
Harrison PJ, Hurd CL (2001) Nutrient physiology of seaweeds: application of concepts to aquaculture. Cah Biol Mar 42:71–82
Hong YP, Chen CC, Cheng HL, Lin CH (1995) Analysis of auxin and cytokinin activity of commercial aqueous seaweed extract. Gartenbauwissenschaft 60:191–194
Lin S, Gobler CJ, Carpenter EJ (2001) Cytological and biochemical responses of Dunaliella tertiolecta (Volvocales, Chlorophyta) to iron stress. Phycologia 40:403–410
Mary Leema JT, Kirubagaran R, Vinithkumar NV, Dheenan PS, Karthikayulu S (2010) High value pigment production from Arthrospira (Spirulina) platensis cultured in seawater. Bioresour Technol 101:9221–9227
Not F, Simon N, Biegala IC, Vaulot D (2002) Application of fluorescent in situ hybridization coupled with tyramide signal amplification (FISHTSA) to assess eukaryotic picoplankton composition. Aquat Microb Ecol 28:157–166
Orset S, Young AJ (1999) Low-temperature induced synthesis of a-carotene in the microalga, Dunaliella salina (Chlorophyta). J Phycol 35:527–549
Pittman JK, Dean AP, Osundeko O (2011) The potential of sustainable algal biofuel production using wastewater resources. Biores Technol 102:17–25
Raja R (2003) Studies on Dunaliella salina (Dunal) Teod. with special reference to its anticancer properties. Ph.D. Thesis, University of Madras, Chennai, India
Raja R, Anbazhagan C, Ganesan V, Rengasamy R (2004a) Efficacy of Dunaliella salina (Volvocales, Chlorophyta) in Salt effluent treatment. Asian J Chem 16:1081–1088
Raja R, Anbazhagan C, Lakshmi D, Rengasamy R (2004b) Nutritional studies on Dunaliella salina (Volvocales, Chlorophyta) under laboratory conditions. Seaweed Res Utili 26:127–146
Raja R, Hemaiswarya S, Balasubramanyam D, Ramasamy R (2007a) Protective effect of Dunaliella salina (Volvocales, Chlorophyta) on experimentally induced fibrosarcoma on wistar rats. Microbiol Res 162:177–184
Raja R, Hemaiswarya S, Balasubramanyam D, Rengasamy R (2007b) PCR identification of Dunaliella salina (Volvocales, Chlorophyceae) and its growth characteristics. Microbiol Res 162:168–176
Reddy CRK, Bhavanath J, Yuji F, Masao O (2009) Seaweed micropropagation techniques and their potentials: an overview. Dev Appl Phycol Ninet Inter Seaweed Symp 2:159–167
Senthil C, Ashish B, Ryan WH, Das KC (2010) Microalgae cultivation in a wastewater dominated by carpet mill effluents for biofuel applications. Bioresour Technol 101:3097–3105
Shi J, Podola B, Melkonian M (2007) Removal of nitrogen and phosphorus from wastewater using microalgae immobilized on twin layers: an experimental study. J Appl Phycol 19:417–423
Sridhar S, Rengasamy R (2002) Effect of seaweed liquid fertilizer obtained from Ulva lactuca on the biomass, pigments and protein content of Spirulina platensis. Sea Res Utili 24:145–149
Syono K, Torrey JG (1976) Identification of cytokinins of root nodules of the garden pea, Pisum sativum. Plant Physiol 57:602–606
Wijffels RH, Barbosa MJ, Eppink MHM (2010) Microalgae for the production of bulk chemicals and biofuels. Biofuels Bioprod Bioref 4:287–295
Zodape ST (2001) Seaweeds as a biofertilizer. J Sci Ind Res (India) 60:378–382
Acknowledgments
The authors are grateful to Foundation for Science and Technology (FCT), Portugal for funding the researcher, Rathinam Raja.
Conflict of interest
Authors do not have any conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Rathinam Raja and Shanmugam Hemaiswarya contributed equally to this work.
Rights and permissions
About this article
Cite this article
Raja, R., Hemaiswarya, S., Carvalho, I.S. et al. Impact of industrial salt effluent and seaweed liquid fertilizers on three microalgae. Braz. J. Bot 38, 547–553 (2015). https://doi.org/10.1007/s40415-015-0170-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40415-015-0170-x