Abstract
Purpose
The present systematic review aims to describe the incidence and severity of chemotherapy-related oral mucositis in children and adolescents affected by hematologic and solid tumors.
Methods
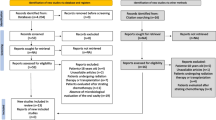
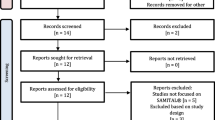
An electronic search was performed on PubMed, Scopus, Web of Science, Cochrane Library, and EBSCO up to the 8th November 2020. The PRISMA protocol was followed for the article selection and inclusion. The risk of bias in individual studies was evaluated through the Newcastle–Ottawa Scale. Data were summarized using mean and standard deviation for continuous variables, while categorical ones were described with frequency and percentage.
Results
A number of 9940 records were obtained after the electronic search. Seventeen of them were included in the qualitative analysis after the two stages of screening, while none of these articles was considered eligible for the quantitative analysis. The mean incidence of oral mucositis was 53.6% and it ranged from 16.7 to 91.5%, while severe oral mucositis accounted for the 15.8% (0.0–35.2%) among selected studies. Most of the articles included both patients with solid and hematologic tumors, while only five of them described oral mucositis in children with acute lymphoblastic leukemia. Even the kinds of chemotherapy administered were extremely variable.
Conclusion
In conclusion, about half of the patients submitted to cancer chemotherapy developed oral mucositis with an incidence and severity that varies depending on the primary disease and the kind of drugs administered.

Similar content being viewed by others
Availability of data and materials
Not applicable.
Code availability
Not applicable.
References
Allen G, Logan R, Revesz T, Keefe D, Gue S. The prevalence and investigation of risk factors of oral mucositis in a pediatric oncology inpatient population; a prospective study. J Pediatr Hematol Oncol. 2017;40(1):15–21. https://doi.org/10.1097/mph.0000000000000970.
Attinà G, Romano A, Maurizi P, D’Amuri S, Mastrangelo S, Capozza MA, Triarico S, Ruggiero A. Management of oral mucositis in children with malignant solid tumors. Front Oncol. 2021;11: 599243. https://doi.org/10.3389/fonc.2021.599243.
Azher U, Shiggaon N. Oral health status of children with acute lymphoblastic leukemia undergoing chemotherapy. Ind J Dent Res. 2013;24(4):523. https://doi.org/10.4103/0970-9290.118371.
Bartelink IH, Bredius RG, Belitser SV, Suttorp MM, Bierings M, Knibbe CA, Egeler M, Lankester AC, Egberts AC, Zwaveling J, Boelens JJ. Association between busulfan exposure and outcome in children receiving intravenous busulfan before hematologic stem cell transplantation. Biol Blood Marrow Transplant. 2009;15(2):231–41. https://doi.org/10.1016/j.bbmt.2008.11.022.
Carreón-Burciaga RG, Castañeda-Castaneira E, González-González R, Molina-Frechero N, Gaona E, Bologna-Molina R. Severity of oral mucositis in children following chemotherapy and radiotherapy and its implications at a single oncology centre in durango state. Mexico Int J Pediatr. 2018;2018:3252765. https://doi.org/10.1155/2018/3252765.
Cheng KK, Molassiotis A, Chang AM, Wai WC, Cheung SS. Evaluation of an oral care protocol intervention in the prevention of chemotherapy-induced oral mucositis in paediatric cancer patients. Eur J Cancer. 2001;37(16):2056–63. https://doi.org/10.1016/s0959-8049(01)00098-3.
Cheng KK, Chang AM, Yuen MP. Prevention of oral mucositis in paediatric patients treated with chemotherapy; a randomised crossover trial comparing two protocols of oral care. Eur J Cancer. 2004;40(8):1208–16. https://doi.org/10.1016/j.ejca.2003.10.023.
Cheng KK, Goggins WB, Lee VW, Thompson DR. Risk factors for oral mucositis in children undergoing chemotherapy: a matched case-control study. Oral Oncol. 2008;44(11):1019–25. https://doi.org/10.1016/j.oraloncology.2008.01.003.
Cheng KK, Lee V, Li CH, Goggins W, Thompson DR, Yuen HL, Epstein JB. Incidence and risk factors of oral mucositis in paediatric and adolescent patients undergoing chemotherapy. Oral Oncol. 2011;47(3):153–62. https://doi.org/10.1016/j.oraloncology.2010.11.019.
Cheng KK, Lee V, Li CH, Yuen HL, Ip WY, He HG, Epstein JB. Impact of oral mucositis on short-term clinical outcomes in paediatric and adolescent patients undergoing chemotherapy. Supp Care Cancer. 2013;21(8):2145–52. https://doi.org/10.1007/s00520-013-1772-0.
de Mendonca RMH, de Araujo M, Levy CE, Morari J, Silva RA, Yunes JA, Brandalise SR. Oral mucositis in pediatric acute lymphoblastic Leukemia patients: evaluation of microbiological and hematological factors. Pediatr Hematol Oncol. 2015;32(5):322–30. https://doi.org/10.3109/08880018.2015.1034819.
El-Housseiny AA, Saleh SM, El-Masry AA, Allam AA. Assessment of oral complications in children receiving chemotherapy. J Clin Pediatr Dent. 2007;31(4):267–73. https://doi.org/10.17796/jcpd.31.4.cq752m6173142r28.
Fadda G, Campus G, Lugliè P. Risk factors for oral mucositis in paediatric oncology patients receiving alkylant chemotherapy. BMC Oral Health. 2006;6:13. https://doi.org/10.1186/1472-6831-6-13.
Figliolia SLC, Oliveira DT, Pereira MC, Lauris JRP, Mauricio AR, Oliveira DT, de Andrea MLM. Oral mucositis in acute lymphoblastic leukaemia: analysis of 169 paediatric patients. Oral Dis. 2008;14(8):761–6. https://doi.org/10.1111/j.1601-0825.2008.01468.x.
Foucher-Estliarova E, Collombet M, Ries LAG, Moreno F, Dolya A, Bray F, Hesseling P, Shin HY, Stiller C. International incidence of Childhood Cancer 2001–10: a population based registry study. Lancet Oncol. 2017;18(6):719–31.
Gandhi K, Datta G, Ahuja S, Saxena T, Datta AG. Prevalence of oral complications occurring in a population of pediatric cancer patients receiving chemotherapy. Int J Clin Pediatr Dent. 2017;10(2):166–71. https://doi.org/10.5005/iD-iournals-10005-1428.
Gussgard AM, Hope AJ, Jokstad A, Tenenbaum H, Wood R. Assessment of cancer therapy-induced oral mucositis using a patient-reported oral mucositis experience questionnaire. PLoS One. 2014;9(3): e91733. https://doi.org/10.1371/journal.pone.0091733.
Hagan J, Shaw J, Duncan P (2017) Bright futures: guidelines for health supervision of infants, children, and adolescents (4th Edition) In.
Maiguma T, Hayashi Y, Ueshima S, Kaji H, Egawa T, Chayama K, Morishima T, Kitamura Y, Sendo T, Gomita Y, Teshima D. Relationship between oral mucositis and high-dose methotrexate therapy in pediatric acute lymphoblastic leukemia. Int J Clin Pharmacol Ther. 2008;46(11):584–90. https://doi.org/10.5414/cpp46584.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA. J Clin Epidemiol. 2009;62(10):1006–12.
Otmani N, Alami R, Hessissen L, Mokhtari A, Soulaymani A, Khattab M. Determinants of severe oral mucositis in paediatric cancer patients: a prospective study. Int J Paediatri Dent. 2011;21(3):210–6. https://doi.org/10.1111/j.1365-263X.2011.01113.x.
Parra JJ, Alvarado MC, Monsalve P, Costa ALF, Montesinos GA, Parra PA. Oral health in children with acute lymphoblastic leukaemia: before and after chemotherapy treatment. Eur Arch Paediatr Dent. 2020;21(1):129–36. https://doi.org/10.1007/s40368-019-00454-4.
Proc P, Szczepańska J, Zubowska M, Wyka K, Młynarski W. Salivary immunoglobulin A level during steroids and chemotherapy treatment administered in remission induction phase among pediatric patients with acute lymphoblastic leukemia. Medicine. 2020;99(42): e22802. https://doi.org/10.1097/md.0000000000022802.
Raber-Drulacher JE, Elad S, Barasch A. Oral mucositis. Oral Oncol. 2010;46:452–6.
Rask C, Albertioni F, Bentzen SM, Schroeder H, Peterson C. Clinical and pharmacokinetic risk factors for high-dose methotrexate-induced toxicity in children with acute lymphoblastic leukemia–a logistic regression analysis. Acta Oncol. 1998;37(3):277–84. https://doi.org/10.1080/028418698429586.
Ribeiro ILA, Melo ACR, Limão NP, Bonan PRF, Lima Neto EA, Valença AMG. Oral mucositis in pediatric oncology patients: a nested case-control to a prospective cohort. Braz Dent J. 2020;31(1):78–88. https://doi.org/10.1590/0103-6440201802881.
Robien K, Schubert MM, Bruemmer B, Lloid ME, Potter JD, Ulrich CM. Predictors of oral mucositis in patients receiving hematopoietic cell transplants for chronic myelogenous leukemia. J Clin Oncol. 2004;22(7):1268–75. https://doi.org/10.1200/jco.2004.05.147.
Rubenstein EB, Peterson DE, Schubert M, Keefe D, McGuire D, Epstein J, Elting LS, Fox PC, Cooksley C, Sonis ST. Clinical practice guidelines for the prevention and treatment of cancer therapy-induced oral and gastrointestinal mucositis. Cancer. 2004;100(S9):2026–46.
Saunders DP, Rouleau T, Cheng K, Yarom N, Kandwal A, Joy J, Bektas Kayhan K, van de Wetering M, Brito-Dellan N, Kataoka T, Chiang K, Ranna V, Vaddi A, Epstein J, Lalla RV, Bossi P, Elad S. Systematic review of antimicrobials, mucosal coating agents, anesthetics, and analgesics for the management of oral mucositis in cancer patients and clinical practice guidelines. Supp Care Cancer. 2020;28(5):2473–84. https://doi.org/10.1007/s00520-019-05181-6.
Sonis ST. Oral mucositis in cance therapy. J Support Oncol. 2004;2:3–8.
Sonis ST. Mucositis: the impact, biology and therapeutic opportunities of oral mucositis. Oral Oncol. 2009;45(12):1015–20.
Sonis S, Clark J. Prevention and management of oral mucositis induced by antineoplastic therapy. Oncology. 1991;5:11–7.
Sonis ST, Elting LS, Keefe D, Peterson DE, Schubert M, Hauer-Jensen M, Bekele BN, Raber-Durlacher J, Donnelly JP, Rubenstein EB, Cancer MSSotMAfSCi, Oncology. ISfO. Perspectives on cancer therapy-induced mucosal injury: pathogenesis, measurement, epidemiology, and consequences for patients. Cancer. 2004;100(9):1995–2025.
Tydings C, Kim A. Technology and precision therapy delivery in childhood cancer. Curr Opin Pediatr. 2020;32(1):1–6.
Valer JB, Curra M, Gabriel AF, Schmidt TR, Ferreira MBC, Roesler R, Evangelista Junior MC, Martins MAT, Gregianin L, Martins MD. Oral mucositis in childhood cancer patients receiving high-dose methotrexate: prevalence, relationship with other toxicities, and methotrexate elimination. Int J Paediatr Dent. 2020. https://doi.org/10.1111/ipd.12718.
Velten DB, Zandonade E, Miotto M. Prevalence of oral manifestations in children and adolescents with cancer submitted to chemotherapy. BMC Oral Health. 2017. https://doi.org/10.1186/s12903-016-0331-8.
Wardill HR, Sonis ST, Blijlevens NMA, Van Sebille YZA, Ciorba MA, Loeffen EAH, Cheng KKF, Bossi P, Porcello L, Castillo DA, Elad S, Bowen JM. Prediction of mucositis risk secondary to cancer therapy: a systematic review of current evidence and call to action. Supp Care Cancer. 2020. https://doi.org/10.1007/s00520-020-05579-7.
Wardley AM, Jayson GC, Swindell R, Morgenstern GR, Chang J, Bloor R, Fraser CJ, Scarffe JH. Prospective evaluation of oral mucositis in patients receiving myeloablative conditioning regimens and haemopoietic progenitor rescue. Br J Haematol. 2000;110(2):292–9. https://doi.org/10.1046/j.1365-2141.2000.02202.x.
Wells GA, Shea B, O'Connell D, Peterson J, Welch V, Losos M, Tugwell P. The Newcastle–Ottawa Scale (NOS) for assessing the quality of non-randomized studies in meta-analysis. Environ Sci. 2000
Funding
No funds, grants, or other support was received.
Author information
Authors and Affiliations
Contributions
BC and DR contributed to the study conception and design. Material preparation, data collection, and analysis were performed by BC. The first draft of the manuscript was written by BC and DR, while BC and AMD participated in the revision process. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethics approval
Not required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Docimo, R., Anastasio, M.D. & Bensi, C. Chemotherapy-induced oral mucositis in children and adolescents: a systematic review. Eur Arch Paediatr Dent 23, 501–511 (2022). https://doi.org/10.1007/s40368-022-00727-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40368-022-00727-5