Abstract
Aim
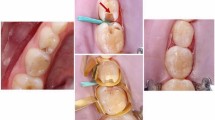
This study was to compare the success of resin-modified Portland cement-based material (TheraCal) with MTA in direct pulp capping (DPC) of primary molars.
Methods
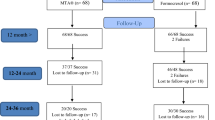
Symmetrical bilateral primary molars (92) from 46 healthy subjects aged 5–7 years were included in this split-mouth randomised clinical trial. DPC for small non-contaminated pulp exposures using either TheraCal or MTA were randomly performed in symmetrical molars. Thereafter, teeth were restored with amalgam. Clinical and radiographic evaluations were performed at 6 and 12 month follow-ups. Data were analysed using Chi square test at a significance level of 0.05.
Results
At the final follow-up session 74 teeth were available. After 12 months, the overall success rates for MTA and TheraCal were 94.5 and 91.8%, respectively. The difference between outcomes of the two groups was not statistically significant (P > 0.05).
Conclusion
Within the limitations of the current study, radiographic and clinical findings revealed that TheraCal exhibited a comparable outcome to MTA in DPC of primary molars after 12 months.

Similar content being viewed by others
References
Aminabadi NA, Huang B, Samiei M, Agheli S, Jamali Z, Shirazi S. A randomized trial using 3Mixtatin compared to MTA in primary molars with inflammatory root resorption: a novel endodontic biomaterial. J Clin Paediatr Dent. 2016;40(2):95–102.
Aminabadi NA, Parto M, Emamverdizadeh P, Jamali Z, Shirazi S. Pulp bleeding color is an indicator of clinical and histohematologic status of primary teeth. Clin Oral Invest. 2017;21(5):1831–41.
Asgary S, Kamrani FA, Taheri S. Evaluation of antimicrobial effect of MTA, calcium hydroxide, and CEM cement. Iran Endod J. 2008;2(3):105–9.
Asl Aminabadi N, Satrab S, Najafpour E, et al. A randomised trial of direct pulp capping in primary molars using MTA compared to 3Mixtatin: a novel pulp capping biomaterial. Int J Paediatr Dent. 2016;26(4):281–90.
Bortoluzzi EA, Niu L-n, Palani CD, et al. Cytotoxicity and osteogenic potential of silicate calcium cements as potential protective materials for pulpal revascularisation. Dent Mater. 2015;31(12):1510–22.
Camilleri J. Characterisation of hydration products of mineral trioxide aggregate. Int Endod J. 2008;41(5):408–17.
Camilleri J. Hydration characteristics of Biodentine and Theracal used as pulp capping materials. Dent Mater. 2014;30(7):709–15.
Cannon M, Gerodias N, Vieira A, Percinoto C, Jurado R. Primate pulpal healing after exposure and TheraCal application. J Clin Pediatr Dent. 2014;38(4):333–7.
Cengiz E, Yilmaz HG. Efficacy of erbium, chromium-doped: yttrium, scandium, gallium, and garnet laser irradiation combined with resin-based tricalcium silicate and calcium hydroxide on direct pulp capping: a randomised clinical trial. J Endod. 2016;42(3):351–5.
Cox C, Sübay R, Ostro E, Suzuki S, Suzuki S. Tunnel defects in dentin bridges: their formation following direct pulp capping. Oper Dent. 1995;21(1):4–11.
Dammaschke T. The history of direct pulp capping. J Hist Dent. 2007;56(1):9–23.
Dammaschke T, Leidinger J, Schäfer E. Long-term evaluation of direct pulp capping—treatment outcomes over an average period of 6.1 years. Clin Oral Investig. 2010;14(5):559–67.
Davidovich E, Wated A, Shapira J, Ram D. The influence of location of local anaesthesia and complexity/duration of restorative treatment on children’s behaviour during dental treatment. Pediatr Dent. 2013;35(4):333–6.
Formosa L, Mallia B, Camilleri J. A quantitative method for determining the anti washout characteristics of cement-based dental materials including mineral trioxide aggregate. Int Endod J. 2013;46(2):179–86.
Fuks AB. Pulp therapy for the primary and young permanent dentitions. Dent Clin N Am. 2000;44(3):571.
Gandolfi MG, Taddei P, Siboni F, et al. Development of the foremost light-curable calcium-silicate MTA cement as root-end in oral surgery. Chemical–physical properties, bioactivity and biological behaviour. Dent Mater. 2011;27(7):e134–e57.
Gandolfi M, Siboni F, Prati C. Chemical–physical properties of TheraCal, a novel light-curable MTA-like material for pulp capping. Int Endod J. 2012;45(6):571–9.
Ghajari MF, Jeddi TA, Iri S, Asgary S. Direct pulp-capping with calcium enriched mixture in primary molar teeth: a randomised clinical trial. Iran Endod J. 2010;5(1):27–30.
Ghajari MF, Jeddi TA, Iri S, Asgary S. Treatment outcomes of primary molars direct pulp capping after 20 months: a randomised controlled trial. Iran Endod J. 2013;8(4):149–52.
Gong V, Franca R. Nanoscale chemical surface characterization of four different types of dental pulp-capping materials. J Dent. 2017;58:11–8.
Ha WN, Kahler B, Walsh LJ. Clinical manipulation of mineral trioxide aggregate: lessons from the construction industry and their relevance to clinical practice. J Can Dent Assoc. 2015;81:f4.
Hebling J, Lessa F, Nogueira I, Carvalho RMD, Costa C. Cytotoxicity of resin-based light-cured liners. Am J Dent. 2009;22(3):137–42.
Hilton T, Ferracane J, Mancl L. Dentistry NP-bRCiE-b. Comparison of CaOH with MTA for direct pulp capping a PBRN randomised clinical trial. J Dent Res. 2013;92(suppl.no.1):16s–22s.
Kennedy D, Kapala J. The dental pulp: biological considerations of protection and treatment. Text book of pediatric dentistry. Baltimore: Williams&Wilkins. 1985.
Komabayashi T, Qiang Z, Eberhart R. Current status of direct pulp-capping materials for permanent teeth. Dent Mater J. 2016;35(1):1–12.
Lee H, Shin Y, Kim S-O, et al. Comparative study of pulpal responses to pulpotomy with ProRoot MTA, RetroMTA, and TheraCal in Dogs’ teeth. J Endod. 2015;41(8):1317–24.
Liang C, SUH BI. Cytotoxicity and biocompatibility of resin-free and resin-modified direct pulp capping materials: a state-of-the-art review. Dent Mater J. 2017;36(1):1–7.
Nielsen M, Casey J, VanderWeele R, Vandewalle K. Mechanical properties of new dental pulp-capping materials. Gen Dent. 2015;64(1):44–8.
Parirokh M, Torabinejad M. Mineral trioxide aggregate: a comprehensive literature review—part I: chemical, physical, and antibacterial properties. J Endod. 2010a;36(1):16–27.
Parirokh M, Torabinejad M. Mineral trioxide aggregate: a comprehensive literature review—part III: clinical applications, drawbacks, and mechanism of action. J Endod. 2010b;36(3):400–13.
Petrolo F, Comba A, Scansetti M, et al. Effects of light-cured MTA like material on direct pulp capping. Dent Mater. 2014;30:e151.
Poggio C, Arciola CR, Beltrami R, et al. Cytocompatibility and antibacterial properties of capping materials. Sci World J 2014;2014:1–10.
Poggio C, Ceci M, Dagna A, et al. In vitro cytotoxicity evaluation of different pulp capping materials: a comparative study. Arh Hig Rada Toksikol. 2015;66(3):181–8.
Ranjkesh B, Isidor F, Dalstra M, Løvschall H. Diametral tensile strength of novel fast-setting calcium silicate cement. Dent Mater J. 2016;35(4):559–63.
Sarkar N, Caicedo R, Ritwik P, Moiseyeva R, Kawashima I. Physicochemical basis of the biologic properties of mineral trioxide aggregate. J Endod. 2005;31(2):97–100.
Sluyk S, Moon P, Hartwell G. Evaluation of setting properties and retention characteristics of mineral trioxide aggregate when used as a furcation perforation repair material. J Endod. 1998;24(11):768–71.
Tomson PL, Grover LM, Lumley PJ, et al. Dissolution of bio-active dentine matrix components by mineral trioxide aggregate. J Dent. 2007;35(8):636–42.
Tuna D, Ölmez A. Clinical long-term evaluation of MTA as a direct pulp capping material in primary teeth. Int Endod J. 2008;41(4):273–8.
Tüzüner T, Alacam A, Altunbas D, Gokdogan F, Gundogdu E. Clinical and radiographic outcomes of direct pulp capping therapy in primary molar teeth following haemostasis with various antiseptics: a randomised controlled trial. Eur J Paediatr Dent. 2012;13(4):289–92.
Acknowledgements
This study was supported by Tabriz University of Medical Sciences. Special thanks to the staff at the Department of Paediatric Dentistry, and the children and parents for their participating in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Professor Leila Erfanparast declares that she has no conflict of interest with respect to the authorship and/or publication of this paper. Dr. Parastoo Iranparvar declares that she has no conflict of interest with respect to the authorship and/or publication of this paper. Professor Ali Vafaei declares that he has no conflict of interest with respect to the authorship and/or publication of this paper.
Research involving human participants
5–7 years old children were included in our study.
Ethical approval
All procedures performed in this study which involved human participants were in accordance with the ethical standards of the “Committee for Ethics in Research on Humans” at Tabriz University of Medical Sciences (Trial no. IR.TBZMED.REC.1395.994) and with the 1964 Helsinki Declaration of Human Rights and its later amendments or comparable ethical standards.
Informed consent
Each experiment was included in the study after reading, understanding, and completing the written informed consent document by his/her parent.
Rights and permissions
About this article
Cite this article
Erfanparast, L., Iranparvar, P. & Vafaei, A. Direct pulp capping in primary molars using a resin-modified Portland cement-based material (TheraCal) compared to MTA with 12-month follow-up: a randomised clinical trial. Eur Arch Paediatr Dent 19, 197–203 (2018). https://doi.org/10.1007/s40368-018-0348-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40368-018-0348-6