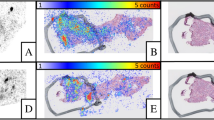
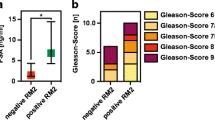
Abstract
Despite significant advances in detection and treatment, prostate cancer (PC) remains the most common malignancy and a major cause of cancer death in men worldwide. Imaging is critically important for the diagnosis, staging, and management of men with prostate cancer (PC). The conventional imaging modalities have been employed for local and metastatic staging with limited performance. Whole-body positron emission tomography (PET) using prostate membrane antigen-based tracers is the most widely used in research and clinical applications in this scenario. However, other ways to image PC are available. Here, we review the clinical literatures on the use of gastrin-releasing peptide receptors (GRPRs) as targets for imaging patients with PC.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66:7–30
Center MM, Jemal A, Lortet-Tieulent J, Ward E, Ferlay J, Brawley O, Bray F (2012) International variation in prostate cancer incidence and mortality rates. Eur Urol 61:1079–1092
Ward JF, Moul JW (2005) Rising prostate-specific antigen after primary prostate cancer therapy. Nat Clin Pract Urol 2:174–182
Van Poppel H, Vekemans K, Da Pozzo L, Bono A, Kliment J, Montironi R, Debois M, Collette L (2006) Radical prostatectomy for locally advanced prostate cancer: results of a feasibility study (EORTC 30001). Eur J Cancer 42:1062–1067
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, Mason M, Matveev V, Wiegel T, Zattoni F, Mottet N (2014) European Association of Urology EAU guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent—update 2013. Eur Urol 65:124–137
Hövels AM, Heesakkers RA, Adang EM, Jager GJ, Strum S, Hoogeveen YL, Severens JL, Barentsz JO (2008) The diagnostic accuracy of CT and MRI in the staging of pelvic lymph nodes in patients with prostate cancer: a meta-analysis. Clin Radiol 63:387–395
Evangelista L, Briganti A, Fanti S, Joniau S, Reske S, Schiavina R, Stief C, Thalmann GN, Picchio M (2016) New clinical indications for (18)F/(11)C-choline, new tracers for positron emission tomography and a promising hybrid device for prostate cancer staging: a systematic review of the literature. Eur Urol 70:161–175
Jensen RT, Battey JF, Spindel ER, Benya RV (2008) International Union of Pharmacology. LXVIII. Mammalian bombesin receptors: nomenclature, distribution, pharmacology, signaling, and functions in normal and disease states. Pharmacol Rev 60:1–42
Mansi R, Fleischmann A, Mäcke HR, Reubi JC (2013) Targeting GRPR in urological cancers—from basic research to clinical application. Nat Rev Urol 10:235–244
Sonni I, Baratto L, Iagaru A (2017) Imaging of prostate cancer using Gallium-68-labeled bombesin. PET Clin 12:159–171
Schroeder RP, van Weerden WM, Bangma C, Krenning EP, de Jong M (2009) Peptide receptor imaging of prostate cancer with radiolabelled bombesin analogues. Methods 48:200–204
Schroeder RP, de Visser M, van Weerden WM, de Ridder CM, Reneman S, Melis M, Breeman WA, Krenning EP, de Jong M (2010) Androgen-regulated gastrin-releasing peptide receptor expression in androgen-dependent human prostate tumor xenografts. Int J Cancer 126:2826–2834
Xiao C, Reitman ML (2016) Bombesin-like receptor 3: physiology of a functional orphan. Trends Endocrinol Metab 27:603–605
Ferreira CA, Fuscaldi LL, Townsend DM, Rubello D, Barros ALB (2017) Radiolabeled bombesin derivatives for preclinical oncological imaging. Biomed Pharmacother 87:58–72
Baum R, Prasad V, Mutloka N, Frischknecht Maecke H, Reubi J (2007) Molecular imaging of bombesin receptors in various tumors by Ga-68 AMBA PET/CT: first results. J Nucl Med 48:79P
Mansi R, Wang X, Forrer F, Kneifel S, Tamma ML, Waser B, Cescato R, Reubi JC, Maecke HR (2009) Evaluation of a 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid-conjugated bombesin-based radioantagonist for the labeling with single-photon emission computed tomography, positron emission tomography, and therapeutic radionuclides. Clin Cancer Res 15:5240–5249
Zhang J, Niu G, Lang L, Li F, Fan X, Yan X, Yao S, Yan W, Huo L, Chen L, Li Z, Zhu Z, Chen X (2017) Clinical translation of a dual integrin αvβ3- and gastrin-releasing peptide receptor—targeting PET radiotracer, 68Ga-BBN-RGD. J Nucl Med 58:228–234
Kähkönen E, Jambor I, Kemppainen J, Lehtiö K, Grönroos TJ, Kuisma A, Luoto P, Sipilä HJ, Tolvanen T, Alanen K, Silén J, Kallajoki M, Roivainen A, Schäfer N, Schibli R, Dragic M, Johayem A, Valencia R, Borkowski S, Minn H (2013) In vivo imaging of prostate cancer using [68Ga]-labeled bombesin analog BAY86-7548. Clin Cancer Res 19:5434–5443
Wieser G, Popp I, Christian Rischke H, Drendel V, Grosu AL, Bartholomä M, Weber WA, Mansi R, Wetterauer U, Schultze-Seemann W, Meyer PT, Jilg CA (2017) Diagnosis of recurrent prostate cancer with PET/CT imaging using the gastrin-releasing peptide receptor antagonist 68Ga-RM2: preliminary results in patients with negative or inconclusive [18F]Fluoroethylcholine-PET/CT. Eur J Nucl Med Mol Imaging 44:1463–1472
Minamimoto R, Sonni I, Hancock S, Vasanawala S, Loening A, Gambhir SS, Iagaru A (2018) Prospective evaluation of 68Ga-RM2 PET/MRI in patients with biochemical recurrence of prostate cancer and negative findings on conventional imaging. J Nucl Med 59:803–808
Baratto L, Harrison C, Davidzon G, Yohannan T, Iagaru A (2018) 68Ga-RM2 PET/MRI detection of recurrent prostate cancer in patients with negative conventional imaging. J Nucl Med 59:453
Maina T, Bergsma H, Kulkarni HR, Mueller D, Charalambidis D, Krenning EP, Nock BA, de Jong M, Baum RP (2016) Preclinical and first clinical experience with the gastrin-releasing peptide receptor-antagonist [68Ga]SB3 and PET/CT. Eur J Nucl Med Mol Imaging 43:964–973
Nock BA, Kaloudi A, Lymperis E, Giarika A, Kulkarni HR, Klette I, Singh A, Krenning EP, de Jong M, Maina T, Baum RP (2017) Theranostic perspectives in prostate cancer with the gastrin-releasing peptide receptor antagonist NeoBOMB1: preclinical and first clinical results. J Nucl Med 58:75–80
Cheng S, Lang L, Wang Z, Jacobson O, Yung B, Zhu G, Gu D, Ma Y, Zhu X, Niu G, Chen X (2018) Positron emission tomography imaging of prostate cancer with Ga-68-labeled gastrin-releasing peptide receptor agonist BBN7–14 and antagonist RM26. Bioconjug Chem 29:410–419
Zhang J, Niu G, Fan X, Lang L, Hou G, Chen L, Wu H, Zhu Z, Li F, Chen X (2018) PET using a GRPR antagonist 68Ga-RM26 in healthy volunteers and prostate cancer patients. J Nucl Med 59:922–928
Sah BR, Burger IA, Schibli R, Friebe M, Dinkelborg L, Graham K, Borkowski S, Bacher-Stier C, Valencia R, Srinivasan A, Hany TF, Mu L, Wild PJ, Schaefer NG (2015) Dosimetry and first clinical evaluation of the new 18F-radiolabeled bombesin analogue BAY 864367 in patients with prostate cancer. J Nucl Med 56:372–378
Wieser G, Mansi R, Grosu AL, Schultze-Seemann W, Dumont-Walter RA, Meyer PT, Maecke HR, Reubi JC, Weber WA (2014) Positron emission tomography (PET) imaging of prostate cancer with a gastrin releasing peptide receptor antagonist—from mice to men. Theranostics 4:412–419
Ananias HJ, Yu Z, Hoving HD, Rosati S, Dierckx RA, Wang F, Yan Y, Chen X, Pruim J, Lub-de Hooge MN, Helfrich W, Elsinga PH, de Jong IJ (2013) Application of (99m)Technetium-HYNIC(tricine/TPPTS)-Aca-Bombesin7–14 SPECT/CT in prostate cancer patients: a first-in-man study. Nucl Med Biol 40:933–938
Minamimoto R, Hancock S, Schneider B, Chin FT, Jamali M, Loening A, Vasanawala S, Gambhir SS, Iagaru A (2016) Pilot comparison of 68Ga-RM2 PET and 68Ga-PSMA-11 PET in patients with biochemically recurrent prostate cancer. J Nucl Med 57:557–562
Author information
Authors and Affiliations
Contributions
LB: literature search, literature review, content planning, and manuscript writing and editing. RL: literature search, content planning, and manuscript writing and editing. MP, SB, and AI: content planning and editing.
Corresponding author
Ethics declarations
Conflict of interest
L. Baratto, R. Laudicella, M. Picchio, S. Baldari, and A. Iagaru declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Baratto, L., Laudicella, R., Picchio, M. et al. Imaging gastrin-releasing peptide receptors (GRPRs) in prostate cancer. Clin Transl Imaging 7, 39–44 (2019). https://doi.org/10.1007/s40336-018-00308-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40336-018-00308-x