Abstract
Background
Increasing evidence supports the notion that human papillomavirus (HPV) DNA integration onto the human genome can influence and alter the molecular cargo in the exosomes derived from head and neck cancer cells. However, the molecular cargo of salivary exosomes derived from HPV-driven oropharyngeal cancer (HPV-driven OPC) remains unelucidated.
Methods and materials
Salivary exosomes morphology and molecular characterizations were examined using the nanoparticle tracking (NTA), western blot analysis, transmission electron microscopy (TEM) and mass spectrometry analysis.
Results
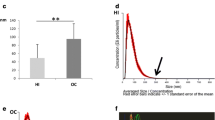
We report that HPV16 DNA was detected (80%) in isolated salivary exosomes of HPV-driven OPC patients. Importantly, we demonstrate elevated protein levels of six main glycolytic enzymes [i.e., aldolase (ALDOA), glyceraldehye-3-phosphate dehydrogenase (GAPDH), lactate dehydrogenase A/B (LDHA and LDHB), phosphoglycerate kinase 1 (PGK1) and pyruvate kinase M1/2 (PKM)] in isolated salivary exosomes of HPV-driven OPC patients, suggesting a novel mechanism underlying the potential role of salivary exosomes in mediating the reciprocal interplay between glucose metabolism and HPV-driven OPC.
Conclusion
Our data demonstrate the potential diagnostic value of HPV16 DNA and glycolytic enzymes in salivary exosomes in discriminating healthy controls from HPV-driven OPC patients, thereby opening new avenues in the future for clinical translation studies.





Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359–86.
Chi AC, Day TA, Neville BW. Oral cavity and oropharyngeal squamous cell carcinoma—an update. CA Cancer J Clin. 2015;65(5):401–21.
Dahlstrom KR, Li G, Tortolero-Luna G, Wei Q, Sturgis EM. Differences in history of sexual behavior between patients with oropharyngeal squamous cell carcinoma and patients with squamous cell carcinoma at other head and neck sites. Head Neck. 2011;33(6):847–55.
D’Souza G, Kreimer AR, Viscidi R, Pawlita M, Fakhry C, Koch WM, et al. Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med. 2007;356(19):1944–56.
Ellington TD, Henley SJ, Senkomago V, O’Neil ME, Wilson RJ, Singh S, et al. Trends in incidence of cancers of the oral cavity and pharynx—United States 2007–2016. MMWR Morb Mortal Wkly Rep. 2020;69(15):433–8.
Lechner M, Breeze CE, O’Mahony JF, Masterson L. Early detection of HPV-associated oropharyngeal cancer. Lancet. 2019;393(10186):2123.
Hong A, Lee CS, Jones D, Veillard AS, Zhang M, Zhang X, et al. Rising prevalence of human papillomavirus-related oropharyngeal cancer in Australia over the last 2 decades. Head Neck. 2016;38(5):743–50.
Sun CX, Bennett N, Tran P, Tang KD, Lim Y, Frazer I, et al. A Pilot Study into the Association between Oral Health Status and Human Papillomavirus-16 Infection. Diagnostics (Basel). 2017;7(1):11.
D’Souza G, McNeel TS, Fakhry C. Understanding personal risk of oropharyngeal cancer: risk-groups for oncogenic oral HPV infection and oropharyngeal cancer. Ann Oncol. 2017;28(12):3065–9.
Gillison ML, Broutian T, Pickard RK, Tong ZY, Xiao W, Kahle L, et al. Prevalence of oral HPV infection in the United States, 2009–2010. JAMA. 2012;307(7):693–703.
Crosbie EJ, Einstein MH, Franceschi S, Kitchener HC. Human papillomavirus and cervical cancer. Lancet. 2013;382(9895):889–99.
Schiffman M, Castle PE, Jeronimo J, Rodriguez AC, Wacholder S. Human papillomavirus and cervical cancer. Lancet. 2007;370(9590):890–907.
Pan C, Issaeva N, Yarbrough WG. HPV-driven oropharyngeal cancer: current knowledge of molecular biology and mechanisms of carcinogenesis. Cancers Head Neck. 2018;3:12.
Kreimer AR, Clifford GM, Boyle P, Franceschi S. Human papillomavirus types in head and neck squamous cell carcinomas worldwide: a systematic review. Cancer Epidemiol Biomark Prev. 2005;14(2):467–75.
Tang KD, Vasani S, Taheri T, Walsh LJ, Hughes BGM, Kenny L, et al. An occult HPV-driven oropharyngeal squamous cell carcinoma discovered through a saliva test. Front Oncol. 2020;10:408. https://doi.org/10.3389/fonc.2020.00408
Koshiol J, Lindsay L, Pimenta JM, Poole C, Jenkins D, Smith JS. Persistent human papillomavirus infection and cervical neoplasia: a systematic review and meta-analysis. Am J Epidemiol. 2008;168(2):123–37.
Munger K, Baldwin A, Edwards KM, Hayakawa H, Nguyen CL, Owens M, et al. Mechanisms of human papillomavirus-induced oncogenesis. J Virol. 2004;78(21):11451–60.
Rampias T, Sasaki C, Weinberger P, Psyrri A. E6 and e7 gene silencing and transformed phenotype of human papillomavirus 16-positive oropharyngeal cancer cells. J Natl Cancer Inst. 2009;101(6):412–23.
Kim SH, Koo BS, Kang S, Park K, Kim H, Lee KR, et al. HPV integration begins in the tonsillar crypt and leads to the alteration of p16, EGFR and c-myc during tumor formation. Int J Cancer. 2007;120(7):1418–25.
Tang KD, Baeten K, Kenny L, Frazer IH, Scheper G, Punyadeera C. Unlocking the potential of saliva-based test to detect HPV-16-driven oropharyngeal cancer. Cancers. 2019;11(4):473.
Parfenov M, Pedamallu CS, Gehlenborg N, Freeman SS, Danilova L, Bristow CA, et al. Characterization of HPV and host genome interactions in primary head and neck cancers. Proc Natl Acad Sci USA. 2014;111(43):15544–9.
Harden ME, Munger K. Human papillomavirus 16 E6 and E7 oncoprotein expression alters microRNA expression in extracellular vesicles. Virology. 2017;508:63–9.
Kannan A, Hertweck KL, Philley JV, Wells RB, Dasgupta S. Genetic mutation and exosome signature of human papilloma virus associated oropharyngeal cancer. Sci Rep. 2017;7(1):46102.
Ludwig S, Sharma P, Theodoraki MN, Pietrowska M, Yerneni SS, Lang S, et al. Molecular and functional profiles of exosomes from HPV(+) and HPV(-) head and neck cancer cell lines. Front Oncol. 2018;8:445.
Ludwig S, Marczak L, Sharma P, Abramowicz A, Gawin M, Widlak P, et al. Proteomes of exosomes from HPV(+) or HPV(-) head and neck cancer cells: differential enrichment in immunoregulatory proteins. Oncoimmunology. 2019;8(7):1593808.
Tang KD, Menezes L, Baeten K, Walsh LJ, Whitfield BCS, Batstone MD, et al. Oral HPV16 prevalence in oral potentially malignant disorders and oral cavity cancers. Biomolecules. 2020;10(2):223.
Tang KD, Vasani S, Menezes L, Taheri T, Walsh LJ, Hughes BGM, et al. Oral HPV16 DNA as a screening tool to detect early oropharyngeal squamous cell carcinoma. Cancer Sci. 2020;111(10):3854–61.
Zlotogorski-Hurvitz A, Dayan D, Chaushu G, Korvala J, Salo T, Sormunen R, et al. Human saliva-derived exosomes: comparing methods of isolation. J Histochem Cytochem. 2015;63(3):181–9.
Tang KD, Kenny L, Frazer IH, Punyadeera C. High-risk human papillomavirus detection in oropharyngeal cancers: comparison of saliva sampling methods. Head Neck. 2019;41(5):1484–9.
Tang KD, Liu J, Russell PJ, Clements JA, Ling MT. Gamma-tocotrienol induces apoptosis in prostate cancer cells by targeting the Ang-1/Tie-2 signalling pathway. Int J Mol Sci. 2019;20(5):1164.
Zhang X, Sadowski P, Punyadeera C. Evaluation of sample preparation methods for label-free quantitative profiling of salivary proteome. J Proteom. 2020;210:103532.
Rappsilber J, Mann M, Ishihama Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat Protoc. 2007;2(8):1896–906.
Zhang X, Walsh T, Atherton JJ, Kostner K, Schulz B, Punyadeera C. Identification and Validation of a Salivary Protein Panel to Detect Heart Failure Early. Theranostics. 2017;7(18):4350–8.
Clough T, Thaminy S, Ragg S, Aebersold R, Vitek O. Statistical protein quantification and significance analysis in label-free LC-MS experiments with complex designs. BMC Bioinformatics. 2012;13(Suppl 16):S6.
Wang Z, Li F, Rufo J, Chen C, Yang S, Li L, et al. Acoustofluidic salivary exosome isolation: a liquid biopsy compatible approach for human papillomavirus-associated oropharyngeal cancer detection. J Mol Diagn. 2020;22(1):50–9.
Nair S, Tang KD, Kenny L, Punyadeera C. Salivary exosomes as potential biomarkers in cancer. Oral Oncol. 2018;84:31–40.
Lin Y, Dong H, Deng W, Lin W, Li K, Xiong X, et al. Evaluation of salivary exosomal chimeric GOLM1-NAA35 RNA as a potential biomarker in esophageal carcinoma. Clin Cancer Res. 2019;25(10):3035–45.
Muller Bark J, Kulasinghe A, Amenabar JM, Punyadeera C. Exosomes in cancer. Adv Clin Chem. 2021;101:1–40.
Pfaffe T, Cooper-White J, Beyerlein P, Kostner K, Punyadeera C. Diagnostic potential of saliva: current state and future applications. Clin Chem. 2011;57(5):675–87.
Tang KD, Kenny L, Perry C, Frazer I, Punyadeera C. The overexpression of salivary cytokeratins as potential diagnostic biomarkers in head and neck squamous cell carcinomas. Oncotarget. 2017;8(42):72272–80.
Guenat D, Hermetet F, Pretet JL, Mougin C. Exosomes and other extracellular vesicles in HPV transmission and carcinogenesis. Viruses. 2017;9(8):211.
Honegger A, Schilling D, Bastian S, Sponagel J, Kuryshev V, Sultmann H, et al. Dependence of intracellular and exosomal microRNAs on viral E6/E7 oncogene expression in HPV-positive tumor cells. PLoS Pathog. 2015;11(3):e1004712.
Wang HF, Wang SS, Tang YJ, Chen Y, Zheng M, Tang YL, et al. The double-edged sword-how human papillomaviruses interact with immunity in head and neck cancer. Front Immunol. 2019;10:653.
Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tan PF, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010;363(1):24–35.
Schmidt H, Kulasinghe A, Perry C, Nelson C, Punyadeera C. A liquid biopsy for head and neck cancers. Expert Rev Mol Diagn. 2016;16(2):165–72.
Hanna GJ, Lau CJ, Mahmood U, Supplee JG, Mogili AR, Haddad RI, et al. Salivary HPV DNA informs locoregional disease status in advanced HPV-associated oropharyngeal cancer. Oral Oncol. 2019;95:120–6.
Toussaint-Smith E, Donner DB, Roman A. Expression of human papillomavirus type 16 E6 and E7 oncoproteins in primary foreskin keratinocytes is sufficient to alter the expression of angiogenic factors. Oncogene. 2004;23(17):2988–95.
Bardos JI, Ashcroft M. Negative and positive regulation of HIF-1: a complex network. Biochim Biophys Acta. 2005;1755(2):107–20.
Rodolico V, Arancio W, Amato MC, Aragona F, Cappello F, Di Fede O, et al. Hypoxia inducible factor-1 alpha expression is increased in infected positive HPV16 DNA oral squamous cell carcinoma and positively associated with HPV16 E7 oncoprotein. Infect Agent Cancer. 2011;6(1):18.
Guo Y, Meng X, Ma J, Zheng Y, Wang Q, Wang Y, et al. Human papillomavirus 16 E6 contributes HIF-1alpha induced Warburg effect by attenuating the VHL-HIF-1alpha interaction. Int J Mol Sci. 2014;15(5):7974–86.
Fan R, Hou WJ, Zhao YJ, Liu SL, Qiu XS, Wang EH, et al. Overexpression of HPV16 E6/E7 mediated HIF-1alpha upregulation of GLUT1 expression in lung cancer cells. Tumour Biol. 2016;37(4):4655–63.
Crusius K, Auvinen E, Alonso A. Enhancement of EGF- and PMA-mediated MAP kinase activation in cells expressing the human papillomavirus type 16 E5 protein. Oncogene. 1997;15(12):1437–44.
Martinez-Ramirez I, Carrillo-Garcia A, Contreras-Paredes A, Ortiz-Sanchez E, Cruz-Gregorio A, Lizano M. Regulation of cellular metabolism by high-risk human papillomaviruses. Int J Mol Sci. 2018;19(7):1839.
Acknowledgements
We would like to thank the members of Saliva and Liquid Biopsy Translational Laboratory for their assistance in sample collection and processing. We also thank Dana Middleton and the staff at the Princess Alexandra Hospital for their assistance in the recruitment of study patients and collection of clinical samples. In addition, we thank the volunteers who took part in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
CP is currently receiving funding from Cancer Australia (APP1145657), NHMRC Ideas Grant (APP 2002576), National Institutes of Health (NIH) (1R21EB030349-01), Garnett Passes and Rodney Williams Foundation and Royal Brisbane Women’s Hospital Foundation.
Conflict of interest
The authors declare that there is no conflict of interest
Ethics approval
This study was approved by the Medical Ethical Institutional Board of University of Queensland (HREC No: 2014000862); Queensland University of Technology (HREC No: 1400000617 and 1400000641) and by the Princess Alexandra Hospital (PAH) Ethics Review Board (HREC Number: HREC/12/QPAH/381), following the Declaration of Helsinki 1964 and its later amendments or comparable ethical standards.
Consent to participate
All participants provided written informed consent, prior to saliva sample collection.
Consent for publication
Not applicable.
Availability of data and material
All datasets generated for this study are included in the article.
Code availability
Not applicable.
Author contributions
All authors have read and agree to the published version of the manuscript. KDT, YX and CP: conceptualization. All authors: methodology, validation, formal analysis, data curation, investigation, and writing—review and editing. KDT: writing—original draft preparation. CP: funding acquisition.
Rights and permissions
About this article
Cite this article
Tang, K.D., Wan, Y., Zhang, X. et al. Proteomic Alterations in Salivary Exosomes Derived from Human Papillomavirus-Driven Oropharyngeal Cancer. Mol Diagn Ther 25, 505–515 (2021). https://doi.org/10.1007/s40291-021-00538-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40291-021-00538-2