Abstract
Background
The aim of this study was to investigate the role of matrix metalloproteinase-9 (MMP-9) C-1562T and 279R/Q (836G>A) polymorphisms in the development of chronic obstructive pulmonary disease (COPD) in Tunisians and to determine their impact on disease progression and airflow obstruction.
Methods
Pulmonary functional tests were evaluated by body plethysmography. MMP-9 genotypes were determined in patients with COPD (n = 138) and healthy controls (n = 216) by polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP). Serum MMP-9 and interleukin-6 (IL-6) levels were determined by enzyme-linked immunosorbent assays (ELISA) and activity of MMP-9 was evaluated by gelatin zymography.
Results
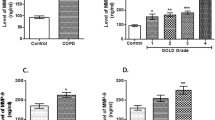
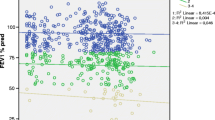
No significant association was found between genetic variations in MMP-9 C-1562T and 279R/Q polymorphisms and the risk of development of COPD. However, a significant correlation was retrieved between the 279 R/Q polymorphism and disease severity (P = 0.02). In addition, homozygous Q (A) genotype was associated with a poorer lung function with a fall in forced expiratory volume in 1 s (FEV1) (%) and forced vital capacity (FVC%) among COPD patients compared with both AG and GG individuals (52.06 ± 19.6 vs. 59.08 ± 17.19, P = 0.03 and 72.41 ± 21.42 vs. 82.98 ± 16.48, P = 0.002, respectively). Using ELISA, a higher level of MMP-9 was found in patients with the CT genotype (P = 0.03), while no significant impact of the 279R/Q polymorphism was observed (P = 0.48). In contrast, by using zymography gel analysis, MMP-9 activity was enhanced in individuals carrying the R(G) allele in comparison with those homozygous for the Q(A) variant (P = 0.02).
Conclusion
Our results support a role for the 279R/Q polymorphism in physiological alterations that may affect progression and severity of COPD. These findings could be related to the decreased activity of MMP-9 among COPD patients carrying the 279Q variant.




Similar content being viewed by others
References
Baraldo S, Turato G, Saetta M. Pathophysiology of the small airways in chronic obstructive pulmonary disease. Respiration. 2012;84:89–97.
Aoshiba K, Nagai A. Differences in airway remodeling between asthma and chronic obstructive pulmonary disease. Clin Rev Allergy Immunol. 2004;27:35–43.
Vandenbroucke RE, Dejonckheere E, Libert C. A therapeutic role for matrix metalloproteinase inhibitors in lung diseases? Eur Respir J. 2011;38:1200–14.
Parks WC, Wilson CL, Lopez-Boado YS. Matrix metalloproteinases as modulators of inflammation and innate immunity. Nat Rev Immunol. 2004;4:617–29.
Nyberg P, Xie L, Kalluri R. Endogenous inhibitors of angiogenesis. Cancer Res. 2005;65:3967–79.
Newby AC. Dual role of matrix metalloproteinases (matrixins) in intimal thickening and atherosclerotic plaque rupture. Physiol Rev. 2005;85:1–31.
Kumar M, Phougat N, Ruhil S, Dhankhar S, Balhara M, Chhillar AK. Genomics of chronic obstructive pulmonary disease (COPD); exploring the SNPs of protease-antiprotease pathway. Curr Genomics. 2013;14:204–13.
Renckens R, Roelofs JJ, Florquin S, de Vos AF, Lijnen HR, van’t Veer C, et al. Matrix metalloproteinase-9 deficiency impairs host defense against abdominal sepsis. J Immunol. 2006;176:3735–41.
Albaiceta GM, Gutiérrez-Fernández A, Parra D, Astudillo A, García-Prieto E, Taboada F, et al. Lack of matrix metalloproteinase-9 worsens ventilator-induced lung injury. Am J Physiol Lung Cell Mol Physiol. 2008;294:535–43.
Zhang B, Ye S, Herrmann SM, Eriksson P, de Maat M, Evans A, et al. Functional polymorphism in the regulatory region of gelatinase B gene in relation to severity of coronary atherosclerosis. Circulation. 1999;99:1788–94.
Zhang B, Henney A, Eriksson P, Hamsten A, Watkins H, Ye S. Genetic variation at the matrix metalloproteinase-9 locus on chromosome 20q12.2-13.1. Hum Genet. 1999;105:418–23.
Schirmer H, Basso da Silva L, Teixeira PJ, Moreira JS, Moreira AL, Simon D. Matrix metalloproteinase gene polymorphisms: lack of association with chronic obstructive pulmonary disease in a Brazilian population. Genet Mol Res. 2009;8:1028–34.
Lee SY, Kim MJ, Kang HG, Yoo SS, Choi YY, Lee WK, et al. Polymorphisms in matrix metalloproteinase-1, -9 and -12 genes and the risk of chronic obstructive pulmonary disease in a Korean population. Respiration. 2010;80:133–8.
American Thoracic Society. Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 1995;152:S77–S121.
Pires DE, Ascher DB, Blundell TL. DUET: a server for predicting effects of mutations on protein stability using an integrated computational approach. Nucleic Acids Res. 2014;42:314–9.
Cheng J, Randall A, Baldi P. Prediction of protein stability changes for single-site mutations using support vector machines. Proteins. 2006;62:1125–32.
Pires DE, Ascher DB, Blundell TL. mCSM: predicting the effects of mutations in proteins using graph-based signatures. Bioinformatics. 2014;30:335–42.
Opdenakker G, Van den Steen PE, Dubois B, Nelissen I, Van Coillie E, Masure S. Gelatinase B functions as regulator and effector in leukocyte biology. J Leukoc Biol. 2001;59:851–9.
Brajer B, Batura-Gabryel H, Nowicka A, Kuznar-Kaminska B, Szczepanik A. Concentration of matrix metalloproteinase-9 in serum of patients with chronic obstructive pulmonary disease and a degree of airway obstruction and disease progression. J Physiol Pharmacol. 2008;59(Suppl 6):145–52.
Skarmoutsou E, D’Amico F, Marchini M, Stivala F, Malaponte G, Scorza R, et al. Analysis of matrix metalloproteinase-9 gene polymorphism 1562 C/T in patients suffering from systemic sclerosis with and without ulcers. Int J Mol Med. 2011;27:873–7.
Montaner J, Fernández-Cadenas I, Molina CA, Monasterio J, Arenillas JF, Ribó M, et al. Safety profile of tissue plasminogen activator treatment among stroke patients carrying a common polymorphism (C-1562T) in the promoter region of the matrix metalloproteinase-9 gene. Stroke. 2003;34:2851–5.
Shan K, Lian-Fu Z, Hui D, Wei G, Na W, Xia J, et al. Polymorphisms in the promoter regions of the matrix metalloproteinases-7, -9 and the risk of endometriosis and adenomyosis in China. Mol Hum Reprod. 2006;12:35–9.
Peres RC, Line SR. Analysis of MMP-9 and TIMP-2 gene promoter polymorphisms in individuals with hypodontia. Braz Dent J. 2005;16:231–6.
Korytina GF, Akhmadishina LZ, Ianbaeva DG, Viktorova TV. Polymorphism in promoter regions of matrix metalloproteinases (MMP1, MMP9, and MMP12) in chronic obstructive pulmonary disease patients [in Russian]. Genetika. 2008;44:242–9.
Ito I, Nagai S, Handa T, Muro S, Hirai T, Tsukino M, et al. Matrix metalloproteinase-9 promoter polymorphism associated with upper lung dominant emphysema. Am J Respir Crit Care Med. 2005;172:1378–82.
Haq I, Chappell S, Johnson SR, Lotya J, Daly L, Morgan K, et al. Association of MMP-2 polymorphisms with severe and very severe COPD: a case control study of MMPs-1, 9 and12 in a European population. BMC Med Genet. 2010;11:7. doi:10.1186/1471-2350-11-7.
Chen L, Wang T, Liu L, Shen Y, Wan C, Wen F. Matrix metalloproteinase-9-1562C/T promoter polymorphism confers risk for COPD: a meta-analysis. PLoS One. 2013;8:e60523.
Tesfaigzi Y, Myers OB, Stidley CA, Schwalm K, Picchi M, Crowell RE, et al. Genotypes in matrix metalloproteinase 9 are a risk factor for COPD. Int J Chron Obstruct Pulmon Dis. 2006;1:267–78.
Minematsu N, Nakamura H, Tateno H, Nakajima T, Yamaguchi K. Genetic polymorphism in matrix metalloproteinase-9 and pulmonary emphysema. Biochem Biophys Res Commun. 2001;289:116–9.
Kukkonen MK, Tiili E, Vehmas T, Oksa P, Piirilä P, Hirvonen A. Association of genes of protease-antiprotease balance pathway to lung function and emphysema subtypes BMC Pulm Med. 2013;13:36.
Joos L, He JQ, Shepherdson MB, Connett JE, Anthonisen NR, Paré PD, et al. The role of matrix metalloproteinase polymorphisms in the rate of decline in lung function. Hum Mol Genet. 2002;11:569–76.
Medley TL, Cole TJ, Dart AM, Gatzka CD, Kingwell BA. Matrix metalloproteinase-9 genotype influences large artery stiffness through effects on aortic gene and protein expression. Arterioscler Thromb Vasc Biol. 2004;24:1479–84.
Awadalla MS, Burdon KP, Kuot A, Hewitt AW, Craig JE. Matrix metalloproteinase-9 genetic variation and primary angle closure glaucoma in a Caucasian population. Mol Vis. 2011;17:1420–4.
Lose F, Thompson PJ, Duffy D, Stewart GA, Kedda MA. A novel tissue inhibitor of metalloproteinase-1 (TIMP-1) polymorphism associated with asthma in Australian women. Thorax. 2005;60:623–8.
Singh R, Srivastava P, Srivastava A, Mittal RD. Matrix metalloproteinase (MMP-9 and MMP-2) gene polymorphisms influence allograft survival in renal transplant recipients. Nephrol Dial Transplant. 2010;25:3393–401.
Natividad A, Cooke G, Holland MJ, Burton MJ, Joof HM, Rockett K, et al. A coding polymorphism in matrix metalloproteinase 9 reduces risk of scarring sequelae of ocular Chlamydia trachomatis infection. BMC Med Genet. 2006;7:40.
Goldberg GI, Strongin A, Collier IE, Genrich LT, Marmer BL. Interaction of 92-kDa type IV collagenase with the tissue inhibitor of metalloproteinases prevents dimerization, complex formation with interstitial collagenase, and activation of the proenzyme with stromelysin. J Biol Chem. 1992;267:4583–91.
Inoue H, Mashimo Y, Funamizu M, Yonekura S, Horiguchi S, Shimojo N, et al. Association of the MMP9 gene with childhood cedar pollen sensitization and pollinosis. J Hum Genet. 2012;57:176–83.
Perotin JM, Adam D, Vella-Boucaud J, Delepine G, Sandu S, Jonvel AC, et al. Delay of airway epithelial wound repair in COPD is associated with airflow obstruction severity. Respir Res. 2014;15:151.
Matsumoto H, Niimi A, Takemura M, Ueda T, Minakuchi M, Tabuena R, et al. Relationship of airway wall thickening to an imbalance between matrix metalloproteinase-9 and its inhibitor in asthma. Thorax. 2005;60:277–81.
Ahluwalia TS, Khullar M, Ahuja M, Kohli HS, Bhansali A, Mohan V, et al. Common variants of inflammatory cytokine genes are associated with risk of nephropathy in type 2 diabetes among Asian Indians. PLoS One. 2009;4:e5168.
Acknowledgments
This work was supported by the Ministry of Higher Education and Scientific Research and by the Ministry of Public Health of the Tunisian Government. The authors wish to thank all of the patients and controls who participated in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors SB, HbN, IRH, AbA, SY, AG, MB, BB, ZT, and KC have no conflicts of interest that are directly relevant to the content of this article.
Funding
The authors SB, HbN, IRH, AbA, SY, AG, MB, BB, ZT, and KC declare that this study was funded by the Tunisian Ministry of Higher Education and Research (UR12ES06).
Ethical approval and informed consent
The study was approved by the local ethical committee of Farhat Hachad Hospital (Sousse, Tunisia; approval number: 08-2011).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bchir, S., Nasr, H.b., Hakim, I.R. et al. Matrix Metalloproteinase-9 (279R/Q) Polymorphism is Associated with Clinical Severity and Airflow Limitation in Tunisian Patients with Chronic Obstructive Pulmonary Disease. Mol Diagn Ther 19, 375–387 (2015). https://doi.org/10.1007/s40291-015-0163-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40291-015-0163-2