Abstract
Background
Second-generation antipsychotics (SGAs) are prescribed for a variety of indications and are strongly associated with adverse metabolic effects. Studies of pediatric outpatients have revealed several deficiencies in monitoring practices for adverse effects associated with SGAs.
Objective
Our objective was to characterize SGA prescribing and metabolic parameter monitoring (MPM) in an inpatient pediatric population.
Methods
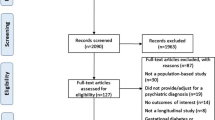
Patients aged <18 years and discharged on SGA treatment between 1 November 2013 and 30 April 2014 from an inpatient psychiatric institution in Pittsburgh, PA, USA were included. Electronic medical records (EMRs) were reviewed for patient age and weight and for parameters used by the International Diabetes Federation (IDF) to define metabolic syndrome: waist circumference, fasting blood glucose, triglycerides, high-density lipoprotein, and blood pressure. The primary outcome was the percent of patients with completed MPM, defined as all parameters being available within the patient’s EMR in any form, except estimates. Secondary outcomes included percent of patients with existing metabolic syndrome or obesity according to IDF criteria, average total daily dose of individual SGAs, and frequency of individual SGA utilization. Data were analyzed utilizing univariate descriptive statistics.
Results
A total of 243 patients met inclusion criteria and were included in the analysis. For the primary outcome, 13.2% (n = 32) of patients had completed MPM for all parameters. Blood pressure was the most frequently documented parameter (n = 241; 99.2%), whereas waist circumference was the least (n = 67; 28%). Risperidone was the most commonly prescribed SGA (n = 99; 41%; average daily dose 1.92 mg).
Conclusions
Compared with outpatient studies, rates of documented MPM for certain parameters (i.e., fasting blood glucose, lipids) is higher for pediatric inpatients treated with SGAs. However, several monitoring deficiencies are still noted.




Similar content being viewed by others
References
Cascade E, Kalali A, Findling R. Use of antipsychotics in children. Psychiatry (Edgemont). 2009;6:21–3.
Rodday AM, Parsons SK, Correll CU, Robb AS, Zima BT, Saunders TS, Leslie LK. Child and adolescent psychiatrists’ attitudes and practices prescribing second generation antipsychotics. J Child Adolesc Psychopharmacol. 2014;24(2):90–3.
Olfson M, Blanco C, Liu L, Moreno C, Laje G. National trends in the office-based treatment of children, adolescents, and adults with antipsychotics. Arch Gen Psychiatry. 2012;69(12):1247–56.
Olfson M, King M, Schoenbaum M. Treatment of young people with antipsychotic medications in the United States. JAMA Psychiatry. 2015;72(9):867–74.
Correll CU, Manu P, Olshanskiy V, Napolitano B, Kane JM, Malhotra AK. Cardiometabolic risk of second-generation antipsychotic medications during first-time use in children and adolescents. JAMA. 2009;302(16):1765–73.
Zimmet P, Alberti KG, Kaufman F, Tajima N, Silink M, Arslanian S, Wong G, Bennett P, Shaw J, Caprio S. IDF Consensus Group. The metabolic syndrome in children and adolescents—an IDF consensus report. Pediatr Diabetes. 2007;8:299–306.
Panagiotopoulos C, Ronsley R, Kuzeljevic B, Davidson J. Waist circumference is a sensitive screening tool for assessment of metabolic syndrome risk in children treated with second-generation antipsychotics. Can J Psychiatry. 2012;57(1):34–44.
Lohr WD, Honaker J. Atypical antipsychotics for the treatment of disruptive behavior. Pediatr Ann. 2013;42:2.
Honey B, Ramos L, Brahm N. Evaluation of monitoring for metabolic effects in children treated with second generation antipsychotics in a pediatric clinic. J Pediatr Pharmacol Ther. 2013;18(4):292–7.
Center for Disease Control and Prevention. BMI percentile calculator for child and teen English Version. Body mass index BMI (BMI) percentile calculator for child and teen. https://nccd.cdc.gov/dnpabmi/calculator.aspx. Accessed 27 Apr 2015.
Fernandez JR, Redden DT, Pietrobelli A, Allison DB. Waist circumference percentiles in nationally representative samples of African-American, European-American, and Mexican-American children and adolescents. J Pediatr. 2004;145(4):439–44.
Morrato EH, Nicol GE, Maahs D, Druss BG, Hartung DM, Valuck RJ, Campagna E, Newcomer JW. Metabolic screening in children receiving antipsychotic drug treatment. Arch Pediatr Adolesc Med. 2010;164(4):344–51.
The American Diabetes Association, American Psychiatric Association, American Association of Clinical Endocrinologists, and North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;7(2):596–601.
Dong B, Wang Z, Yang Y, Wang HJ, Ma J. Intensified association between waist circumference and hypertension in abdominally overweight children. Obes Res Clin Pract. 2016;10(1):24–32. doi:10.1016/j.orcp.2015.04.002.
Matone M, Localio R, Huang YS, Reis R, Feudtner C, Rubin D. The relationship between mental health diagnosis and treatment with second-generation antipsychotics over time: a national study of U.S. Medicaid-enrolled children. Health Serv Res. 2012;47(5):1836–60.
The Centers for Medicare and Medicaid Services. Atypical antipsychotic medications: use in pediatric patients. 2015. https://www.cms.gov/Medicare-Medicaid-Coordination/Fraud-Prevention/Medicaid-Integrity-Education/Pharmacy-Education-Materials/Downloads/atyp-antipsych-pediatric-factsheet11-14.pdf. Accessed 12 Dec 2016.
Acknowledgements and disclosures
Preliminary results of this work were presented at the 24th Annual Pediatric Pharmacy Advocacy Group Meeting (April 2015) and American Society of Health-System Pharmacists Midyear Meeting (December 2014.) The corresponding author was completing a PGY-2 Psychiatric Pharmacy residency at the time this project was conducted. Special thanks to Mr. Richard Crabtree for his assistance with the EMR.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
There are no grants, funding sources, or commercial interests to report in relation to this publication.
Conflict of interest
Valerie D. Nolt, Alexandra Victoria Kibler, G. Lucy Wilkening, and Tanya J. Fabian have no conflicts of interest to disclose.
Rights and permissions
About this article
Cite this article
Nolt, V.D., Kibler, A.V., Wilkening, G.L. et al. Second-Generation Antipsychotic Utilization and Metabolic Parameter Monitoring in an Inpatient Pediatric Population: A Retrospective Analysis. Pediatr Drugs 19, 139–146 (2017). https://doi.org/10.1007/s40272-016-0209-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40272-016-0209-x