Abstract
Background
Nephrotoxic medications are a common cause of acute kidney injury (AKI). Critically ill children receive more medication than other inpatients; however, the risk of nephrotoxic medication-induced AKI in these children is not well understood.
Objective
The aim of this study was to determine the association between exposure to nephrotoxic medications in the intensive care unit (ICU) and the development of AKI amongst critically ill children, adjusting for differences in underlying risk.
Methods
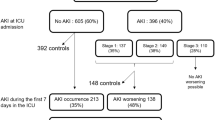
We conducted a nested case–control study among a cohort of patients admitted to a paediatric intensive care unit between January 2006 and June 2009. Cases were identified according to the RIFLE criteria. Using incidence density sampling, controls were matched 1:1 according to pre-ICU nephrotoxic drug exposure. Administration of nephrotoxic medications and other known risk factors of AKI were evaluated during the ICU stay prior to the diagnosis of AKI.
Results
A total of 914 patients in the cohort developed AKI and had an identifiable matched control. Eighty-seven percent of cases and 74% of controls were exposed to one or more nephrotoxic medications in the ICU during the study period. Furosemide (administered to 67.8% of patients), vancomycin (28.7%), and gentamicin (21.4%) were the most frequently administered nephrotoxic drugs. Patients who developed AKI were more likely to be exposed to at least one nephrotoxic medication and risk increased with increasing number of nephrotoxic medications. Ganciclovir (adjusted odds ratio [AOR] 4.7; 95% CI 1.7–13.0), furosemide (AOR 1.9; 95% CI 1.4–2.4), and gentamicin (AOR 1.8; 95% CI 1.4–2.4) significantly increased the odds of developing AKI after adjusting for underlying differences in risk factors of AKI.
Conclusion
This is the first study to assess the association between risk-adjusted nephrotoxic medication exposure and the development of AKI in critically ill children. Nephrotoxic medication exposure was common amongst children in the ICU and we found AKI was associated with the administration of specific drugs after adjustment for important risk factors.

Similar content being viewed by others
References
Akcan-Arikan A, Zappitelli M, Loftis LL, Washburn KK, Jefferson LS, Goldstein SL. Modified RIFLE criteria in critically ill children with acute kidney injury. Kidney Int. 2007;71(10):1028–35.
Plotz FB, Bouma AB, van Wijk JA, Kneyber MC, Bokenkamp A. Pediatric acute kidney injury in the ICU: an independent evaluation of pRIFLE criteria. Intensive Care Med. 2008;34(9):1713–7.
Schneider J, Khemani R, Grushkin C, Bart R. Serum creatinine as stratified in the RIFLE score for acute kidney injury is associated with mortality and length of stay for children in the pediatric intensive care unit. Crit Care Med. 2010;38(3):933–9.
Washburn KK, Zappitelli M, Arikan AA, Loftis L, Yalavarthy R, Parikh CR, et al. Urinary interleukin-18 is an acute kidney injury biomarker in critically ill children. Nephrol Dial Transpl. 2008;23(2):566–72.
Zappitelli M, Washburn KK, Arikan AA, Loftis L, Ma Q, Devarajan P, et al. Urine neutrophil gelatinase-associated lipocalin is an early marker of acute kidney injury in critically ill children: a prospective cohort study. Crit Care. 2007;11(4):R84.
Bresolin N, Bianchini AP, Haas CA. Pediatric acute kidney injury assessed by pRIFLE as a prognostic factor in the intensive care unit. Pediatr Nephrol. 2013;28(3):485–92.
Alkandari O, Eddington KA, Hyder A, Gauvin F, Ducruet T, Gottesman R, et al. Acute kidney injury is an independent risk factor for pediatric intensive care unit mortality, longer length of stay and prolonged mechanical ventilation in critically ill children: a two-center retrospective cohort study. Crit Care. 2011;15(3):R146.
Zwiers AJ, de Wildt SN, Hop WC, Dorresteijn EM, Gischler SJ, Tibboel D, et al. Acute kidney injury is a frequent complication in critically ill neonates receiving extracorporeal membrane oxygenation: a 14-year cohort study. Crit Care. 2013;17(4):R151.
Hui-Stickle S, Brewer ED, Goldstein SL. Pediatric ARF epidemiology at a tertiary care center from 1999 to 2001. Am J Kidney Dis. 2005;45(1):96–101.
Askenazi DJ, Feig DI, Graham NM, Hui-Stickle S, Goldstein SL. 3–5 year longitudinal follow-up of pediatric patients after acute renal failure. Kidney Int. 2006;69(1):184–9.
Askenazi DJ, Griffin R, McGwin G, Carlo W, Ambalavanan N. Acute kidney injury is independently associated with mortality in very low birthweight infants: a matched case–control analysis. Pediatric Nephrol. 2009;24(5):991–7.
Sanchez-Pinto LN, Goldstein SL, Schneider JB, Khemani RG. Association between progression and improvement of acute kidney injury and mortality in critically ill children. Pediatr Crit Care Med. 2015;16(8):703–10.
Selewski DT, Cornell TT, Heung M, Troost JP, Ehrmann BJ, Lombel RM, et al. Validation of the KDIGO acute kidney injury criteria in a pediatric critical care population. Intensive Care Med. 2014;40(10):1481–8.
Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Working Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int. 2012;Supplement 2:1–138.
Slater MB, Gruneir A, Rochon PA, Howard AW, Koren G, Parshuram CS. Risk factors of acute kidney injury in critically ill children. Pediatr Crit Care Med. 2016;17(9):e391–8.
Moffett BS, Goldstein SL. Acute kidney injury and increasing nephrotoxic-medication exposure in noncritically-ill children. Clin J Am Soc Nephrol. 2011;6(4):856–63.
Cullen DJ, Sweitzer BJ, Bates DW, Burdick E, Edmondson A, Leape LL. Preventable adverse drug events in hospitalized patients: a comparative study of intensive care and general care units. Crit Care Med. 1997;25(8):1289–97.
Rhone ET, Carmody JB, Swanson JR, Charlton JR. Nephrotoxic medication exposure in very low birth weight infants. J Matern Fetal Neonatal Med. 2014;27(14):1485–90.
Sinclair EA, Yenokyan G, McMunn A, Fadrowski JJ, Milstone AM, Lee CK. Factors associated with acute kidney injury in children receiving vancomycin. Ann Pharmacother. 2014;48(12):1555–62.
Totapally BR, Machado J, Lee H, Paredes A, Raszynski A. Acute kidney injury during vancomycin therapy in critically ill children. Pharmacotherapy. 2013;33(6):598–602.
Moffett BS, Hilvers PS, Dinh K, Arikan AA, Checchia P, Bronicki R. Vancomycin-associated acute kidney injury in pediatric cardiac intensive care patients. Congenit Heart Dis. 2015;10(1):E6–10.
Rao S, Abzug MJ, Carosone-Link P, Peterson T, Child J, Siparksy G, et al. Intravenous acyclovir and renal dysfunction in children: a matched case control study. J Pediatr. 2015;166(6):1462–8e1–4.
Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P. Acute renal failure—definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):R204–12.
Schwartz GJ, Haycock GB, Spitzer A. Plasma creatinine and urea concentration in children: normal values for age and sex. J Pediatr. 1976;88(5):828–30.
Szklo M, Nieto J. Epidemiology: beyond the basics, 2nd ed. Burlington: Jones and Bartlett Learning; 2006.
World Health Organization. International statistical classification of diseases and related health problems 10th revision. 2010 [cited 2013 February 12, 2013]; Available from: http://apps.who.int/classifications/icd10/browse/2010/en.
Pollack MM, Ruttimann UE, Getson PR. Pediatric risk of mortality (PRISM) score. Crit Care Med. 1988;16(11):1110–6.
Leteurtre S, Martinot A, Duhamel A, Gauvin F, Grandbastien B, Nam TV, et al. Development of a pediatric multiple organ dysfunction score: use of two strategies. Med Decis Making. 1999;19(4):399–410.
Knoderer CA, Gritzman AL, Nichols KR, Wilson AC. Late-occurring vancomycin-associated acute kidney injury in children receiving prolonged therapy. Ann Pharmacother. 2015;49(10):1113–9.
Aydin SI, Seiden HS, Blaufox AD, Parnell VA, Choudhury T, Punnoose A, et al. Acute kidney injury after surgery for congenital heart disease. Ann Thorac Surg. 2012;94(5):1589–95.
Morgan CJ, Zappitelli M, Robertson CM, Alton GY, Sauve RS, Joffe AR, et al. Risk factors for and outcomes of acute kidney injury in neonates undergoing complex cardiac surgery. J Pediatr. 2013;162(1):120–7e1.
Viswanathan S, Manyam B, Azhibekov T, Mhanna MJ. Risk factors associated with acute kidney injury in extremely low birth weight (ELBW) infants. Pediatric Nephrol. 2012;27(2):303–11.
Bailey D, Phan V, Litalien C, Ducruet T, Merouani A, Lacroix J, et al. Risk factors of acute renal failure in critically ill children: a prospective descriptive epidemiological study. Pediatr Crit Care Med. 2007;8(1):29–35.
Roy AK, Mc Gorrian C, Treacy C, Kavanaugh E, Brennan A, Mahon NG, et al. A comparison of traditional and novel definitions (RIFLE, AKIN, and KDIGO) of acute kidney injury for the prediction of outcomes in acute decompensated heart failure. Cardiorenal Med. 2013;3(1):26–37.
Rodrigues FB, Bruetto RG, Torres US, Otaviano AP, Zanetta DM, Burdmann EA. Incidence and mortality of acute kidney injury after myocardial infarction: a comparison between KDIGO and RIFLE criteria. PLoS One. 2013;8(7):e69998.
Bastin AJ, Ostermann M, Slack AJ, Diller GP, Finney SJ, Evans TW. Acute kidney injury after cardiac surgery according to risk/injury/failure/loss/end-stage, acute kidney injury network, and kidney disease: improving global outcomes classifications. J Crit Care. 2013;28(4):389–96.
Sutherland SM, Byrnes JJ, Kothari M, Longhurst CA, Dutta S, Garcia P, et al. AKI in hospitalized children: comparing the pRIFLE, AKIN, and KDIGO definitions. Clin J Am Soc Nephrol. 2015;10(4):554–61.
Siew ED, Matheny ME, Ikizler TA, Lewis JB, Miller RA, Waitman LR, et al. Commonly used surrogates for baseline renal function affect the classification and prognosis of acute kidney injury. Kidney Int. 2010;77(6):536–42.
Zappitelli M, Parikh CR, Akcan-Arikan A, Washburn KK, Moffett BS, Goldstein SL. Ascertainment and epidemiology of acute kidney injury varies with definition interpretation. Clin J Am Soc Nephrol. 2008;3(4):948–54.
Zavada J, Hoste E, Cartin-Ceba R, Calzavacca P, Gajic O, Clermont G, et al. A comparison of three methods to estimate baseline creatinine for RIFLE classification. Nephrol Dial Transpl. 2010;25(12):3911–8.
Thongprayoon C, Cheungpasitporn W, Harrison AM, Kittanamongkolchai W, Ungprasert P, Srivali N, et al. The comparison of the commonly used surrogates for baseline renal function in acute kidney injury diagnosis and staging. BMC Nephrol. 2016;17:6.
Girardeau Y, Trivin C, Durieux P, Le Beller C, Louet Agnes LL, Neuraz A, et al. Detection of drug–drug interactions inducing acute kidney injury by electronic health records mining. Drug Saf. 2015;38(9):799–809.
Menon S, Kirkendall ES, Nguyen H, Goldstein SL. Acute kidney injury associated with high nephrotoxic medication exposure leads to chronic kidney disease after 6 months. J Pediatr. 2014;165(3):522–7e2.
Ali T, Khan I, Simpson W, Prescott G, Townend J, Smith W, et al. Incidence and outcomes in acute kidney injury: a comprehensive population-based study. J Am Soc Nephrol. 2007;18(4):1292–8.
Hsu RK, McCulloch CE, Dudley RA, Lo LJ, Hsu CY. Temporal changes in incidence of dialysis-requiring AKI. J Am Soc Nephrol. 2013;24(1):37–42.
Yamout H, Levin ML, Rosa RM, Myrie K, Westergaard S. Physician prevention of acute kidney injury. Am J Med. 2015;128(9):1001–6.
Goldstein SL, Kirkendall E, Nguyen H, Schaffzin JK, Bucuvalas J, Bracke T, et al. Electronic health record identification of nephrotoxin exposure and associated acute kidney injury. Pediatrics. 2013;132(3):e756–67.
Acknowledgements
The authors would like to thank Dr. Valeria Rac, Dr. Joseph Amuah, Helena Frndova, Winnie Seto, Tony Pyle, Traci Peggie, Sarah Ashley, and Christina Stevancec for their advice and assistance. Morgan Slater was supported through a Canadian Institutes of Health Research Frederick Banting and Charles Best Canada Graduate Scholarship, the Ontario Student Opportunity Trust Fund—Hospital for Sick Children Foundation Student Scholarship Program, and an Ontario Graduate Scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure of potential conflicts of interest
All authors (M. Slater, A. Gruneir, P. Rochon, A. Howard, G. Koren, C. Parshuram) declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Funding sources
This work was supported by the following scholarships awarded to Morgan Slater: a Canadian Institutes of Health Research Frederick Banting and Charles Best Canada Graduate Scholarship, the Ontario Student Opportunity Trust Fund—The Hospital for Sick Children Foundation Student Scholarship Program, and an Ontario Graduate Scholarship. Dr. C Parshuram received operational funding from the Canadian Institutes of Health Research that supported completion of the study and was administered by the SickKids Research Institute.
Rights and permissions
About this article
Cite this article
Slater, M.B., Gruneir, A., Rochon, P.A. et al. Identifying High-Risk Medications Associated with Acute Kidney Injury in Critically Ill Patients: A Pharmacoepidemiologic Evaluation. Pediatr Drugs 19, 59–67 (2017). https://doi.org/10.1007/s40272-016-0205-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40272-016-0205-1