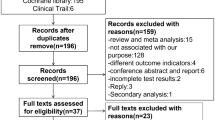
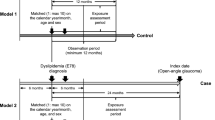
Abstract
Background and Objective
There is a relative lack of head-to-head comparisons of denosumab against other osteoporosis drugs on safety. We aimed to explore ocular outcomes in patients with osteoporosis initiating denosumab vs zoledronic acid.
Methods
We conducted a cohort study using claims data (2010–15) from two large US commercial insurance databases including patients with osteoporosis who were aged 50 years or older and initiators of denosumab or zoledronic acid. The primary outcomes were (1) receipt of cataract surgery and development of (2) wet age-related macular degeneration and (3) dry age-related macular degeneration within 365 days after initiation of denosumab vs zoledronic acid. Propensity score fine stratification and weighting were used to control for potential confounding, and we calculated the incidence rate and hazard ratio for each outcome in the cohorts. The estimates from the two databases were combined with a fixed-effects model meta-analysis.
Results
The study cohort included 50,821 denosumab and 67,471 zoledronic acid initiators. In the propensity score-weighted analysis, compared to zoledronic acid use, denosumab was associated with a modestly decreased risk of undergoing cataract surgery (hazard ratio 0.91; 95% confidence interval 0.85–0.98) but not with the risk of wet age-related macular degeneration (hazard ratio 1.29; 95% confidence interval 0.99–1.70) or dry age-related macular degeneration (hazard ratio 1.03; 95% confidence interval 0.98–1.09).
Conclusions
In this large population-based cohort study of 118,292 patients with osteoporosis, initiation of denosumab was associated with a modestly decreased risk of cataract surgery vs zoledronic acid. The risk of age-related macular degeneration was similar between the two drugs.


Similar content being viewed by others
References
Black DM, Rosen CJ. Clinical practice: postmenopausal osteoporosis. N Engl J Med. 2016;374(3):254–62.
Melton LJ 3rd. Who has osteoporosis? A conflict between clinical and public health perspectives. J Bone Miner Res. 2000;15(12):2309–14.
Cummings SR, Melton LJ. Epidemiology and outcomes of osteoporotic fractures. Lancet. 2002;359(9319):1761–7.
Khosla S, Hofbauer LC. Osteoporosis treatment: recent developments and ongoing challenges. Lancet Diabetes Endocrinol. 2017;5(11):898–907.
Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, et al. Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. N Engl J Med. 2007;356(18):1809–22.
Deeks ED, Perry CM. Zoledronic acid: a review of its use in the treatment of osteoporosis. Drugs Aging. 2008;25(11):963–86.
Wu J, Zhang Q, Yan G, Jin X. Denosumab compared to bisphosphonates to treat postmenopausal osteoporosis: a meta-analysis. J Orthop Surg Res. 2018;13(1):194.
Choi NK, Solomon DH, Tsacogianis TN, Landon JE, Song HJ, Kim SC. Comparative safety and effectiveness of denosumab versus zoledronic acid in patients with osteoporosis: a cohort study. J Bone Miner Res. 2017;32(3):611–7.
Gnant M, Pfeiler G, Steger GG, Egle D, Greil R, Fitzal F, et al. Adjuvant denosumab in postmenopausal patients with hormone receptor-positive breast cancer (ABCSG-18): disease-free survival results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019;20(3):339–51.
Papapetrou PD. Bisphosphonate-associated adverse events. Hormones (Athens, Greece). 2009;8(2):96–110.
Fraunfelder FW, Fraunfelder FT, Jensvold B. Scleritis and other ocular side effects associated with pamidronate disodium. Am J Ophthalmol. 2003;135(2):219–22.
Patel DV, Horne A, House M, Reid IR, McGhee CN. The incidence of acute anterior uveitis after intravenous zoledronate. Ophthalmology. 2013;120(4):773–6.
Patel DV, Bolland M, Nisa Z, Al-Abuwsi F, Singh M, Horne A, et al. Incidence of ocular side effects with intravenous zoledronate: secondary analysis of a randomized controlled trial. Osteoporos Int. 2015;26(2):499–503.
Woo TC, Joseph DJ, Wilkinson R. Serious ocular complications of zoledronate. Clin Oncol (R Coll Radiol). 2006;18(7):545–6.
Kaur H, Uy C, Kelly J, Moses AM. Orbital inflammatory disease in a patient treated with zoledronate. Endocr Pract. 2011;17(4):e101–3.
Chehade LK, Curragh D, Selva D. Bisphosphonate-induced orbital inflammation: more common than once thought? Osteoporos Int. 2019;30(5):1117–20.
Desai RJ, Rothman KJ, Bateman BT, Hernandez-Diaz S, Huybrechts KF. A propensity-score-based fine stratification approach for confounding adjustment when exposure is infrequent. Epidemiology. 2017;28(2):249–57.
Saito T, Sterbenz JM, Malay S, Zhong L, MacEachern MP, Chung KC. Effectiveness of anti-osteoporotic drugs to prevent secondary fragility fractures: systematic review and meta-analysis. Osteoporos Int. 2017;28(12):3289–300.
Mammo Z, Guo M, Maberley D, Matsubara J, Etminan M. Oral bisphosphonates and risk of wet age-related macular degeneration. Am J Ophthalmol. 2016;168:62–7.
Hewitt RE, Lissina A, Green AE, Slay ES, Price DA, Sewell AK. The bisphosphonate acute phase response: rapid and copious production of proinflammatory cytokines by peripheral blood gd T cells in response to aminobisphosphonates is inhibited by statins. Clin Exp Immunol. 2005;139(1):101–11.
Or C, Cui J, Matsubara J, Forooghian F. Pro-inflammatory and anti-angiogenic effects of bisphosphonates on human cultured retinal pigment epithelial cells. Br J Ophthalmol. 2013;97(8):1074–8.
Honda S, Nagai T, Kondo N, Fukuda M, Kusuhara S, Tsukahara Y, et al. Therapeutic effect of oral bisphosphonates on choroidal neovascularization in the human eye. J Ophthalmol. 2010. https://doi.org/10.1155/2010/206837(Epub 2010 Jul 25).
Tang D, Borchman D, Yappert MC, Vrensen GF, Rasi V. Influence of age, diabetes, and cataract on calcium, lipid-calcium, and protein–calcium relationships in human lenses. Invest Ophthalmol Vis Sci. 2003;44(5):2059–66.
Augoulea A, Tsakonas E, Triantafyllopoulos I, Rizos D, Armeni E, Tsoltos N, et al. Comparative effects of denosumab or bisphosphonate treatment on bone mineral density and calcium metabolism in postmenopausal women. J Musculoskelet Neuronal Interact. 2017;17(1):444–9.
Makras P, Polyzos SA, Papatheodorou A, Kokkoris P, Chatzifotiadis D, Anastasilakis AD. Parathyroid hormone changes following denosumab treatment in postmenopausal osteoporosis. Clin Endocrinol (Oxf). 2013;79(4):499–503.
Breen TL, Shane E. Prolonged hypocalcemia after treatment with zoledronic acid in a patient with prostate cancer and vitamin D deficiency. J Clin Oncol. 2004;22(8):1531–2.
Yoshida K, Solomon DH, Kim SC. Active-comparator design and new-user design in observational studies. Nat Rev Rheumatol. 2015;11(7):437–41.
Reid IR, Gamble GD, Mesenbrink P, Lakatos P, Black DM. Characterization of and risk factors for the acute-phase response after zoledronic acid. J Clin Endocrinol Metab. 2010;95(9):4380–7.
Fraunfelder FW, Fraunfelder FT. Adverse ocular drug reactions recently identified by the National Registry of Drug-Induced Ocular Side Effects. Ophthalmology. 2004;111(7):1275–9.
Umunakwe OC, Herren D, Kim SJ, Kohanim S. Diffuse ocular and orbital inflammation after zoledronate infusion: case report and review of the literature. Digit J Ophthalmol. 2017;23(4):109–12.
Author information
Authors and Affiliations
Contributions
Hemin Lee verified the analytical methods and drafted the manuscript. Yinzhu Jin conducted the statistical analysis and drafted the manuscript. Miin Roh contributed to the study conception and reviewed the manuscript for important intellectual content. Theodore N. Tsacogianis conducted the statistical analysis. Sangshin Park verified the analytical methods and reviewed the manuscript for important intellectual content. Nam-Kyong Choi contributed to the study conception, verified the analytical methods, and reviewed the manuscript for important intellectual content. Seoyoung C. Kim supervised the project, contributed to the study conception, verified the analytical methods, and reviewed the manuscript for important intellectual content.
Corresponding author
Ethics declarations
Funding
This study was supported by the Division of Pharmacoepidemiology and Pharmacoeconomics at Brigham and Women’s Hospital.
Conflict of interest
Seoyoung C. Kim has received research grants to the Brigham and Women’s Hospital from Pfizer, Roche, and Bristol-Myers Squibb for unrelated topics. Hemin Lee, Yinzhu Jin, Miin Roh, Theodore N. Tsacogianis, Sangshin Park, and Nam-Kyong Choi have no conflicts of interest that are directly relevant to the content of this article.
Ethics approval
Personal identifiers were removed from the dataset before the analysis to protect subject confidentiality. Therefore, patient informed consent was not required. The study protocol was approved by the Institutional Review Board of Brigham and Women’s Hospital.
Consent to participate
Not applicable.
Prior posting and presentation
This work is the sole product of the authors and has been presented at the 2018 International Conference on Pharmacoepidemiology and Therapeutic Risk Management.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, H., Jin, Y., Roh, M. et al. Risk of Cataract Surgery and Age-Related Macular Degeneration After Initiation of Denosumab vs Zoledronic Acid for Osteoporosis: A Multi-Database Cohort Study. Drugs Aging 37, 311–320 (2020). https://doi.org/10.1007/s40266-020-00745-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-020-00745-2