Abstract
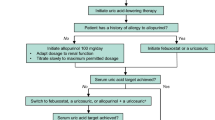
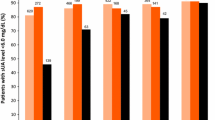
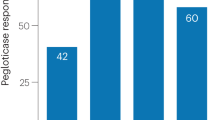
Lesinurad (Zurampic®) is an oral selective inhibitor of the URAT1 and OAT4 uric acid (UA) transporters of the kidney, via which it inhibits UA reabsorption and thus increases renal UA excretion and lowers serum UA (sUA) levels. Lesinurad 200 mg once daily is indicated for use in combination with a xanthine oxidase inhibitor (XOI) to treat hyperuricaemia in adults with gout who have not achieved target sUA levels with an XOI alone. Approval was based on three 12-month phase 3 trials that evaluated lesinurad in combination with allopurinol in adults with gout inadequately responsive to allopurinol (CLEAR 1 and 2) and in combination with febuxostat in adults with tophaceous gout (CRYSTAL). The target sUA level of <6 mg/dL at 6 months (primary endpoint) was achieved by significantly more lesinurad plus allopurinol than placebo plus allopurinol recipients in the CLEAR trials. In CRYSTAL (which enrolled patients regardless of prior XOI experience, and included 3 weeks of febuxostat before randomization), the proportion of patients who achieved an sUA target of <5 mg/dL did not reach statistical significance between lesinurad plus febuxostat and placebo plus febuxostat at 6 months (primary endpoint), although significantly favoured the lesinurad plus febuxostat group at 12 months. Notably, the sUA target of <5 mg/dL at 6 months was met with lesinurad plus febuxostat in the CRYSTAL subgroup that had uncontrolled hyperuricaemia at baseline, despite having received febuxostat pre-randomization. Lesinurad plus XOI regimens were generally not associated with improvements in flares and tophi in these trials, although clinical benefit became more apparent in 12-month extension studies; the regimens were also generally well tolerated. Thus, lesinurad, in combination with an XOI, is an emerging option for the treatment of hyperuricaemia in adults with gout who have not achieved target sUA levels with an XOI alone.

Similar content being viewed by others
References
Dalbeth N, Merriman TR, Stamp LK. Gout. Lancet. 2016;388(10055):2039–52.
Perez-Ruiz F, Dalbeth N, Bardin T. A review of uric acid, crystal deposition disease, and gout. Adv Ther. 2015;32(1):31–41.
Richette P, Doherty M, Pascual E, et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann Rheum Dis. 2017;76(1):29–42.
National Kidney Foundation. Gout, hyperuricemia & chronic kidney disease. 2016. https://www.kidney.org/atoz/content/gout. Accessed 24 Mar 2017.
Khanna P, Tafesse E, Baumgartner S, et al. Comparing the burden of illness of patients with tophaceous and non-tophaceous gout in France, Germany, Italy, Spain, UK, and USA [abstract no. 226]. Arthritis Rheumatol. 2016;68(Suppl 10).
Nyberg F, Horne L, Morlock R, et al. Comorbidity burden in trial-aligned patients with established gout in Germany, UK, US, and France: a retrospective analysis. Adv Ther. 2016;33(7):1180–98.
Khanna D, Fitzgerald JD, Khanna PP, et al. 2012 American College of Rheumatology guidelines for management of gout. Part 1: systematic nonpharmacologic and pharmacologic therapeutic approaches to hyperuricemia. Arthritis Care Res. 2012;64(10):1431–46.
Zurampic® (lesinurad) tablets, for oral use: US prescribing information. 2016. http://irwdpi.com/zurampic/zurampic.pdf. Accessed 3 Apr 2017.
Grünenthal GmbH. Zurampic 200 mg film-coated tablets: EU summary of product characteristics. 2016. http://www.ema.europa.eu/. Accessed 24 Mar 2017.
Miner J, Tan PK, Hyndman D, et al. Lesinurad, a novel, oral compound for gout, acts to decrease serum uric acid through inhibition of urate transporters in the kidney. Arthritis Res Ther. 2016;18(1):214.
Shen Z, Yeh LT, Wallach K, et al. In vitro and in vivo interaction studies between lesinurad, a selective urate reabsorption inhibitor, and major liver or kidney transporters. Clin Drug Investig. 2016;36(6):443–52.
Shen Z, Rowlings C, Kerr B, et al. Pharmacokinetics, pharmacodynamics, and safety of lesinurad, a selective uric acid reabsorption inhibitor, in healthy adult males. Drug Des Devel Ther. 2015;9:3423–34.
Perez-Ruiz F, Sundy JS, Miner JN, et al. Lesinurad in combination with allopurinol: results of a phase 2, randomised, double-blind study in patients with gout with an inadequate response to allopurinol. Ann Rheum Dis. 2016;75(6):1074–80.
Fleischmann R, Kerr B, Yeh LT, et al. Pharmacodynamic, pharmacokinetic and tolerability evaluation of concomitant administration of lesinurad and febuxostat in gout patients with hyperuricaemia. Rheumatology. 2014;53(12):2167–74.
Saag KG, Fitz-Patrick D, Kopicko J, et al. Lesinurad combined with allopurinol. A randomized, double-blind, placebo-controlled study in gout patients with an inadequate response to standard-of-care allopurinol (a US-based study). Arthritis. Rheumatol. 2017;69(1):203–12.
Bardin T, Keenan RT, Khanna PP, et al. Lesinurad in combination with allopurinol: a randomised, double-blind, placebo-controlled study in patients with gout with inadequate response to standard of care (the multinational CLEAR 2 study). Ann Rheum Dis. 2016. doi:10.1136/annrheumdis-2016-209213.
Dalbeth N, Jones G, Terkeltaub R, et al. Lesinurad, a novel selective uric acid reabsorption inhibitor, in combination with febuxostat, in patients with tophaceous gout: the CRYSTAL phase III clinical trial [abstract no. SAT0329 plus poster]. Ann Rheum Dis. 2015;74(Suppl 2):778.
Saag KG, Becker MA, Storgard C, et al. Examination of serum uric acid (SUA) lowering and safety with extended treatment with lesinurad and allopurinol in subjects with gout [abstract no. THU0495]. Ann Rheum Dis. 2016;75(Suppl 2):371.
Bardin T, Dalbeth N, Terkeltaub R, et al. Clinical response of tophus and flares to extended use of lesinurad in combination with a xanthine oxidase inhibitor in patients with gout [abstract no. 209]. Arthritis Rheumatol. 2016;68(Suppl 10).
Shen Z, Gillen M, Tieu K, et al. Supratherapeutic dose evaluation and effect of lesinurad on cardiac repolarization: a thorough QT/QTc study. Drug Des Devel Ther. 2016;10:3509–17.
Gillen M, Valdez S, Zhou D, et al. Effects of renal function on pharmacokinetics and pharmacodynamics of lesinurad in adult volunteers. Drug Des Devel Ther. 2016;10:3555–62.
US National Institutes of Health. ClinicalTrials.gov identifier NCT01510769. 2016. https://clinicaltrials.gov. Accessed 24 Mar 2017.
Dalbeth N, Jones G, Terkeltaub R, et al. Efficacy and safety in patients with tophaceous gout receiving lesinurad and febuxostat combination therapy: interim analysis of an extension study [abstract no. 2352]. Arthritis Rheumatol. 2015;67(Suppl 10).
Saag KG, Bardin T, So A, et al. Analysis of gout subjects receiving lesinurad and allopurinol combination therapy by baseline renal function [abstract no. 2112]. Arthritis Rheumatol. 2015;67(Suppl 10).
Dalbeth N, Jones G, Terkeltaub R, et al. Lesinurad and febuxostat combination therapy: analysis of treatment based on patient baseline renal function [abstract no. 2353]. Arthritis Rheumatol. 2015;67(Suppl 10).
Khanna PP, Baumgartner S, Robinson J. Lesinurad, a selective uric acid reabsorption inhibitor, in combination with xanthane oxidase inhibitors: SUA and flares composite endpoint from phase III studies in gout patients (CLEAR 1, CLEAR 2 and CRYSTAL) [abstract no. THU0527]. Ann Rheum Dis. 2016;75(Suppl 2):382–3.
Becker MA, Keenan RT, Khanna P, et al. Integrated safety of lesinurad, a novel uric acid reabsorption inhibitor for the treatment of gout [abstract no. 207]. Arthritis Rheumatol. 2016;68(Suppl 10).
Terkeltaub R, Malamet R, Bos K, et al. Renal safety of lesinurad: a pooled analysis of phase III and extension studies [abstract no. 206]. Arthritis Rheumatol. 2016;68(Suppl 10).
Sattui SE, Gaffo AL. Treatment of hyperuricemia in gout: current therapeutic options, latest developments and clinical implications. Ther Adv Musculoskelet Dis. 2016;8(4):145–59.
Keenan RT, Schlesinger N. New and pipeline drugs for gout. Curr Rheumatol Rep. 2016;18(6):32.
Terkeltaub R. Emerging uricosurics for gout. Expert Rev Clin Pharmacol. 2016:1–3. doi:10.1080/17512433.2017.1271709.
Lepist EI, Ray AS. Renal drug-drug interactions: what we have learned and where we are going. Expert Opin Drug Metab Toxicol. 2012;8(4):433–48.
Robbins N, Koch SE, Tranter M, et al. The history and future of probenecid. Cardiovasc Toxicol. 2012;12(1):1–9.
Terkeltaub R, Perez-Ruiz F, Storgard C, et al. Relationship between sustained lowering of serum urate levels and improvements in gout flares and tophus area: pooled exploratory analysis of gout subjects receiving lesinurad and xanthine oxidase inhibitor combination therapy [abstract no. SAT0313]. Ann Rheum Dis. 2015;74(Suppl 2):771–2.
Wood R, Fermer S, Ramachandran S, et al. Patients with gout treated with conventional urate-lowering therapy: association with disease control, health-related quality of life, and work productivity. J Rheumatol. 2016;43(10):1897–903.
Khanna P, Baumgartner S, Robinson J. The impact of gout flares on patient-reported assessments of pain and disability [abstract no. FRI0589 plus poster]. Ann Rheum Dis. 2016;75(Suppl 2):655–6.
Qaseem A, Harris RP, Forciea MA. Management of acute and recurrent gout: a clinical practice guideline from the American College of Physicians. Ann Intern Med. 2017;166(1):58–68.
Singh JA, Storgard C, Baumgartner S, et al. Use of high-dose allopurinol to reach serum uric acid targets in patients with gout across multiple countries [abstract no. 1186]. In: ACR/ARHP Annual Meeting; 2013.
Becker MA, Fitz-Patrick D, Choi HK, et al. An open-label, 6-month study of allopurinol safety in gout: The LASSO study. Semin Arthritis Rheum. 2015;45(2):174–83.
Stamp LK, Chapman PT. Urate-lowering therapy: current options and future prospects for elderly patients with gout. Drugs Aging. 2014;31(11):777–86.
Bach MH, Simkin PA. Uricosuric drugs: the once and future therapy for hyperuricemia? Curr Opin Rheumatol. 2014;26(2):169–75.
Stack A, Blak B, Johnson M, et al. Association of gout with risk of advanced chronic kidney disease [abstract no. 3188]. Arthritis Rheumatol. 2016;68(Suppl 10).
Borghi C, Nuevo J, Medina J, et al. Association between serum uric acid levels and cardiovascular risk. A post hoc analysis of the European Cardiovascular Risk Patients: Disease Prevention and Management in Usual Daily Practice (EURIKA) Study [poster no. FRI0337]. In: 16th EULAR Annual European Congress of Rheumatology; 2015.
Baumgartner S, Morlock R. Hyperuricemia and the risk for cardiovascular disease: individual patient meta-analysis of 77,045 individuals enrolled in 15 epidemiologic studies [poster no. FRI0591]. In: EULAR Annual European Congress of Rheumatology; 2016.
Cheetham TC, Nichols GA, Harrold LR, et al. The association between serum uric acid levels and acute myocardial infarction in incident gout patients [abstract no. PMS18]. In: ISPOR 21st Annual International Meeting; 2016.
Acknowledgements
During the peer review process, the manufacturer of lesinurad was offered the opportunity to review this article. Changes resulting from comments received were made on the basis of scientific and editorial merit.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Conflict of interest
Emma Deeks is a salaried employee of Adis/Springer, is responsible for the article content and declares no relevant conflicts of interest.
Additional information
The manuscript was reviewed by: S. Chohan, Arizona Arthritis and Rheumatology Associates, Phoenix, AZ, USA; A. L. Gaffo, University of Alabama at Birmingham and Rheumatology Section, Birmingham VA Medical Center, Birmingham, AL, USA; P. Khanna, Medicine-Rheumatology, University of Michigan, Ann Arbor, MI, USA.
Rights and permissions
About this article
Cite this article
Deeks, E.D. Lesinurad: A Review in Hyperuricaemia of Gout. Drugs Aging 34, 401–410 (2017). https://doi.org/10.1007/s40266-017-0461-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-017-0461-y