Abstract
Avacincaptad pegol (IZERVAY™; formerly Zimura®) is a complement C5 inhibitor that is being developed by IVERIC Bio, an Astellas company, for the treatment of geographic atrophy secondary to age-related macular degeneration. Avacincaptad pegol recently received approval for the treatment of adults with geographic atrophy secondary to age-related macular degeneration. This article summarizes the milestones in the development of avacincaptad pegol leading to this first approval for geographic atrophy secondary to age-related macular degeneration.
Similar content being viewed by others
References
IVERIC Bio. IZERVAY™ (avacincaptad pegol intravitreal solution): US prescribing information. 2023. https://ivericbio.com. Accessed 12 Sep 2023.
Jaffe GJ, Westby K, Csaky KG, et al. C5 inhibitor avacincaptad pegol for geographic atrophy due to age-related macular degeneration: a randomized pivotal phase 2/3 trial. Ophthalmology. 2021;128(4):576–86.
IVERIC Bio. Iveric Bio receives U.S. FDA approval for IZERVAY™ (avacincaptad pegol intravitreal solution), a new treatment for geographic atrophy [media release]. 5 Aug 2023. https://www.astellas.com/en/news/28281.
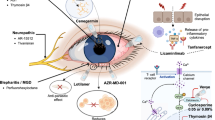
IVERIC Bio. Our science. 2023. https://ivericbio.com/our-science. Accessed 12 Sep 2023.
Ophthotech Corporation. Ophthotech raises $36 million; in-licenses two compounds for macular degeneration [media release]. 21 Feb 2008. https://www.fiercebiotech.com/biotech/press-release-ophthotech-raises-36-million-licenses-two-compounds-for-macular-degeneration.
IVERIC bio. Ophthotech transitions to a gene therapy focused company developing treatments for orphan inherited retinal diseases and changes name to IVERIC bio [media release]. 16 Apr 2019. https://investors.ivericbio.com/news-releases/news-release-details/ophthotech-transitions-gene-therapy-focused-company-developing.
United States Securities and Exchange Commission. Form 10-K for IVERIC bio, Inc. 2020. https://www.sec.gov/ix?doc=/Archives/edgar/data/1410939/000141093921000007/opht-20201231.htm. Accessed 12 Sep 2023.
IVERIC Bio. Iveric Bio and DelSiTech enter exclusive agreement for development of sustained release Zimura® [media release]. 5 Jul 2022. https://ivericbio.com/iveric-bio-and-delsitech-enter-exclusive-agreement-for-development-of-sustained-release-zimura.
IVERIC Bio. Astellas enters into definitive agreement to acquire Iveric Bio [media release]. 30 Apr 2023. https://ivericbio.com/astellas-enters-into-definitive-agreement-to-acquire-iveric-bio.
United States Securities and Exchange Commission. Form 10-K for IVERIC bio, Inc. 2022. https://www.sec.gov/ix?doc=/Archives/edgar/data/1410939/000141093923000025/isee-20221231.htm. Accessed 12 Sep 2023.
OphthoTech Corporation. Ophthotech announces results from phase 2a safety trial of Zimura® in combination with Lucentis® in wet age-related macular degeneration [media release]. 16 Nov 2018. https://www.businesswire.com/news/home/20181112005203/en/Ophthotech-Announces-Results-from-Phase-2a-Safety-Trial-of-Zimura%C2%AE-in-Combination-with-Lucentis%C2%AE-in-Wet-Age-Related-Macular-Degeneration.
Patel SS, Lally DR, Hsu J, et al. Avacincaptad pegol for geographic atrophy secondary to age-related macular degeneration: 18-month findings from the GATHER1 trial. Eye (Lond). 2023. https://doi.org/10.1038/s41433-023-02497-w.
Jaffe GJ, Zemborain Z, Farsiu S, et al. Evaluation of GA lesion growth by minimum distance to the fovea center: post hoc analysis of the GATHER1 trial [presentation]. Retina World Conference. 2022.
Khanani AM, Patel SS, Staurenghi G, et al. GATHER2 pivotal phase 3 study results: efficacy of intravitreal avacincaptad pegol in geographic atrophy [presentation]. In: American Academy of Ophthalmology Annual Meeting. 2022.
Heier JS, Khanani AM, Eichenbaum D, et al. GATHER2 pivotal phase 3 study results: safety of intravitreal avacincaptad pegol in geographic atrophy [presentation]. In: American Academy of Ophthalmology Annual Meeting. 2022.
Astellas Pharma. European Medicines Agency accepts Iveric Bio’s marketing authorization application for avacincaptad pegol for geographic atrophy [media release]. 18 Aug 2023. https://www.astellas.com/en/news/28446.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Authorship and Conflict of Interest
During the peer review process the manufacturer of the agent under review was offered an opportunity to comment on the article. Changes resulting from any comments received were made by the authors on the basis of scientific completeness and accuracy. Connie Kang is a salaried employee of Adis International Ltd/Springer Nature, and declares no relevant conflicts of interest. All authors contributed to this article and are responsible for its content.
Ethics Approval, Consent to Participate, Consent to Publish, Availability of Data and Material, Code Availability
Not applicable.
Additional information
This profile has been extracted and modified from the AdisInsight database. AdisInsight tracks drug development worldwide through the entire development process, from discovery, through pre-clinical and clinical studies to market launch and beyond.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kang, C. Avacincaptad Pegol: First Approval. Drugs 83, 1447–1453 (2023). https://doi.org/10.1007/s40265-023-01948-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-023-01948-8