Abstract
Objective
To evaluate and compare the risk of erectile dysfunction (ED) associated with the use of allopurinol and febuxostat in adult male gout patients.
Methods
We conducted a cohort study using TriNetX (Cambridge, MA, USA), a global federated health research network that provides real-time electronic medical record datasets. We analyzed and compared the associated risk of ED in gout patients who started taking allopurinol or febuxostat within 12 months. Propensity score matching was performed to adjust for demographic variables, comorbidities, and medication use. Kaplan–Meier analysis was used to estimate the probability of the outcome of interest. The hazard ratio (HR) and associated confidence intervals were calculated along with the proportionality test using R's Survival Package v3.2-3.
Results
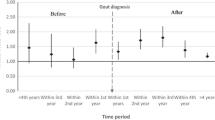
We identified 679,862 patients with gout among 107,517,445 patients in the database. Of these patients, 24,000 were treated with febuxostat and 299,726 with allopurinol. After propensity matching, 9075 patients receiving febuxostat without allopurinol (febuxostat group) and 9075 corresponding patients receiving allopurinol without febuxostat (allopurinol group) were analyzed for comparison. Among all male patients over 19 years of age, febuxostat was associated with a significantly higher risk of ED versus allopurinol (HR 1.354; 95% confidence interval (CI) 1.003–1.829; log rank test, p = 0.047). After subgroup analysis, in gout patients aged 19–64 years, a significantly higher incidence of ED was observed in the febuxostat group than in the allopurinol group (HR 2.002, 95% CI 1.282–3.126). The risk of ED did not differ significantly between the allopurinol and febuxostat groups in gout patients older than 65 years.
Conclusions
Febuxostat may be associated with a higher risk of ED than allopurinol in adult male patients with gout. Future large-scale prospective studies are warranted to confirm our results.



Similar content being viewed by others
Data availability statement
Data availability statement has been introduced in the section “Availability of data and materials (data sharing statement)”.
References
Jin M, Yang F, Yang I, et al. Uric acid, hyperuricemia and vascular diseases. Front Biosci (Landmark Ed). 2012;17:656–69.
Singh G, Lingala B, Mithal A. Gout and hyperuricaemia in the USA: prevalence and trends. Rheumatology (Oxford). 2019;58:2177–80.
Perez-Ruiz F, Sundy JS, Miner JN, et al. Lesinurad in combination with allopurinol: results of a phase 2, randomised, double-blind study in patients with gout with an inadequate response to allopurinol. Ann Rheum Dis. 2016;75:1074–80.
Kuo CF, Grainge MJ, Mallen C, et al. Rising burden of gout in the UK but continuing suboptimal management: a nationwide population study. Ann Rheum Dis. 2015;74:661–7.
Smolen LJ, Gahn JC, Mitri G, et al. the budget impact of increased use of febuxostat in the management of gout: a US health plan managed care pharmacy and medical costs perspective. Clin Ther. 2016;38:1710–25.
Hu M, Tomlinson B. Febuxostat in the management of hyperuricemia and chronic gout: a review. Ther Clin Risk Manag. 2008;4:1209–20.
Khanna D, Fitzgerald JD, Khanna PP, et al. 2012 American College of Rheumatology guidelines for management of gout. Part 1: systematic nonpharmacologic and pharmacologic therapeutic approaches to hyperuricemia. Arthritis Care Res (Hoboken). 2012;64:1431–46.
White WB, Saag KG, Becker MA, et al. Cardiovascular safety of febuxostat or allopurinol in patients with gout. N Engl J Med. 2018;378:1200–10.
Ayta IA, McKinlay JB, Krane RJ. The likely worldwide increase in erectile dysfunction between 1995 and 2025 and some possible policy consequences. BJU Int. 1999;84(1):50–6.
Schlesinger N, Radvanski DC, Cheng JQ, et al. Erectile dysfunction is common among patients with gout. J Rheumatol. 2015;42:1893–7.
Luo L, Xiang Q, Deng Y, et al. Gout is associated with elevated risk of erectile dysfunction: a systematic review and meta-analysis. Rheumatol Int. 2019;39:1527–35.
Sansone A, Reisman Y, Jannini EA. Relationship between hyperuricemia with deposition and sexual dysfunction in males and females. J Endocrinol Invest. 2022;45:691–703.
Shamloul R, Ghanem H. Erectile dysfunction. Lancet. 2013;381:153–65.
Gagliardi AC, Miname MH, Santos RD. Uric acid: a marker of increased cardiovascular risk. Atherosclerosis. 2009;202:11–7.
Puddu P, Puddu GM, Cravero E, et al. Relationships among hyperuricemia, endothelial dysfunction and cardiovascular disease: molecular mechanisms and clinical implications. J Cardiol. 2012;59:235–42.
Maruhashi T, Hisatome I, Kihara Y, et al. Hyperuricemia and endothelial function: from molecular background to clinical perspectives. Atherosclerosis. 2018;278:226–31.
Schlesinger N, Lu N, Choi HK. Gout and the risk of incident erectile dysfunction: a body mass index-matched population-based study. J Rheumatol. 2018;45:1192–7.
Chen YF, Lin HH, Lu CC, et al. Gout and a subsequent increased risk of erectile dysfunction in men aged 64 and under: a nationwide cohort study in Taiwan. J Rheumatol. 2015;42:1898–905.
Nishino T, Okamoto K, Eger BT, et al. Mammalian xanthine oxidoreductase—mechanism of transition from xanthine dehydrogenase to xanthine oxidase. FEBS J. 2008;275(13):3278–89.
Chung HY, Baek BS, Song SH, et al. Xanthine dehydrogenase/xanthine oxidase and oxidative stress. Age (Omaha). 1997;20(3):127–40.
Jeremy JY, Jones RA, Koupparis AJ, et al. Reactive oxygen species and erectile dysfunction: possible role of NADPH oxidase. Int J Impot Res. 2007;19:265–80.
Tanaka A, Taguchi I, Teragawa H, et al. Febuxostat does not delay progression of carotid atherosclerosis in patients with asymptomatic hyperuricemia: a randomized, controlled trial. PLoS Med. 2020;17: e1003095.
FitzGerald JD, Dalbeth N, Mikuls T, et al. 2020 American College of Rheumatology guideline for the management of gout. Arthritis Rheumatol. 2020;72(6):879–95.
Hui M, Carr A, Cameron S, et al. The British Society for Rheumatology guideline for the management of gout. Rheumatology (Oxford). 2017;56(7):1056–9.
Author information
Authors and Affiliations
Contributions
DR SMD and Dr CCW had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Study conception and design: Dr SMD. Acquisition, analysis, or interpretation of data: All authors. Drafting the manuscript: Dr QT. Editing the manuscript: Dr SMD. Statistical analysis: Dr SIW and Dr QT.
Corresponding authors
Ethics declarations
Funding
This article was supported by funding from NSFC 81900795.
Conflict of interest
The authors declare that there are no competing interests.
Ethics approval
The TriNetX platform is compliant with the Health Insurance Portability and Accountability Act (HIPAA) and the General Data Protection Regulation (GDPR). The Western Institutional Review Board (WIRB) has granted a waiver to TriNetX because it contains only aggregated counts and statistical summaries of anonymized information. The use of TriNetX for the present study was approved by the Institutional Review Board of Chung Shan Medical University Hospital (CSMUH No: CS2-21176).
Consent to participate
Not applicable.
Consent for publication
All the listed authors have read and approved the final manuscript and agreed to publication.
Availability of data and materials (data sharing statement)
All data relevant to the study are included in the article.
Code availability
https://live.trinetx.com/tnx/study/111600/analytics/632a78c05745ce4a5743338a/outcomes/results.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tong, Q., Du, Y., Cui, R. et al. Risk of Erectile Dysfunction in Male Patients with Gout Treated with Febuxostat or Allopurinol: A Propensity Score-Matched Cohort Study. Drugs 82, 1717–1726 (2022). https://doi.org/10.1007/s40265-022-01816-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-022-01816-x