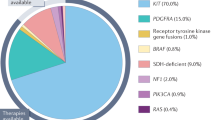
Abstract
Ripretinib (Qinlock®) is a small molecule inhibitor of the receptor tyrosine kinases KIT and platelet-derived growth factor receptor α (PDGFRA) and is approved for the treatment of gastrointestinal stromal tumours as a fourth-line of therapy. After successive cycles of treatment, gastrointestinal stromal tumours can carry a wide array of mutations, which makes resistance to treatment more likely. Ripretinib has a dual mechanism of action that allows it to target a broad spectrum of mutations in KIT or PDGFRA. The pivotal phase III INVICTUS trial demonstrated an increase of progression-free survival in patients receiving ripretinib compared with placebo, with efficacy being maintained across patients with KIT exon 9, 11, 13 and 17 mutations. Ripretinib has acceptable tolerability, with the most common drug-related grade 3 or 4 adverse events being lipase increases, hypertension, fatigue and hypophosphataemia. Ripretinib is therefore a valuable additional line of therapy available for the management of gastrointestinal stromal tumours.
Plain Language Summary
The receptor tyrosine kinases KIT and platelet-derived growth factor receptor α (PDGFRA) regulate cell growth and survival; mutations in the genes of these proteins are the most common causes of gastrointestinal stromal tumours. Drugs that target kinases are a mainstay in the treatment of these cancers; however, new mutations often occur that make tumours resistant to kinase inhibitors. Ripretinib (Qinlock®) is approved for patients with gastrointestinal stromal tumours after the tumour has become resistant to three or more other kinase inhibitors. In a pivotal phase III clinical trial in patients with gastrointestinal stromal tumours and who had failure with three or more prior treatments, ripretinib significantly delayed disease progression compared with placebo. Ripretinib has an acceptable side effect profile, with the most common drug-related side effects being alopecia, muscle pain and nausea. Thus, ripretinib is a valuable treatment option in the management of advanced gastrointestinal stromal tumours.

Similar content being viewed by others
References
Klug LR, Khosroyani HM, Kent JD, et al. New treatment strategies for advanced-stage gastrointestinal stromal tumours. Nat Rev Clin Oncol. 2022;19(5):328–41.
Patel SR, Reichardt P. An updated review of the treatment landscape for advanced gastrointestinal stromal tumors. Cancer. 2021;127(13):2187–95.
Verschoor AJ, Bovee J, Overbeek LIH, et al. The incidence, mutational status, risk classification and referral pattern of gastro-intestinal stromal tumours in the Netherlands: a nationwide pathology registry (PALGA) study. Virchows Arch. 2018;472(2):221–9.
Corless CL, Schroeder A, Griffith D, et al. PDGFRA mutations in gastrointestinal stromal tumors: frequency, spectrum and in vitro sensitivity to imatinib. J Clin Oncol. 2005;23(23):5357–64.
Manley PW, Cowan-Jacob SW, Buchdunger E, et al. Imatinib: a selective tyrosine kinase inhibitor. Eur J Cancer. 2002;38(Suppl 5):S19-27.
Heinrich MC, Corless CL, Blanke CD, et al. Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J Clin Oncol. 2006;24(29):4764–74.
US FDA. Qinlock® (ripretinib) tablets: US prescribing information. 2021. https://www.qinlockhcp.com/. Accessed 28 Sep 2022.
European Medicines Agency. QINLOCK 50 mg tablets: summary of product characteristics. 2021. https://www.ema.europa.eu/. Accessed 28 Sep 2022.
Smith BD, Kaufman MD, Lu WP, et al. Ripretinib (DCC-2618) is a switch control kinase inhibitor of a broad spectrum of oncogenic and drug-resistant KIT and PDGFRA variants. Cancer Cell. 2019;35(5):738-51.e9.
Janku F, Abdul Razak AR, Chi P, et al. Switch control inhibition of KIT and PDGFRA in patients with advanced gastrointestinal stromal tumor: a phase I study of ripretinib. J Clin Oncol. 2020;38(28):3294–303.
Li X, Shelton MJ, Wang J, et al. Effects of CYP3A inhibition, CYP3A induction, and gastric acid reduction on the pharmacokinetics of ripretinib, a switch control KIT tyrosine kinase inhibitor. Clin Pharmacol Drug Dev. 2022. https://doi.org/10.1002/cpdd.1110.
Blay J-Y, Serrano C, Heinrich MC, et al. Ripretinib in patients with advanced gastrointestinal stromal tumours (INVICTUS): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2020;21(7):923–34.
Bauer S, Heinrich MC, George S, et al. Clinical activity of ripretinib in patients with advanced gastrointestinal stromal tumor harboring heterogeneous KIT/PDGFRA mutations in the phase III INVICTUS study. Clin Cancer Res. 2021;27(23):6333–42.
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–47.
Demetri GD, Reichardt P, Kang Y-K, et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet. 2013;381(9863):295–302.
von Mehren M, Heinrich MC, George S, et al. Ripretinib as ≥ 4th-line treatment in patients with advanced gastrointestinal stromal tumor: long-term update from the phase III INVICTUS study [abstract no. 1540P and poster]. Ann Oncol. 2021;32(Suppl 5):S1120–1.
Zalcberg JR, Heinrich MC, George S, et al. Clinical benefit of ripretinib dose escalation after disease progression in advanced gastrointestinal stromal tumor: an analysis of the INVICTUS study. Oncologist. 2021;26(11):e2053–60.
Bauer S, Jones RL, Blay JY, et al. Ripretinib versus sunitinib in patients with advanced gastrointestinal stromal tumor after treatment with imatinib (INTRIGUE): a randomized, open-label, phase III trial. J Clin Oncol. 2022;2022:JCO2200294.
Robert C, Soria JC, Spatz A, et al. Cutaneous side-effects of kinase inhibitors and blocking antibodies. Lancet Oncol. 2005;6(7):491–500.
Patel A, George S, Janku F, et al. Dermatopathological review of cutaneous squamous cell carcinoma events in patients with gastrointestinal stromal tumors treated with ripretinib [abstract no. P-107 and poster]. Ann Oncol. 2021;32(Suppl 3):S134–5.
Chen MH, Kerkela R, Force T. Mechanisms of cardiac dysfunction associated with tyrosine kinase inhibitor cancer therapeutics. Circulation. 2008;118(1):84–95.
Li X, Psoinos CM, Shelton MJ, et al. Effect of hepatic impairment on the pharmacokinetics of ripretinib [abstract no. e16031]. J Clin Oncol. 2022;40(16 Suppl):e16031.
Casali PG, Blay JY, Abecassis N, et al. Gastrointestinal stromal tumours: ESMO–EURACAN–GENTURIS clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2022;33(1):20–33.
National Comprehensive Cancer Network. Gastroinestinal stromal tumors (GISTs): NCCN guidelines version 2.2022. 2022. https://www.nccn.org/. Accessed 28 Sep 2022.
Taiho Pharmaceutical Co. Taiho pharmaceutical obtains approval to manufacture and market HSP90 inhibitor Jeselhy® tablets 40 mg (pimitespib) for gastrointestinal stromal tumor (GIST) [media release]. 20 June 2022. https://www.taiho.co.jp/.
Acknowledgments
During the peer review process, the manufacturer of ripretinib was also offered an opportunity to review this article. Changes resulting from comments received were made on the basis of scientific and editorial merit.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The preparation of this review was not supported by any external funding.
Authorship and conflict of interest
S. Fung and M. Shirley are salaried employees of Adis International Ltd/Springer Nature, and declare no relevant conflicts of interest. All authors contributed to the review and are responsible for the article content.
Ethics approval, Consent to participate, Consent to publish, Availability of data and material, Code availability
Not applicable.
Additional information
The manuscript was reviewed by: Y. Hayashi, Enteric Neuroscience Program and Department of Physiology and Biomedical Engineering, Mayo Clinic College of Medicine and Science, Rochester, Minnesota, USA; C.-N. Yeh, General Surgery Department and Liver Research Center, Chang Gung Memorial Hospital, Chang Gung University.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fung, S., Shirley, M. Ripretinib: A Review in Gastrointestinal Stromal Tumours as Fourth-or Later-Line of Therapy. Drugs 82, 1541–1548 (2022). https://doi.org/10.1007/s40265-022-01794-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-022-01794-0