Abstract
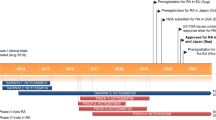
Baricitinib (Olumiant™) is an orally-administered, small-molecule, janus-associated kinase (JAK) inhibitor developed by Eli Lilly and Incyte Corporation for the treatment of rheumatoid arthritis (RA), atopic dermatitis and systemic lupus erythematosus. JAKs transduce intracellular signals from cell surface receptors for various cytokines and growth factors involved in inflammation and immune function, suggesting JAK inhibitors may be of therapeutic benefit in inflammatory conditions. In February 2017, baricitinib was approved in the EU, as monotherapy or in combination with methotrexate, for the treatment of moderate to severe active rheumatoid arthritis in adult patients who have responded inadequately to, or who are intolerant to one or more disease-modifying anti-rheumatic drugs (DMARDs). Regulatory approval to market baricitinib as a treatment for RA has also been sought in the USA and Japan. This article summarizes the milestones in the development of baricitinib leading to this first global approval for the treatment for moderate to severe active RA in adult patients who have responded inadequately to, or are intolerant to one or more DMARDs.
Similar content being viewed by others
References
Eli Lilly and Company, Incyte Corporation. European Commission approves once-daily olumiant tablets for treatment of adults with moderate-to-severe active rheumatoid arthritis [media release]. 13 Feb 2017.
Eli Lilly and Company, Incyte Corporation. US FDA extends review period for baricitinib, an investigational rheumatoid arthritis treatment [media release]. 13 Jan 2017.
Eli Lilly and Company, Incyte Corporation. Lilly and Incyte announce collaboration for development and commercialization of oral anti-inflammatory and autoimmune therapies [media release]. 21 Dec 2009.
Fridman JS, Scherle PA, Collins R, et al. Selective inhibition of JAK1 and JAK2 is efficacious in rodent models of arthritis: preclinical characterization of INCB028050. J Immunol. 2010;184(9):5298–307.
Shi JG, Chen X, Lee F, et al. The pharmacokinetics, pharmacodynamics, and safety of baricitinib, an oral JAK 1/2 inhibitor, in healthy volunteers. J Clin Pharmacol. 2014;54(12):1354–61.
European Medicines Agency. Baricitinib (Olumiant): summary of product characteristics 2017. http://ec.europa.eu/health/documents/community-register/2017/20170213136870/anx_136870_en.pdf. Accessed 17 Feb 2017.
Payne C, Zhang X, Shahri N, et al. Evaluation of potential drug-drug interactions with baricitinib [abstract no. AB0492]. Ann Rheum Dis. 2015;74(Suppl 2):1063.
Fleischmann R, Schiff M, van der Heijde D, et al. Baricitinib, methotrexate, or combination in patients with rheumatoid arthritis and no or limited prior disease-modifying antirheumatic drug treatment. Arthritis Rheumatol. 2017;69(3):506–17.
Dougados M, van der Heijde D, Chen YC, et al. Baricitinib in patients with inadequate response or intolerance to conventional synthetic DMARDs: results from the RA-BUILD study. Ann Rheum Dis. 2017;76(1):88–95.
Taylor PC, Keystone EC, van der Heijde D, et al. Baricitinib versus placebo or adalimumab in rheumatoid arthritis. N Engl J Med. 2017;376(7):652–62.
Genovese MC, Kremer J, Zamani O, et al. Baricitinib in patients with refractory rheumatoid arthritis. N Engl J Med. 2016;374(13):1243–52.
Takeuchi T, Genovese M, Xie L, et al. Baricitinib dose step-down following disease control in patients with rheumatoid arthritis [abstract no. OP0228]. Ann Rheum Dis. 2016;75(Suppl 2):144.
Keystone EC, Taylor PC, Drescher E, et al. Safety and efficacy of baricitinib at 24 weeks in patients with rheumatoid arthritis who have had an inadequate response to methotrexate. Ann Rheum Dis. 2015;74(2):333–40.
Keystone E, Taylor P, Genovese M, et al. Safety and efficacy of baricitinib through 128 weeks in an open-label, long-term extension study in patients with rheumatoid arthritis [abstract no. 67]. J Rheumatol. 2015;42(7):1292.
Tanaka Y, Emoto K, Cai Z, et al. Efficacy and safety of baricitinib in Japanese patients with active rheumatoid arthritis receiving background methotrexate therapy: a 12-week, double-blind, randomized placebo-controlled study. J Rheumatol. 2016;43(3):504–11.
Tanaka Y, Emoto K, Cai Z, et al. Efficacy and safety of baricitinib in Japanese rheumatoid arthritis patients during a 52 week extension phase [abstract no. 1483]. Arthritis. Rheumatol. 2014;66(Suppl 10):S652.
Papp KA, Menter MA, Raman M, et al. A randomized phase 2b trial of baricitinib, an oral Janus kinase (JAK) 1/JAK2 inhibitor, in patients with moderate-to-severe psoriasis. Br J Dermatol. 2016;174(6):1266–76.
Papp K, Menter A, Raman M, et al. A Phase 2b dose-ranging trial of baricitinib, an oral JAK 1/JAK 2 inhibitor, in patients with moderate-to-severe psoriasis: Results from the randomized withdrawal and re-treatment periods [abstract no. P032]. J Invest Dermatol. 2015;135(Suppl 3):S7.
Tuttle KR, Brosius III FC, Adler SG, et al. Baricitinib in diabetic kidney disease: results from a phase 2, multicenter, randomized, double-blind, placebo-controlled study [abstract no. 114-LB]. Diabetes. 2015;64(Suppl 1):LB29.
Kremer J, Genovese MC, Keystone E, et al. Effects of baricitinib on lipid, apolipoprotein, and lipoprotein particle profiles in a phase 2b study in patients with active rheumatoid arthritis. Arthritis Rheumatol. 2016. doi:10.1002/art.40036.
Smolen J, Genovese M, Takeuchi T, et al. Safety profile of baricitinib in patients with active RA: an integrated analysis [abstract no. THU0166]. Ann Rheum Dis. 2016;75(Suppl 2):243–4.
Disclosure
The preparation of this review was not supported by any external funding. During the peer review process the manufacturer of the agent under review was offered an opportunity to comment on the article. Changes resulting from any comments received were made by the author on the basis of scientific completeness and accuracy. A. Markham is a contracted employee of Adis, Springer SBM.
Author information
Authors and Affiliations
Corresponding author
Additional information
This profile has been extracted and modified from the AdisInsight database. AdisInsight tracks drug development worldwide through the entire development process, from discovery, through pre-clinical and clinical studies to market launch and beyond.
Rights and permissions
About this article
Cite this article
Markham, A. Baricitinib: First Global Approval. Drugs 77, 697–704 (2017). https://doi.org/10.1007/s40265-017-0723-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-017-0723-3