Abstract
Introduction
Fluoroquinolones are widely used to treat bacterial infections. Many in vitro and in vivo studies have established a chemical relationship between fluoroquinolones’ particular chemical structure and photosensitivity. The aim of this study was to establish a relationship between the chemical structure of fluoroquinolones and the risk of photosensitivity adverse effects from real-world data.
Methods
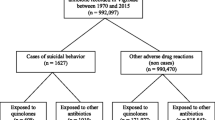
All the Individual Case Safety Reports (ICSRs) related to fluoroquinolones and registered in the World Health Organization global database (VigiBase®) up to December 31, 2017 were collected. A disproportionality analysis was performed in order to quantify the photosensitivity risk for each fluoroquinolone by calculating their reporting odds ratio (ROR).
Results
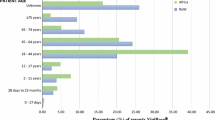
Up to December 31, 2017, 282,805 ICSRs related to fluoroquinolones were selected, of which 1647 were photosensitivity adverse event cases. Sparfloxacin had the highest adjusted ROR of 161.10 (95% confidence interval [CI] 133.66–194.02) followed by grepafloxacin (40.30 [26.30–59.60]) closely followed by lomefloxacin (32.61 [28.61–37.07]), then enoxacin (11.04 [8.33–14.32]) and fleroxacin (8.22 [5.06–12.56]).
Conclusion
This study confirms the high reporting rate of photosensitivity adverse effects for sparfloxacin from real-world data. Moreover, our data suggest more photosensitivity adverse effects reporting for fluoroquinolones with a halogen at their 8th position.


Similar content being viewed by others
References
Domagala JM. Structure-activity and structure-side-effect relationships for the quinolone antibacterials. J Antimicrob Chemother. 1994;33:685–706.
Sousa J, Alves G, Fortuna A, Falcão A. Third and fourth generation fluoroquinolone antibacterials: a systematic review of safety and toxicity profiles. Curr Drug Saf. 2014;9:89–105.
Effect of fluoroquinolones on melanogenesis in normal human melanocytes HEMn-DP: a comparative in vitro study. PubMed—NCBI. https://www.ncbi.nlm.nih.gov/pubmed/27572617. Accessed 12 Jan 2020.
Hayashi N, Nakata Y, Yazaki A. New findings on the structure–phototoxicity relationship and photostability of fluoroquinolones with various substituents at position 1. Antimicrob Agents Chemother. 2004;48:799–803.
Yabe K, Goto K, Jindo T, Sekiguchi M, Furuhama K. Structure–phototoxicity relationship in Balb/c mice treated with fluoroquinolone derivatives, followed by ultraviolet-A irradiation. Toxicol Lett. 2005;157:203–10.
Seto Y, Inoue R, Ochi M, Gandy G, Yamada S, Onoue S. Combined use of in vitro phototoxic assessments and cassette dosing pharmacokinetic study for phototoxicity characterization of fluoroquinolones. AAPS J. 2011;13:482–92.
Zhao J, Liu Y, Jiang X, Guo P, Xu Y, Zhang P, et al. Effect of C-5 position on the photochemical properties and phototoxicity of antofloxacin and levofloxacin: a stable and transient study. J Photochem Photobiol B. 2016;155:122–9.
UMC|VigiBase. https://www.who-umc.org/vigibase/vigibase/. Accessed 15 Oct 2018.
Montastruc J-L, Sommet A, Bagheri H, Lapeyre-Mestre M. Benefits and strengths of the disproportionality analysis for identification of adverse drug reactions in a pharmacovigilance database. Br J Clin Pharmacol. 2011;72:905–8.
Faillie J-L. Case–non case studies: principles, methods, bias and interpretation. Therapie. 2018;73:247–55.
UMC|vigiMethods. https://www.who-umc.org/vigibase/vigilyze/vigimethods/. Accessed 31 Oct 2018.
Hamanaka H, Mizutani H, Asahig K, Shimizu M. Melanocyte melanin augments sparfloxacin-induced phototoxicity. J Dermatol Sci. 1999;21:27–33.
Tokura Y, Iwamoto Y, Mizutani K, Takigawa M. Sparfloxacin phototoxicity: potential photoaugmentation by ultraviolet A and B sources. Arch Dermatol Res. 1996;288:45–50.
Stricker BH. Adverse reaction signal detection methodology in pharmacoepidemiology. Eur J Epidemiol. 2018;33:507–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Yoann Zelmat, Vanessa Rousseau, Leila Chebane, Jean-Louis Montastruc, Haleh Bagheri, and Agnès Sommet have no conflicts of interest that are directly relevant to the content of this study.
Funding
No sources of funding were used to assist in the preparation of this study.
Data sharing
The data sharing policy type is 2.
Rights and permissions
About this article
Cite this article
Zelmat, Y., Rousseau, V., Chebane, L. et al. Fluoroquinolone-Induced Photosensitivity: A Chemical Fragment-Based Approach by a Case/Non-case Study in VigiBase®. Drug Saf 43, 561–566 (2020). https://doi.org/10.1007/s40264-020-00917-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-020-00917-4