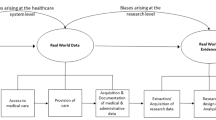
Abstract
Research that makes secondary use of administrative and clinical healthcare databases is increasingly influential for regulatory, reimbursement, and other healthcare decision-making. Consequently, there are numerous guidance documents on reporting for studies that use ‘real-world’ data captured in administrative claims and electronic health record (EHR) databases. These guidance documents are intended to improve transparency, reproducibility, and the ability to evaluate validity and relevance of design and analysis decisions. However, existing guidance does not differentiate between structured and unstructured information contained in EHRs, registries, or other healthcare data sources. While unstructured text is convenient and readily interpretable in clinical practice, it can be difficult to use for investigation of causal questions, e.g., comparative effectiveness and safety, until data have been cleaned and algorithms applied to extract relevant information to structured fields for analysis. The goal of this paper is to increase transparency for healthcare decision makers and causal inference researchers by providing general recommendations for reporting on steps taken to make unstructured text-based data usable for comparative effectiveness and safety research. These recommendations are designed to be used as an adjunct for existing reporting guidance. They are intended to provide sufficient context and supporting information for causal inference studies involving use of natural language processing- or machine learning-derived data fields, so that researchers, reviewers, and decision makers can be confident in their ability to evaluate the validity and relevance of derived measures for exposures, inclusion/exclusion criteria, covariates, and outcomes for the causal question of interest.
Similar content being viewed by others
References
National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Health Sciences Policy; Forum on Drug Discovery, Development, and Translation. Real-world evidence generation and evaluation of therapeutics: proceedings of a workshop. Washington, DC: National Academies Press (US). 2017.
Psaty BM, Breckenridge AM. Mini-Sentinel and regulatory science—big data rendered fit and functional. N Engl J Med. 2014;370(23):2165–7.
Fleurence RL, Curtis LH, Califf RM, Platt R, Selby JV, Brown JS. Launching PCORnet, a national patient-centered clinical research network. J Am Med Inform Assoc. 2014;21(4):578–82.
Oliveira JL, Lopes P, Nunes T, Campos D, Boyer S, Ahlberg E, et al. The EU-ADR web platform: delivering advanced pharmacovigilance tools. Pharmacoepidemiol Drug Saf. 2013;22(5):459–67.
Collaborators A, Andersen M, Bergman U, Choi N-K, Gerhard T, Huang C, et al. The Asian Pharmacoepidemiology Network (AsPEN): promoting multi-national collaboration for pharmacoepidemiologic research in Asia. Pharmacoepidemiol Drug Saf. 2013;22(7):700–4.
Behrman RE, Benner JS, Brown JS, McClellan M, Woodcock J, Platt R. Developing the Sentinel system—a national resource for evidence development. N Engl J Med. 2011;364(6):498–9.
Suissa S, Henry D, Caetano P, Dormuth CR, Ernst P, Hemmelgarn B, et al. CNODES: the Canadian network for observational drug effect studies. Open Med. 2012;6(4):e134–40.
Trifiro G, Coloma PM, Rijnbeek PR, Romio S, Mosseveld B, Weibel D, et al. Combining multiple healthcare databases for postmarketing drug and vaccine safety surveillance: why and how? J Intern Med. 2014;275(6):551–61.
Engel P, Almas MF, De Bruin ML, Starzyk K, Blackburn S, Dreyer NA. Lessons learned on the design and the conduct of post-authorization safety studies: review of 3 years of PRAC oversight. Br J Clin Pharmacol. 2017;83(4):884–93.
Eichler H-G, Hurts H, Broich K, Rasi G. Drug regulation and pricing—can regulators influence affordability? N Engl J Med. 2016;374(19):1807–9.
Makady A, Ham RT, de Boer A, Hillege H, Klungel O, Goettsch W, et al. Policies for use of real-world data in health technology assessment (HTA): a comparative study of six HTA agencies. Value Health. 2017;20(4):520–32.
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. PLoS Med. 2007;4(10):e296.
Vandenbroucke JP, von Elm E, Altman DG, Gøtzsche PC, Mulrow CD, Pocock SJ, et al. Strengthening the reporting of observational studies in epidemiology (STROBE): explanation and elaboration. PLoS Med. 2007;4(10):e297.
Wang SV, Schneeweiss S, Berger ML, Brown J, de Vries F, Douglas I, et al. Reporting to improve reproducibility and facilitate validity assessment for healthcare database studies V1.0. Pharmacoepidemiol Drug Saf. 2017;26(9):1018–32.
Benchimol EI, Smeeth L, Guttmann A, Harron K, Moher D, Petersen I, et al. The REporting of studies conducted using observational routinely-collected health Data (RECORD) statement. PLoS Med. 2015;12(10):e1001885.
EMA. ENCePP guide on methodological standards in pharmacoepidemiology. London: EMA; 2014.
Us FDA. Guidance for industry and FDA staff: best practices for conducting and reporting pharmacoepidemiologic safety studies using electronic healthcare data. Rockville: US FDA; 2013.
Collins GS, Reitsma JB, Altman DG, Moons KGM. Transparent Reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. BMJ. 2015;350:g7594.
Wong A, Plasek JM, Montecalvo SP, Zhou L. Natural language processing and its implications for the future of medication safety: a narrative review of recent advances and challenges. Pharmacotherapy. 2018;38(8):822–41.
Ford E, Carroll JA, Smith HE, Scott D, Cassell JA. Extracting information from the text of electronic medical records to improve case detection: a systematic review. J Am Med Inform Assoc. 2016;23(5):1007–15.
Weiss LS, Zhou X, Walker AM, Ananthakrishnan AN, Shen R, Sobel RE, et al. A case study of the incremental utility for disease identification of natural language processing in electronic medical records. Pharm Med. 2018;32(1):31–7.
Walker AM, Zhou X, Ananthakrishnan AN, Weiss LS, Shen R, Sobel RE, et al. Computer-assisted expert case definition in electronic health records. Int J Med Inform. 2016;86:62–70.
Khalifa A, Meystre S. Adapting existing natural language processing resources for cardiovascular risk factors identification in clinical notes. J Biomed Inform. 2015;58:S128–32.
Li Q, Spooner SA, Kaiser M, Lingren N, Robbins J, Lingren T, et al. An end-to-end hybrid algorithm for automated medication discrepancy detection. BMC Med Inform Decis Mak. 2015;15:37.
White RW, Wang S, Pant A, Harpaz R, Shukla P, Sun W, et al. Early identification of adverse drug reactions from search log data. J Biomed Inform. 2016;59:42–8.
Han L, Ball R, Pamer CA, Altman RB, Proestel S. Development of an automated assessment tool for MedWatch reports in the FDA adverse event reporting system. J Am Med Inform Assoc. 2017;24(5):913–20.
Sarker A, Gonzalez G. Portable automatic text classification for adverse drug reaction detection via multi-corpus training. J Biomed Inform. 2015;53:196–207.
Strandell J, Caster O, Bate A, Norén N, Edwards IR. Reporting patterns indicative of adverse drug interactions. Drug Saf. 2011;34(3):253–66.
Botsis T, Buttolph T, Nguyen MD, Winiecki S, Woo EJ, Ball R. Vaccine adverse event text mining system for extracting features from vaccine safety reports. J Am Med Inform Assoc. 2012;19(6):1011–8.
Wunnava S, Qin X, Kakar T, Kong X, Rundensteiner EA, Sahoo SK, et al. One size does not fit all: an ensemble approach towards information extraction from adverse drug event narratives. In: Proceedings of the 11th international joint conference on biomedical engineering systems and technologies, vol 5. HEALTHINF. 2018. p. 176–188
Chen ES, Hripcsak G, Xu H, Markatou M, Friedman C. Automated acquisition of disease—drug knowledge from biomedical and clinical documents: an initial study. J Am Med Inform Assoc. 2008;15(1):87–98.
Califf RM. The patient-centered outcomes research network: a national infrastructure for comparative effectiveness research. N C Med J. 2014;75(3):204–10.
Ball R, Robb M, Anderson SA, Dal Pan G. The FDA’s sentinel initiative—a comprehensive approach to medical product surveillance. Clin Pharmacol Ther. 2016;99(3):265–8.
US FDA. Safety: FDA’s sentinel initiative. http://www.fda.gov/Safety/FDAsSentinelInitiative/ucm2007250.htm. Accessed Jan 2016.
Duke-Margolis Center for Health Policy. Discussion guide. Improving the efficiency of outcome validation in the Sentinel System. Washington, DC: Duke-Margolis Center for Health Policy. 2018.
PCORNet. PTNP-CCR. PCORnet common data model. 2016. http://www.pcornet.org/pcornet-common-data-model/. Accessed Jan 2016.
Brown JB, N; Curtis, L; Raebel, MA; Haynes, K, Rosofsky, R. Sentinel common data model. 2017. https://www.sentinelinitiative.org/sentinel/data/distributed-database-common-data-model/sentinel-common-data-model. Accessed 14 June 2018.
Ball R, Toh S, Nolan J, Haynes K, Forshee R, Botsis T. Evaluating automated approaches to anaphylaxis case classification using unstructured data from the FDA sentinel system. Pharmacoepidemiol Drug Saf. 2018;27(10):1077–84.
Seninel. Surveillance tools. Health outcome of interest validations and literature reviews. https://www.sentinelinitiative.org/sentinel/surveillance-tools/validations-lit-review. Accessed 11 Dec 2018.
Huerta C, Abbing-Karahagopian V, Requena G, Oliva B, Alvarez Y, Gardarsdottir H, et al. Exposure to benzodiazepines (anxiolytics, hypnotics and related drugs) in seven European electronic healthcare databases: a cross-national descriptive study from the PROTECT-EU Project. Pharmacoepidemiol Drug Saf. 2016;25(Suppl. 1):56–65.
Lai ECC, Stang P, Yang YHK, Kubota K, Wong ICK, Setoguchi S. International multi-database pharmacoepidemiology: potentials and pitfalls. Curr Epidemiol Rep. 2015;2(4):229–38.
Pratt N, Andersen M, Bergman U, Choi N-K, Gerhard T, Huang C, et al. Multi-country rapid adverse drug event assessment: the Asian Pharmacoepidemiology Network (AsPEN) antipsychotic and acute hyperglycaemia study. Pharmacoepidemiol Drug Saf. 2013;22(9):915–24.
Wang S, Verpillat P, Rassen J, Patrick A, Garry E, Bartels D. Transparency and reproducibility of observational cohort studies using large healthcare databases. Clin Pharmacol Ther. 2016;99(3):325–32.
Schneeweiss S, Rassen JA, Brown JS, Rothman KJ, Happe L, Arlett P, et al. Graphical depiction of longitudinal study designs in health care databases. Ann Intern Med. 2019;170(6):398–406.
Datta-Nemdharry P, Thomson A, Beynon J. Opportunities and challenges in developing a cohort of patients with type 2 diabetes mellitus using electronic primary care data. PloS One. 2016;11(11):e0162236.
Reeves D, Springate DA, Ashcroft DM, Ryan R, Doran T, Morris R, et al. Can analyses of electronic patient records be independently and externally validated? The effect of statins on the mortality of patients with ischaemic heart disease: a cohort study with nested case–control analysis. BMJ Open. 2014;4(4):e004952.
Shiloach M, Frencher SK, Steeger JE, Rowell KS, Bartzokis K, Tomeh MG, et al. Toward robust information: data quality and inter-rater reliability in the American College of Surgeons National Surgical Quality Improvement Program. J Am Coll Surg. 2010;210(1):6–16.
Hallgren KA. Computing inter-rater reliability for observational data: an overview and tutorial. Tutor Quant Methods Psychol. 2012;8(1):23–34.
Rothman KJ, Greenland S, Lash TL. Modern epidemiology. Philadelphia: Lippincott, Williams & Wilkins; 2008.
van Zaane B, Vergouwe Y, Donders ART, Moons KGM. Comparison of approaches to estimate confidence intervals of post-test probabilities of diagnostic test results in a nested case–control study. BMC Med Res Methodol. 2012;12:166.
Pencina MJ, D’Agostino RB, Massaro JM. Understanding increments in model performance metrics. Lifetime Data Anal. 2013;19(2):202–18.
Demler OV, Paynter NP, Cook NR. Tests of calibration and goodness-of-fit in the survival setting. Stat Med. 2015;34(10):1659–80.
Berger ML, Sox H, Willke R, Brixner D, Eichler H-G, Goettsch W, et al. Good practices for real-world data studies of treatment and/or comparative effectiveness: Recommendations from the joint ISPOR-ISPE Special Task Force on real-world evidence in health care decision making. Pharmacoepidemiol Drug Saf. 2017;20(8):1003–8
Requena G, Huerta C, Gardarsdottir H, Logie J, González-González R, Abbing-Karahagopian V, et al. Hip/femur fractures associated with the use of benzodiazepines (anxiolytics, hypnotics and related drugs): a methodological approach to assess consistencies across databases from the PROTECT-EU project. Pharmacoepidemiol Drug Saf. 2016;25(Suppl 1):66–78.
Nosek BA, Alter G, Banks GC, Borsboom D, Bowman SD, Breckler SJ, et al. Promoting an open research culture. Science. 2015;348(6242):1422–5.
Sarmiento RF, Dernoncourt F. Improving patient cohort identification using natural language processing. In: MIT Critical Data, editor. Secondary analysis of electronic health records. Springer, Cham. 2016, pp 405–17.
Miller M, Banerjee T, Muppalla R, Romine W, Sheth A. What are people tweeting about zika? An exploratory study concerning its symptoms, treatment, transmission, and prevention. JMIR Public Health Surveill. 2017;3(2):e38.
Toh S, Reichman ME, Houstoun M, Ross Southworth M, Ding X, Hernandez AF, et al. Comparative risk for angioedema associated with the use of drugs that target the renin-angiotensin-aldosterone system. Arch Intern Med. 2012;172(20):1582–9.
Tian Z, Sun S, Eguale T, Rochefort CM. Automated extraction of VTE events from narrative radiology reports in electronic health records: a validation study. Med Care. 2017;55(10):e73–80.
Carroll RJ, Thompson WK, Eyler AE, Mandelin AM, Cai T, Zink RM, et al. Portability of an algorithm to identify rheumatoid arthritis in electronic health records. J Am Med Inform Assoc. 2012;19(e1):e162–9.
Wright A, Pang J, Feblowitz JC, Maloney FL, Wilcox AR, Ramelson HZ, et al. A method and knowledge base for automated inference of patient problems from structured data in an electronic medical record. J Am Med Inform Assoc. 2011;18(6):859–67.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr Shirley V Wang has received salary support on investigator-initiated grants from Novartis Pharmaceuticals Corporation, Boehringer Ingelheim, and J&J to Brigham and Women’s Hospital, and was a consultant to Aetion, Inc., all for unrelated work. Dr Olga V. Patterson receives research grants from the following for-profit organizations: Amgen Inc., Anolinx LLC, AstraZeneca Pharmaceuticals LP, Genentech Inc., Genomic Health, Inc., Gilead Sciences Inc., HITEKS Solutions Inc., Merck & Co., Inc., Northrop Grumman Information Systems, Novartis International AG, PAREXEL International Corporation, and Shire PLC through the University of Utah or Western Institute for Biomedical Research. Dr Patterson also receives research funding from the following federal and non-profit organizations: Agency for Healthcare Research and Quality, Brigham and Women’s Hospital, Centers for Disease Control and Prevention, Department of Defense, Department of Veterans Affairs, Intermountain Healthcare, National Heart, Lung, and Blood Institute, National Institute on Alcohol Abuse and Alcoholism, National Institute of Arthritis and Musculoskeletal and Skin Diseases, National Institute of General Medical Sciences, National Institute of Standards and Technology, National Library of Medicine, National Science Foundation, Patient Centered Outcomes Research Institute, and RAND Corporation. Dr Joshua J. Gagne has received salary support from grants from Novartis Pharmaceuticals Corporation and Eli Lilly and company to Brigham and Women’s Hospital and is a consultant to Aetion, Inc. and to Optum, Inc., all for unrelated work. Dr Andrew Bate is an employee and shareholder of Pfizer. The views expressed in this paper are those of Dr Bate and may not necessarily reflect those of Pfizer. Dr Robert Ball is an author of US Patent 9,075,796, “Text mining for large medical text datasets and corresponding medical text classification using informative feature selection”. Dr Li Zhou has received research funding from the Agency of Healthcare Research and Quality (AHRQ): R01HS022728 and CRICO/RMF. Dr Jeffrey S Brown, Dr Pall Jonsson, Dr Adam Wright, and Dr Wim Goettsch have no conflicts of interest that are directly relevant to the content of this article. The views expressed in this article are the personal views of the authors and may not be understood or quoted as being made on behalf of or reflecting the position of the US Food and Drug Administration or the National Institute for Health and Care Excellence.
Funding
This study was supported by funds from the Division of Pharmacoepidemiology and Pharmacoeconomics and Brigham and Women’s Hospital.
Ethical Approval
Not applicable; no data were analyzed.
Patient Consent
No patient contact or data were involved.
Rights and permissions
About this article
Cite this article
Wang, S.V., Patterson, O.V., Gagne, J.J. et al. Transparent Reporting on Research Using Unstructured Electronic Health Record Data to Generate ‘Real World’ Evidence of Comparative Effectiveness and Safety. Drug Saf 42, 1297–1309 (2019). https://doi.org/10.1007/s40264-019-00851-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-019-00851-0