Abstract
Introduction
Leukotriene-modifying agents (LTMAs) including montelukast, zafirlukast, and zileuton are approved by the US Food and Drug Administration (FDA) for the treatment of asthma and allergic rhinitis. Various neuropsychiatric events (NEs) have been reported; however, the evidence of the association is conflicting. This systematic review investigates the association between NEs and LTMAs by assessing the relevant published literature.
Methods
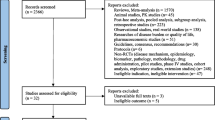
PubMed, EMBASE, MEDLINE, and Cochrane Library were searched using keywords. Studies designed to investigate the association were eligible for inclusion without restriction to any study design or language. The primary outcome was defined as suicidal conditions, while secondary outcomes included all other NEs.
Results
Thirty-three studies were included for a narrative review. Four observational studies did not find a significant association, while ten pharmacovigilance studies using different global databases detected the signals. Notably, some studies suggest that the FDA warning issued in 2008 might have influenced the reporting rate of NEs as a result of increased awareness.
Limitations
The risk of NEs was not quantified, because of the lack of randomized controlled trials and observational studies investigating the association.
Conclusion
Many pharmacovigilance studies have been conducted to determine the association between NEs and LTMAs, but there is limited evidence from observational studies. High-quality epidemiological studies should be conducted to evaluate the association and quantify the risk, not only in children, but also in adults.

Similar content being viewed by others
References
US Food and Drug Administration (FDA). Accolate (zafirlukast) product label. 2013. http://www.fda.gov/Drugs. Accessed 14 Nov 2016.
US Food and Drug Administration (FDA). Zyflo (zileuton) product label. 2012. http://www.fda.gov/Drugs. Accessed 14 Nov 2016.
US Food and Drug Administration (FDA). Singulair (montelukast sodium) product label. 2014. http://www.fda.gov/Drugs. Accessed 14 Nov 2016.
Biswas P, Wilton L, Pearce G, Freemantle S, Sharkir S, Mann RD. Pharmacosurveillance and safety of the leukotriene receptor antagonist (LTRA), montelukast. Clin Exp Allergy Rev. 2001;1(3):300–4.
Twaites BR, Wilton LV, Shakir SAW. Safety of zafirlukast: results of a postmarketing surveillance study on 7976 patients in England. Drug Saf. 2007;30(5):419–29.
Bygdell M, Brunlof G, Wallerstedt SM, Kindblom JM. Psychiatric adverse drug reactions reported during a 10-year period in the Swedish pediatric population. Pharmacoepidemiol Drug Saf. 2012;21(1):79–86.
Wallerstedt SM, Brunlof G, Sundstrom A, Eriksson AL. Montelukast and psychiatric disorders in children. Pharmacoepidemiol Drug Saf. 2009;18(9):858–64.
Aagaard L, Hansen EH. Paediatric adverse drug reactions following use of asthma medications in Europe from 2007 to 2011. Int J Clin Pharm. 2014;36(6):1222–9.
Lafay-Chebassier C, Chavant F, Favreliere S, Pizzoglio V, Perault-Pochat MC. French Association of Regional Pharmacovigilance C. Drug-induced depression: a case/non case study in the French pharmacovigilance database. Therapie. 2015;70(5):425–32.
Aldea-Perona A, Garcia-Saiz M, Sanz Alvarez E. Psychiatric disorders and montelukast in children: a disproportionality analysis of the VigiBase. Drug Saf. 2016;39(1):69–78.
Schumock GT, Gibbons RD, Lee TA, Joo MJ, Stayner LT, Valuck RJ. The association between leukotriene-modifying agents and spontaneously reported suicide. Drug Inf J. 2012;46(1):99–106.
Marchand MS, Jonville-Bera AP, Autret-Leca E. Psychiatric disorders associated with montelukast: data from the National Pharmacovigilance Database. Arch Pediatr. 2013;20(3):269–73.
Cereza G, Dolade NG, Laporte JR. Nightmares induced by montelukast in children and adults. Eur Respir J. 2012;40(6):1574–5.
US Food and Drug Administration (FDA). Updated information on leukotriene inhibitors: montelukast (marketed as Singulair), zafirlukast (marketed as Accolate), and zileuton (marketed as Zyflo and Zyflo CR). 2015. http://www.fda.gov/Drugs. Accessed 23 Dec 2016.
Scott SGM. Singulair, Montelukast: asthma medication linked to serious psychotic episodes in children. ABC News, Australia. 2016. http://www.abc.net.au/news/2016-09-05/asthma-tablet-linked-to-serious-psychotic-episodes-in-children/7795474. Accessed 16 Nov 2016.
Philip G, Hustad CM, Malice MP, Noonan G, Ezekowitz A, Reiss TF, et al. Analysis of behavior-related adverse experiences in clinical trials of montelukast. J Allergy Clin Immunol. 2009;124(4):699–706.
Holbrook JT, Harik-Khan R. Montelukast and emotional well-being as a marker for depression: results from 3 randomized, double-masked clinical trials. J Allergy Clin Immunol. 2008;122(4):828–9.
Philip G, Hustad C, Noonan G, Malice MP, Ezekowitz A, Reiss TF, et al. Reports of suicidality in clinical trials of montelukast. J Allergy Clin Immunol. 2009;124(4):691–6.
Bisgaard H, Skoner D, Boza ML, Tozzi CA, Newcomb K, Reiss TF, et al. Safety and tolerability of montelukast in placebo-controlled pediatric studies and their open-label extensions. Pediatr Pulmonol. 2009;44(6):568–79.
Schumock GT, Lee TA, Joo MJ, Valuck RJ, Stayner LT, Gibbons RD. Association between leukotriene-modifying agents and suicide: what is the evidence? Drug Saf. 2011;34(7):533–44.
Jick H, Hagberg KW, Egger P. Rate of suicide in patients taking montelukast. Pharmacotherapy. 2009;29(2):165–6.
Schumock GT, Stayner LT, Valuck RJ, Joo MJ, Gibbons RD, Lee TA. Risk of suicide attempt in asthmatic children and young adults prescribed leukotriene-modifying agents: a nested case-control study. J Allergy Clin Immunol. 2012;130(2):368–75.
Chen VCH, Wang TN, Liao YT, Lin TC, Stewart R, Lee CTC. Asthma and self-harm: a population-based cohort study in Taiwan. J Psychosom Res. 2014;77(6):462–7.
Ali MM, O’Brien CE, Cleves MA, Martin BC. Exploring the possible association between montelukast and neuropsychiatric events among children with asthma: a matched nested case-control study. Pharmacoepidemiol Drug Saf. 2015;24(4):435–45.
Cheng J. Adverse drug reactions induced by montelukast. Chin J New Drugs. 2014;23(4):486–8 (92).
Lu CY, Zhang F, Lakoma MD, Butler MG, Fung V, Larkin EK, et al. Asthma treatments and mental health visits after a Food and Drug Administration label change for leukotriene inhibitors. Clin Ther. 2015;37(6):1280–91.
Zhou EH, Kaplan S, Mosholder A, Moeny D, Ding Y, Wu Y, et al. Antidepressant medication dispensing among montelukast initiators. J Asthma. 2013;50(9):990–5.
Erdem SB, Nacaroglu HT, Unsal Karkiner CS, Gunay I, Can D. Side effects of leukotriene receptor antagonists in asthmatic children. Iran J Pediatr. 2015. doi:10.5812/ijp.3313.
Anandan N, Ibitoye F. Montelukast and worsening of hallucinations in paranoid schizophrenia. Psychiatr Bull. 2008;32(7):276.
Skillman KL, Stumpf JL. Montelukast-induced anxiety in two pediatric patients. Pharmacotherapy. 2011;31(5):90e–5e.
Byrne F, Oluwole B, Whyte V, Fahy S, McGuinness D. Delayed onset of neuropsychiatric effects associated with montelukast. Ir J Psychol Med. 2012;29(2):125–7.
Callero-Viera A, Infante S, Fuentes-Aparicio V, Zapatero L, Alonso-Lebrero E. Neuropsychiatric reactions to montelukast. J Investig Allergol Clin Immunol. 2012;22(6):452–3.
Kocyigit A, Oksuz BG, Yarar F, Uzun F, Igde M, Islek I. Hallucination development with montelukast in a child with asthma: case presentation. Iran J Allergy Asthma Immunol. 2013;12(4):397–9.
Ibarra-Barrueta O, Palacios-Zabalza I, Mora-Atorrasagasti O, Mayo-Suarez J. Effect of concomitant use of montelukast and efavirenz on neuropsychiatric adverse events. Ann Pharmacother. 2014;48(1):145–8.
Gadde DP, Creticos PS, Beakes DE, Dauby PL, Grooms LA, Abeel D, et al. Adverse effects associated with leukotriene antagonist therapy. J Allergy Clin Immunol. 2010;1:AB68.
Aldea-Perona A, Fernandez-Quintana E, Garcia-Sanchez-Colomer M, Boada-Fernandez del Campo C, Rodriguez-Jimenez C, Sanz-Alvarez E, et al. Up-to-date of the neuropsychiatric events in relation with antileukotriens-agents in the Spanish surveillance system (Sistema Espanol de Farmacovigilancia-SEFV). Basic Clin Pharmacol Toxicol. 2011;109:35.
Iessa N, Star K, Wilton L, Curran S, Edwards IR, Aronson JK, et al. Montelukast and suicide: causality assessment using spontaneous reports and Bradford Hill guidelines. Drug Saf. 2011;34:949–50.
Rhee CW, Choi NK, Ahn YO, Lee J, Park BJ. Risk of depression associated with use of leukotriene receptor antagonist in elderly asthma patients in Korea. Pharmacoepidemiol Drug Saf. 2011;20:S251.
Trotta F, Tartaglia L, Ferrazin F, Santuccio C. Psychiatric and behaviour-related adverse events occurring with antiasthmatic drugs reported in the national pharmacovigilance network. Drug Saf. 2011;34:1013–4.
Iessa N, Murray M, Wong I, Man K, Frank B, Santosh P, et al. Leukotriene receptor antagonists and suicide: a self-controlled case series study. Drug Saf. 2013;36:855.
Burgos Pimentel ABP, De Francisco AM, Tavakov A, Avendano JF, Jimenez TD, Perez TC, et al. Neuropsychiatric disorders associated with montelukast. Allergy. 2014;69:439.
Narang R, Narang S, Narang D, Udeani G. Contemporary use of montelukast and its association with depression in asthma and allergic rhinitis patients. Chest. 2014;146(4):13A.
Erdem SB, Karaman S, Nacaroglu HT, Unsal Karkiner CS, Can D. Are leukotriene receptor antagonists safe? Allergy. 2015;70:412–3.
Benard B, Bastien V, Vinet B, Yang R, Krajinovic M, Ducharme FM. Neuropsychiatric adverse drug reactions in children initiated on montelukast in real-life practice. Eur Respir J. 2017. doi:10.1183/13993003.00148-2017.
Iessa N, Murray ML, Curran S, Wong IC. Asthma and suicide-related adverse events: a review of observational studies. Eur Respir Rev. 2011. doi:10.1183/09059180.00005211.
Trojan TD, Khan DA, Defina LF, Akpotaire O, Goodwin RD, Brown ES. Asthma and depression: the Cooper Center Longitudinal Study. Ann Allergy Asthma Immunol. 2014. doi:10.1016/j.anai.2014.02.015.
Goodwin RD, Robinson M, Sly PD, McKeague IW, Susser ES, Zubrick SR, et al. Severity and persistence of asthma and mental health: a birth cohort study. Psychol Med. 2013. doi:10.1017/s0033291712001754.
Goodwin RD, Scheckner B, Pena L, Feldman JM, Taha F, Lipsitz JD. A 10-year prospective study of respiratory disease and depression and anxiety in adulthood. Ann Allergy Asthma Immunol. 2014. doi:10.1016/j.anai.2014.08.003.
Calapai G, Casciaro M, Miroddi M, Calapai F, Navarra M, Gangemi S. Montelukast-induced adverse drug reactions: a review of case reports in the literature. Pharmacology. 2014;94:60–70.
Zhang CT, Lin JR, Wu F, Ghosh A, Tang SS, Hu M, et al. Montelukast ameliorates streptozotocin-induced cognitive impairment and neurotoxicity in mice. Neurotoxicology. 2016;57:214–22.
Kalra J, Kumar P, Majeed ABA, Prakash A. Modulation of LOX and COX pathways via inhibition of amyloidogenesis contributes to mitoprotection against beta-amyloid oligomer-induced toxicity in an animal model of Alzheimer’s disease in rats. Pharmacol Biochem Behav. 2016;146–147:1–12.
Lai J, Hu M, Wang H, Hu M, Long Y, Miao MX, et al. Montelukast targeting the cysteinyl leukotriene receptor 1 ameliorates Abeta1-42-induced memory impairment and neuroinflammatory and apoptotic responses in mice. Neuropharmacology. 2014;79:707–14.
Di Meco A, Lauretti E, Vagnozzi AN, Pratico D. Zileuton restores memory impairments and reverses amyloid and tau pathology in aged Alzheimer’s disease mice. Neurobiol Aging. 2014;35(11):2458–64.
Ghosh A, Chen F, Thakur A, Hong H. Cysteinyl leukotrienes and their receptors: emerging therapeutic targets in central nervous system disorders. CNS Neurosci Ther. 2016. doi:10.1111/cns.12596.
Acknowledgements
We thank Joseph E. Blais (Centre for Safe Medication Practice and Research) for proofreading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The study was partially supported by a grant from the Research Grants Council of the Hong Kong Special Administrative Region, China (Project No. HKU 789813).
Conflict of interest
Sharon W.Y. Law, Angel Y.S. Wong, Shweta Anand, Ian C.K. Wong, and Esther W. Chan declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Law, S.W.Y., Wong, A.Y.S., Anand, S. et al. Neuropsychiatric Events Associated with Leukotriene-Modifying Agents: A Systematic Review. Drug Saf 41, 253–265 (2018). https://doi.org/10.1007/s40264-017-0607-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-017-0607-1