Abstract
Background and Objectives
Obesity and opioid use for chronic pain in obese individuals are both important public health concerns. The pharmacokinetics of oral morphine after Roux-en-Y gastric bypass (RYGB) are unknown. Therefore, we aimed to study the pharmacokinetics of oral morphine in morbidly obese patients before and after RYGB surgery, to identify the effects of RYGB and the subsequent reversal of morbid obesity on the pharmacokinetic parameters of morphine.
Methods
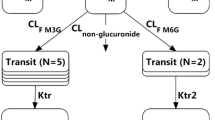
The pharmacokinetics of oral morphine (30 mg) were studied in 30 obese patients before (Visit 1) and then 7–15 days (Visit 2) and 6 months (Visit 3) after RYGB. A population pharmacokinetic model was used to describe the time course of the plasma morphine concentration, to study the effect of RYGB on morphine pharmacokinetics and to estimate inter-patient variability.
Results
The oral morphine time to maximum plasma concentration (t max) was twofold lower and maximum plasma concentration (C max) was 1.7 times higher at Visit 2, and t max was 7.5 times lower and C max 3.3 times higher at Visit 3 than at Visit 1. The mean oral morphine area under the plasma concentration–time curve (AUC) increased significantly (1.55-fold) between Visits 1 and 3. Changes in body mass index (BMI) after RYGB were clearly associated with decreased apparent oral morphine clearance and apparent central and peripheral morphine volumes of distribution. None of the other anthropometric parameters explained the inter-subject variability in morphine exposure better than BMI.
Conclusion
RYGB and the BMI reduction that followed it dramatically increased the rate of morphine absorption and slightly increased morphine exposure. The dose of immediate-release forms of morphine may be divided in obese patients after RYGB to prevent adverse events due to early and high morphine plasma peaks.



Similar content being viewed by others
References
Obesity and overweight. WHO Fact Sheet no. 311. Available at URL:http://www.who.int/mediacentre/factsheets/fs311/en. Accessed 8 Aug 2014.
American Society for Metabolic and Bariatric Surgery (ASMBS). Final report May 2011. Media Press Kit. Gainesville: American Society for Metabolic and Bariatric Surgery; 2011. http://s3.amazonaws.com/publicASMBS/MediaPressKit/MetabolicBariatricSurgeryOverviewJuly2011.pdf. Accessed 31 Jul 2014.
Padwal R, Brocks D, Sharma AM. A systematic review of drug absorption following bariatric surgery and its theoretical implications. Obes Rev. 2010;11:41–50.
Padwal RS, Ben-Eltriki M, Wang X, Langkaas LA, Sharma AM, Birch DW, et al. Effect of gastric bypass surgery on azithromycin oral bioavailability. J Antimicrob Chemother. 2012;67:2203–6.
Padwal RS, Gabr RQ, Sharma AM, Langkaas LA, Birch DW, Karmali S, et al. Effect of gastric bypass surgery on the absorption and bioavailability of metformin. Diabetes Care. 2011;34:1295–300.
Hamad GG, Helsel JC, Perel JM, Kozak GM, McShea MC, Hughes C, et al. The effect of gastric bypass on the pharmacokinetics of serotonin reuptake inhibitors. Am J Psychiatry. 2012;169:256–63.
Roerig JL, Steffen K, Zimmerman C, Mitchell JE, Crosby RD, Cao L. Preliminary comparison of sertraline levels in postbariatric surgery patients versus matched nonsurgical cohort. Surg Obes Relat Dis. 2012;8:62–6.
Skottheim IB, Stormark K, Christensen H, Jakobsen GS, Hjelmesaeth J, Jenssen T, et al. Significantly altered systemic exposure to atorvastatin acid following gastric bypass surgery in morbidly obese patients. Clin Pharmacol Ther. 2009;86:311–8.
De Smet J, Colin P, De Paepe P, Ruige J, Batens H, Van Nieuwenhove Y, et al. Oral bioavailability of moxifloxacin after Roux-en-Y gastric bypass surgery. J Antimicrob Chemother. 2012;67:226–9.
Skottheim IB, Jakobsen GS, Stormark K, Christensen H, Hjelmesaeth J, Jenssen T, et al. Significant increase in systemic exposure of atorvastatin after biliopancreatic diversion with duodenal switch. Clin Pharmacol Ther. 2010;87:699–705.
Hanley MJ, Abernethy DR, Greenblatt DJ. Effect of obesity on the pharmacokinetics of drugs in humans. Clin Pharmacokinet. 2010;49:71–87.
Brill MJ, Diepstraten J, van Rongen A, van Kralingen S, van den Anker JN, Knibbe CA. Impact of obesity on drug metabolism and elimination in adults and children. Clin Pharmacokinet. 2012;51:277–304.
Hooper MM, Stellato TA, Hallowell PT, Seitz BA, Moskowitz RW. Musculoskeletal findings in obese subjects before and after weight loss following bariatric surgery. Int J Obes (Lond). 2007;31:114–20.
Raebel MA, Newcomer SR, Reifler LM, Boudreau D, Elliott TE, DeBar L, et al. Chronic use of opioid medications before and after bariatric surgery. JAMA. 2013;310:1369–76.
Lloret Linares C, Hajj A, Poitou C, Simoneau G, Clement K, Laplanche JL, et al. Pilot study examining the frequency of several gene polymorphisms involved in morphine pharmacodynamics and pharmacokinetics in a morbidly obese population. Obes Surg. 2011;21:1257–64.
Lloret Linares C, Decleves X, Oppert JM, Basdevant A, Clement K, Bardin C, et al. Pharmacology of morphine in obese patients: clinical implications. Clin Pharmacokinet. 2009;48:635–51.
Hitt HC, McMillen RC, Thornton-Neaves T, Koch K, Cosby AG. Comorbidity of obesity and pain in a general population: results from the Southern Pain Prevalence Study. J Pain. 2007;8:430–6.
Sjostrom L, Narbro K, Sjostrom CD, Karason K, Larsson B, Wedel H, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357:741–52.
Lloret Linares C, Ciangura C, Bouillot JL, Coupaye M, Decleves X, Poitou C, et al. Validity of leg-to-leg bioelectrical impedance analysis to estimate body fat in obesity. Obes Surg. 2011;21:917–23.
Boddy K, King PC, Hume R, Weyers E. The relation of total body potassium to height, weight, and age in normal adults. J Clin Pathol. 1972;25:512–7.
Janmahasatian S, Duffull SB, Ash S, Ward LC, Byrne NM, Green B. Quantification of lean bodyweight. Clin Pharmacokinet. 2005;44:1051–65.
Devine BJ. Gentamicin therapy. Drug Intell Clin Pharm. 1974;8:650–5.
Hull JH, Sarubbi FA Jr. Gentamicin serum concentrations: pharmacokinetic predictions. Ann Intern Med. 1976;85:183–9.
Suter M, Giusti V, Heraief E, Zysset F, Calmes JM. Laparoscopic Roux-en-Y gastric bypass: initial 2-year experience. Surg Endosc. 2003;17:603–9.
Dodet P, Perrot S, Auvergne L, Hajj A, Simoneau G, Decleves X, et al. Sensory impairment in obese patients? sensitivity and pain detection thresholds for electrical stimulation after surgery-induced weight loss, and comparison with a nonobese population. Clin J Pain. 2013;29:43–9.
Chast F, Bardin C, Sauvageon-Martre H, Callaert S, Chaumeil JC. Systemic morphine pharmacokinetics after ocular administration. J Pharm Sci. 1991;80:911–7.
Hirt D, Urien S, Ekouevi DK, Rey E, Arrive E, Blanche S, et al. Population pharmacokinetics of tenofovir in HIV-1-infected pregnant women and their neonates (ANRS 12109). Clin Pharmacol Ther. 2009;85:182–9.
Staahl C, Upton R, Foster DJ, Christrup LL, Kristensen K, Hansen SH, et al. Pharmacokinetic-pharmacodynamic modeling of morphine and oxycodone concentrations and analgesic effect in a multimodal experimental pain model. J Clin Pharmacol. 2008;48:619–31.
Lotsch J, Weiss M, Ahne G, Kobal G, Geisslinger G. Pharmacokinetic modeling of M6G formation after oral administration of morphine in healthy volunteers. Anesthesiology. 1999;90:1026–38.
Dirksen C, Damgaard M, Bojsen-Moller KN, Jorgensen NB, Kielgast U, Jacobsen SH, et al. Fast pouch emptying, delayed small intestinal transit, and exaggerated gut hormone responses after Roux-en-Y gastric bypass. Neurogastroenterol Motil. 2013;25:346-e255.
Steffen KJ, Engel SG, Pollert GA, Li C, Mitchell JE. Blood alcohol concentrations rise rapidly and dramatically after Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2013;9:470–3.
Hasselstrom J, Sawe J. Morphine pharmacokinetics and metabolism in humans. Enterohepatic cycling and relative contribution of metabolites to active opioid concentrations. Clin Pharmacokinet. 1993;24:344–54.
Kharasch ED, Hoffer C, Whittington D, Sheffels P. Role of P-glycoprotein in the intestinal absorption and clinical effects of morphine. Clin Pharmacol Ther. 2003;74:543–54.
Xu J, Kulkarni SR, Li L, Slitt AL. UDP-glucuronosyltransferase expression in mouse liver is increased in obesity- and fasting-induced steatosis. Drug Metab Dispos. 2012;40:259–66.
Congiu M, Mashford ML, Slavin JL, Desmond PV. UDP glucuronosyltransferase mRNA levels in human liver disease. Drug Metab Dispos. 2002;30:129–34.
Lumeng CN, Saltiel AR. Inflammatory links between obesity and metabolic disease. J Clin Invest. 2011;121:2111–7.
Tchernof A, Levesque E, Beaulieu M, Couture P, Despres JP, Hum DW, et al. Expression of the androgen metabolizing enzyme UGT2B15 in adipose tissue and relative expression measurement using a competitive RT-PCR method. Clin Endocrinol (Oxf). 1999;50:637–42.
Ziccardi P, Nappo F, Giugliano G, Esposito K, Marfella R, Cioffi M, et al. Reduction of inflammatory cytokine concentrations and improvement of endothelial functions in obese women after weight loss over one year. Circulation. 2002;105:804–9.
van Kralingen S, Taks M, Diepstraten J, van de Garde EM, van Dongen EP, Wiezer MJ, et al. Pharmacokinetics and protein binding of cefazolin in morbidly obese patients. Eur J Clin Pharmacol. 2011;67:985–92.
Sparreboom A, Wolff AC, Mathijssen RH, Chatelut E, Rowinsky EK, Verweij J, et al. Evaluation of alternate size descriptors for dose calculation of anticancer drugs in the obese. J Clin Oncol. 2007;25:4707–13.
Pai MP, Bearden DT. Antimicrobial dosing considerations in obese adult patients. Pharmacotherapy. 2007;27:1081–91.
Green B, Duffull SB. What is the best size descriptor to use for pharmacokinetic studies in the obese? Br J Clin Pharmacol. 2004;58:119–33.
Chatelut E, Puisset F. The scientific basis of body surface area-based dosing. Clin Pharmacol Ther. 2014;95:359–61.
Drake J, Kirkpatrick CT, Aliyar CA, Crawford FE, Gibson P, Horth CE. Effect of food on the comparative pharmacokinetics of modified-release morphine tablet formulations: oramorph SR and MST Continus. Br J Clin Pharmacol. 1996;41:417–20.
Hasselstrom J, Alexander N, Bringel C, Svensson JO, Sawe J. Single-dose and steady-state kinetics of morphine and its metabolites in cancer patients—a comparison of two oral formulations. Eur J Clin Pharmacol. 1991;40:585–91.
Osborne R, Joel S, Trew D, Slevin M. Morphine and metabolite behavior after different routes of morphine administration: demonstration of the importance of the active metabolite morphine-6-glucuronide. Clin Pharmacol Ther. 1990;47:12–9.
Lotsch J, Zimmermann M, Darimont J, Marx C, Dudziak R, Skarke C, Geisslinger G. Does the A118G polymorphism at the mu-opioid receptor gene protect against morphine-6-glucuronide toxicity? Anesthesiology. 2002;97:814–9.
Skarke C, Darimont J, Schmidt H, Geisslinger G, Lotsch J. Analgesic effects of morphine and morphine-6-glucuronide in a transcutaneous electrical pain model in healthy volunteers. Clin Pharmacol Ther. 2003;73:107–21.
Shibutani K, Inchiosa MA Jr, Sawada K, Bairamian M. Accuracy of pharmacokinetic models for predicting plasma fentanyl concentrations in lean and obese surgical patients: derivation of dosing weight (“pharmacokinetic mass”). Anesthesiology. 2004;101:603–13.
Acknowledgments
We thank Juliette Oliary and Guy Simoneau for help throughout the study. The English text was edited by Dr Owen Parkes. This study was funded in part by the Société d’Evaluation et de Traitement de la Douleur (SFETD-France) and the Groupe Pasteur Mutualité.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
C. Lloret-Linares and D. Hirt contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure. Means and Standard Deviations of observed plasma morphine concentrations versus time before surgery (Visit 1), 8-15 days after surgery (Visit 2), and 6 months after surgery (Visit 3)
Rights and permissions
About this article
Cite this article
Lloret-Linares, C., Hirt, D., Bardin, C. et al. Effect of a Roux-en-Y Gastric Bypass on the Pharmacokinetics of Oral Morphine Using a Population Approach. Clin Pharmacokinet 53, 919–930 (2014). https://doi.org/10.1007/s40262-014-0163-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-014-0163-0