Abstract
Background
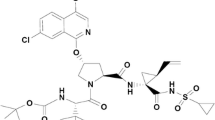
Danoprevir (RG7227) is a potent macrocyclic inhibitor of the hepatitis C virus NS3/4A protease, which is currently in development in combination with low-dose ritonavir for the treatment of chronic hepatitis C infection. Danoprevir is a substrate of cytochrome P450 3A4, and the organic anion transporting polypeptides (OATP) 1B1 and 1B3.
Objective
The objective of this study was to evaluate the effect of a potent OATP inhibitor, ciclosporin, on danoprevir pharmacokinetics, when administered as danoprevir/ritonavir. The effect of danoprevir/ritonavir on ciclosporin pharmacokinetics was also investigated.
Methods
This was a single-dose, randomized, open-label, two-sequence, three-period, crossover study in healthy volunteers. In the first period, subjects were randomized to receive either a single oral dose of danoprevir 100 mg in combination with ritonavir 100 mg or a single oral dose of ciclosporin 100 mg. After a 14-day washout, patients were crossed over to receive the opposite treatment. In period 3, all subjects received the combination of danoprevir/ritonavir and ciclosporin following a 14-day washout from period 2. Blood samples were collected serially with each dose for pharmacokinetic assessment. Pharmacokinetic parameters were estimated using non-compartmental analysis. Geometric mean ratios (GMRs) and 90 % confidence intervals (CIs) were used to compare pharmacokinetic parameters [maximum concentration (C max), area under the concentration–time curve from time zero to infinity (AUC∞), and concentration 12 h post-dose (C 12h)] of danoprevir/ritonavir and ciclosporin when administered alone or in combination. Measures of safety and tolerability were also evaluated.
Results
A total of 18 subjects were enrolled, and 17 completed the study. The C max, AUC∞, and C 12h GMRs (90 % CI) when danoprevir/ritonavir and ciclosporin were co-administered versus danoprevir/ritonavir or ciclosporin alone were 7.22 (5.42–9.62), 13.6 (11.2–16.6), and 22.5 (17.4–29.3), respectively, for danoprevir, 1.97 (1.72–2.27), 2.23 (2.07–2.42), and 2.50 (2.22–2.81), respectively, for ritonavir, and 1.42 (1.29–1.57), 3.65 (3.27–4.08), and 6.15 (5.32–7.11), respectively, for ciclosporin. All treatments were well tolerated, with no laboratory abnormalities, and no clinically significant changes in vital signs, electrocardiograms, or physical examinations observed.
Conclusions
A significant drug–drug interaction was observed between ciclosporin and danoprevir/ritonavir, leading to substantial increases in exposure to danoprevir and a lesser impact on exposure to ritonavir. Therefore, co-administration of danoprevir/ritonavir with potent OATP inhibitors should be undertaken with appropriate precautions.




Similar content being viewed by others
References
Everson GT, Cooper C, Hézode C, et al. High SVR rates with 12 to 24 weeks of ritonavir-boosted danoprevir plus Peg-IFNα-2a (40KD)/RBV in HCV genotype 1 or 4 patients in the DAUPHINE study. Hepatology. 2012;56(4 Suppl):552A.
Gane EJ, Rouzier R, Stedman C, et al. Antiviral activity, safety, and pharmacokinetics of danoprevir/ritonavir plus PEG-IFN α-2a/RBV in hepatitis C patients. J Hepatol. 2011;55(5):972–9.
Reddy MB, Chen Y, Haznedar JO, et al. Impact of low-dose ritonavir on danoprevir pharmacokinetics: results of computer-based simulations and a clinical drug–drug interaction study. Clin Pharmacokinet. 2012;51(7):457–65.
Goelzer P, Morcos PN, Tran J, et al. Coadministration of ritonavir with the HCV protease inhibitor danoprevir substantially reduces reactive metabolite formation both in vitro and in vivo. Hepatology. 2012;56(4 Suppl):580A.
Eley T, Han Y-H, Huang S-P, et al. In vivo and in vitro assessment of asunaprevir as an inhibitor and substrate of OATP transporters in healthy volunteers. Rev Antivir Ther Infect Dis. 2012;6:7.
Park S, Ward W, Beaudet B, et al. In vitro assessment of potential drug–drug interactions between telaprevir and cyclophilin inhibitors in the treatment of chronic hepatitis C. Hepatology. 2011;54(Suppl S1):540A.
Reddy MB, Morcos PN, Le Pogam S, et al. Pharmacokinetic/pharmacodynamic predictors of clinical potency for hepatitis C virus nonnucleoside polymerase and protease inhibitors. Antimicrob Agents Chemother. 2012;56(6):3144–56.
Seiwert SD, Andrews SW, Jiang Y, et al. Preclinical characteristics of the hepatitis C virus NS3/4A protease inhibitor ITMN-191 (R7227). Antimicrob Agents Chemother. 2008;52(12):4432–41.
Portmann R, Meille C, Poirier A. Danoprevir: in vitro studies on hepatic active uptake in human hepatocytes. Nutley: Hoffmann-La Roche Inc; 2012 (data on file).
Wen B, Huang J. In vivo metabolism of danoprevir. Nutley: Hoffmann-La Roche Inc; 2012 (data on file).
Giacomini KM, Huang SM, Tweedie DJ, et al. Membrane transporters in drug development. Nat Rev Drug Discov. 2010;9(3):215–36.
Shitara Y, Maeda K, Ikejiri K, et al. Clinical significance of organic anion transporting polypeptides (OATPs) in drug disposition: their roles in hepatic clearance and intestinal absorption. Biopharm Drug Dispos. 2013;34(1):45–78.
Saeki T, Ueda K, Tanigawara Y, et al. Human P-glycoprotein transports cyclosporin A and FK506. J Biol Chem. 1993;268:6077–80.
Treiber A, Schneiter R, Häusler S, et al. Bosentan is a substrate of human OATP1B1 and OATP1B3: inhibition of hepatic uptake as the common mechanism of its interactions with cyclosporin A, rifampicin, and sildenafil. Drug Metab Dispos. 2007;35(8):1400–7.
Simonson SG, Raza A, Martin PD, et al. Rosuvastatin pharmacokinetics in heart transplant recipients administered an antirejection regimen including cyclosporine. Clin Pharmacol Ther. 2004;76(2):167–77.
Park JW, Siekmeier R, Merz M, et al. Pharmacokinetics of pravastatin in heart-transplant patients taking cyclosporin A. Int J Clin Pharmacol Ther. 2002;40(10):439–50.
Binet I, Wallnöfer A, Weber C, et al. Renal hemodynamics and pharmacokinetics of bosentan with and without cyclosporine A. Kidney Int. 2000;57:224–31.
Neoral® (cyclosporine capsules, USP). US prescribing information. New Jersey: Novartis Pharmaceuticals Corporation; 2012.
Morcos PN, Moreira S, Navarro M, et al. Ritonavir-boosted danoprevir (DNVr) may be co-administered without regard to meals or in combination with ranitidine or omeprazole: results of a healthy volunteer pharmacokinetic study. Rev Antiviral Ther Infect Dis. 2012;6:7.
Kalliokoski A, Niemi M. Impact of OATP transporters on pharmacokinetics. Br J Pharmacol. 2009;158(3):693–705.
Ding R, Tayrouz Y, Reidel KD, et al. Substantial pharmacokinetic interaction between digoxin and ritonavir in healthy volunteers. Clin Pharmacol Ther. 2004;76(1):73–84.
Vaidyanathan S, Camenisch G, Schuetz H, et al. Pharmacokinetics of the oral direct renin inhibitor aliskiren in combination with digoxin, atorvastatin, and ketoconazole in healthy subjects: the role of P-glycoprotein in the disposition of aliskiren. J Clin Pharmacol. 2008;48(11):1323–38.
Keogh J, Kunta J. Development, validation and utility of an in vitro technique for assessment of potential clinical drug–drug interactions involving P-glycoprotein. Eur J Pharm Sci. 2006;27(5):543–54.
Reddy MB, Connor A, Brennan BJ, et al. Physiological modeling and assessments of regional drug bioavailability of danoprevir to determine whether a controlled release formulation is feasible. Biopharm Drug Dispos. 2011;32(5):261–75.
Asberg A, Hartmann A, Fjeldsa E, et al. Bilateral pharmacokinetic interaction between cyclosporine A and atorvastatin in renal transplant recipients. Am J Transplant. 2001;1(4):382–6.
Olbricht C, Wanner C, Eisenhauer T, et al. Accumulation of lovastatin, but not pravastatin, in the blood of cyclosporine-treated kidney graft patients after multiple doses. Clin Pharmacol Ther. 1997;62(3):311–21.
Regazzi MB, Iacona I, Campana C, et al. Altered disposition of pravastatin following concomitant drug therapy with cyclosporin A in transplant recipients. Transplant Proc. 1993;25(4):2732–4.
Drewe J, Gutmann H, Fricker G, et al. HIV protease inhibitor ritonavir: a more potent inhibitor of P-glycoprotein than the cyclosporine analog SDZ PSC 833. Biochem Pharmacol. 1999;57:1147–52.
Morcos PN, Chang L, Zhang Y, et al. Danoprevir (DNV) does not change effects of ritonavir (RTV) on the PK of CYP3A substrate midazolam (MDZ) and CYP2C9 substrate warfarin (WAR). Rev Antiviral Ther Infect Dis. 2011;6:4.
Gomez DY, Wacher VJ, Tomlanovich SJ, et al. The effects of ketoconazole on the intestinal metabolism and bioavailability of cyclosporine. Clin Pharmacol Ther. 1995;58(1):15–9.
Hsu A, Granneman GR, Bertz RJ. Ritonavir. Clinical pharmacokinetics and interactions with other anti-HIV agents. Clin Pharmacokinet. 1998;35(4):275–91.
Vogel M, Voigt E, Michaelis HC, et al. Management of drug-to-drug interactions between cyclosporine A and the protease-inhibitor lopinavir/ritonavir in liver-transplanted HIV-infected patients. Liver Transplant. 2004;10(7):939–44.
Guaraldi G, Cocchi S, Codeluppi M, et al. Pharmacokinetic interaction between amprenavir/ritonavir and FosAmprenavir on cyclosporine in two patients with human immunodeficiency virus infection undergoing orthotopic liver transplantation. Transplant Proc. 2006;38(4):1138–40.
Acknowledgments
Patrick Smith was employed by Hoffmann-La Roche Inc, Nutley, NJ, USA, when the present study was conducted. This study was supported by F. Hoffmann-La Roche Ltd. Support for third-party writing assistance for this manuscript, furnished by Sian Deeves, Ph.D., was provided by F. Hoffmann-La Roche Ltd.
Author contributions
BB designed the research, performed analysis and interpretation of data, and critically reviewed and revised the manuscript for intellectual content. SM wrote the manuscript, designed and performed the research, performed acquisition, analysis, and interpretation of data, and critically reviewed and revised the manuscript for intellectual content. PM, MN, and JA performed analysis and interpretation of data and critically reviewed and revised the manuscript for intellectual content. PG was responsible for the nonclinical study concept and design, and critically reviewed and revised the manuscript for intellectual content. PW performed analysis and interpretation of data and critical revision of the manuscript for important intellectual content. PS designed the research, performed analysis and interpretation of data, and critically reviewed and revised the manuscript for intellectual content.
Conflict of interest
All authors are employees of Hoffmann-La Roche Inc or Genentech.
Author information
Authors and Affiliations
Corresponding author
Additional information
ClinicalTrials.gov: NCT01514968.
Rights and permissions
About this article
Cite this article
Brennan, B.J., Moreira, S.A., Morcos, P.N. et al. Pharmacokinetics of a Three-Way Drug Interaction Between Danoprevir, Ritonavir and the Organic Anion Transporting Polypeptide (OATP) Inhibitor Ciclosporin. Clin Pharmacokinet 52, 805–813 (2013). https://doi.org/10.1007/s40262-013-0077-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-013-0077-2